|
Uronic Acids
300px, The glucose.html" ;"title="Fischer projections of D-glucose">Fischer projections of D-glucose (left) and D-glucuronic acid (right). Glucose's terminal carbon's primary alcohol group has been oxidized to a carboxylic acid. Uronic acids () or alduronic acids are a class of sugar acids with both carbonyl and carboxylic acid functional groups. They are sugars in which the hydroxyl group furthest from the carbonyl group has been oxidized to a carboxylic acid. Usually the sugar is an aldose, but fructuronic acid also occurs. Oxidation of the terminal aldehyde instead yields an aldonic acid, while oxidation of both the terminal hydroxyl group and the aldehyde yields an aldaric acid. The names of uronic acids are generally based on their parent sugars, for example, the uronic acid analog of glucose is glucuronic acid. Uronic acids derived from hexoses are known as hexuronic acids and uronic acids derived from pentoses are known as penturonic acids. Examples Some of these ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Beta D-Glucuronic Acid
Beta (, ; uppercase , lowercase , or cursive Greek, cursive ; or ) is the second letter of the Greek alphabet. In the system of Greek numerals, it has a value of 2. In Ancient Greek, beta represented the voiced bilabial plosive . In Modern Greek, it represents the voiced bilabial fricative while in borrowed words is instead commonly transcribed as μπ. Letters that arose from beta include the Roman letter and the Cyrillic letters and . Name Like the names of most other Greek letters, the name of beta was adopted from the acrophony, acrophonic name of the corresponding letter in Phoenician alphabet, Phoenician, which was the common Semitic languages, Semitic word ('house', compare and ). In Greek, the name was , pronounced in Ancient Greek. It is spelled in modern monotonic orthography and pronounced . History The letter beta was derived from the Phoenician alphabet, Phoenician letter Bet (letter), beth . The letter Β had the largest number of highly divergent lo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Aldaric Acid
Aldaric acids are a group of sugar acids, where the terminal hydroxyl and carbonyl groups of the sugars have been replaced by terminal carboxylic acids, and are characterised by the formula HO2C-(CHOH)n-CO2H. Oxidation of just the aldehyde yields an aldonic acid while oxidation of just the terminal hydroxyl group yields an uronic acid.) Aldaric acids cannot form cyclic hemiacetals like unoxidized sugars, but they can sometimes form lactones. Synthesis and reactions Aldaric acids are usually synthesized by the oxidation of aldoses with nitric acid. In this reaction it is the open-chain (polyhydroxyaldehyde) form of the sugar that reacts. These compounds are of interest as bio-derived chemicals including to bio-derived polyesters. Structure Nomenclature of the aldaric acids is based on the sugars from which they are derived; for example, glucose is oxidized to glucaric acid and xylose to xylaric acid. Unlike their parent sugars, aldaric acids have the same functional group at ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Uronic Acids
300px, The glucose.html" ;"title="Fischer projections of D-glucose">Fischer projections of D-glucose (left) and D-glucuronic acid (right). Glucose's terminal carbon's primary alcohol group has been oxidized to a carboxylic acid. Uronic acids () or alduronic acids are a class of sugar acids with both carbonyl and carboxylic acid functional groups. They are sugars in which the hydroxyl group furthest from the carbonyl group has been oxidized to a carboxylic acid. Usually the sugar is an aldose, but fructuronic acid also occurs. Oxidation of the terminal aldehyde instead yields an aldonic acid, while oxidation of both the terminal hydroxyl group and the aldehyde yields an aldaric acid. The names of uronic acids are generally based on their parent sugars, for example, the uronic acid analog of glucose is glucuronic acid. Uronic acids derived from hexoses are known as hexuronic acids and uronic acids derived from pentoses are known as penturonic acids. Examples Some of these ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Isosaccharinic Acid
Isosaccharinic acid (ISA) is a six-carbon sugar acid which is formed by the action of calcium hydroxide on lactose and other carbohydrates. It is of interest because it may form in intermediate-level nuclear waste stores when cellulose is degraded by the calcium hydroxide in cements such as Portland cement. The calcium salt of the alpha form of ISA is very crystalline and quite insoluble in cold water, but in hot water it is soluble. ISA is thought to form by means of a series of reactions in which calcium ions acting as lewis acids catalyze two of the three steps. The first step is likely to be a rearrangement of the reducing sugar end of the cellulose (or lactose) into a keto sugar, the second step is likely to be a reaction similar to the base catalyzed dehydration which often occurs after an aldol reaction. In this second step an alkoxide (derived from a sugar) takes the role of the hydroxide leaving group, this second step is not likely to require the lewis acidity of the ca ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Glucuronic Acid
Glucuronic acid (GCA, from ) is a uronic acid that was first isolated from urine (hence the name "uronic acid"). It is found in many natural gum, gums such as gum arabic ( 18%), xanthan, and kombucha tea and is important for the metabolism of microorganisms, plants and animals. Properties Glucuronic acid is a sugar acid derived from glucose, with its sixth carbon atom oxidized to a carboxylic acid. In living beings, this primary oxidation occurs with Uridine diphosphate glucose, UDP-α-D-glucose (UDPG), not with the free sugar. Glucuronic acid, like its precursor glucose, can exist as a linear (carboxo-)aldohexose ( 60,000 are too large for renal excretion and will be excreted with bile into the intestine. Neonates are deficient in this conjugating system, making them particularly vulnerable to drugs such as chloramphenicol, which is inactivated by the addition of glucuronic acid, resulting in gray baby syndrome. Bilirubin is excreted in the bile as bilirubin diglucuronid ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Gluconic Acid
Gluconic acid is an organic compound with molecular formula C6H12O7 and condensed structural formula HOCH2(CHOH)4CO2H. A white solid, it forms the gluconate anion in neutral aqueous solution. The salts of gluconic acid are known as "gluconates". Gluconic acid, gluconate salts, and gluconate esters occur widely in nature because such species arise from the oxidation of glucose. Some drugs are injected in the form of gluconates. Chemical structure The chemical structure of gluconic acid consists of a six-carbon chain, with five hydroxyl groups positioned in the same way as in the open-chained form of glucose, terminating in a carboxylic acid group. It is one of the 16 stereoisomers of 2,3,4,5,6-pentahydroxyhexanoic acid. Production Gluconic acid is typically produced by the aerobic oxidation of glucose in the presence of the enzyme glucose oxidase. The conversion produces gluconolactone and hydrogen peroxide. The lactone spontaneously hydrolyzes to gluconic acid in water. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Proteoglycans
Proteoglycans are proteins that are heavily glycosylation, glycosylated. The basic proteoglycan unit consists of a "core protein" with one or more covalent bond, covalently attached glycosaminoglycan (GAG) chain(s). The point of attachment is a serine (Ser) residue to which the glycosaminoglycan is joined through a tetrasaccharide bridge (e.g. chondroitin sulfate-GlcA-Galactose, Gal-Gal-Xylose, Xyl-PROTEIN). The Ser residue is generally in the sequence -Ser-Gly-X-Gly- (where X can be any amino acid residue but proline), although not every protein with this sequence has an attached glycosaminoglycan. The chains are long, linear carbohydrate polymers that are negatively charged under physiological conditions due to the occurrence of sulfate and uronic acid groups. Proteoglycans occur in connective tissue. Types Proteoglycans are categorized by their relative size (large and small) and the nature of their glycosaminoglycan chains. Types include: Certain members are considered m ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Iduronic Acid
-Iduronic acid (IUPAC abbr.: IdoA) is the major uronic acid component of the glycosaminoglycans (GAGs) dermatan sulfate, and heparin. It is also present in heparan sulfate, although here in a minor amount relative to its carbon-5 epimer glucuronic acid. IdoA is a pyranose sugar. Most pyranoses are stable in one of two chair conformations 1C4 or 4C1. -iduronate is different and adopts more than one solution conformation, with an equilibrium existing between three low-energy conformers. These are the 1C4 and 4C1 chair forms and an additional 2S0 skew-boat conformation. left, α--idopyranuronic acid, 120px IdoA may be modified by the addition of an ''O''-sulfate group at carbon position 2 to form 2-''O''-sulfo--iduronic acid (IdoA2S). In 2000, LK Hallak described the importance of this sugar in respiratory syncytial virus (RSV) infection. Dermatan sulfate and heparan sulfate were the only GAGs containing IdoA, and they were the only ones that inhibited RSV infection in cell cul ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Glucuronate
Glucuronic acid (GCA, from ) is a uronic acid that was first isolated from urine (hence the name "uronic acid"). It is found in many gums such as gum arabic ( 18%), xanthan, and kombucha tea and is important for the metabolism of microorganisms, plants and animals. Properties Glucuronic acid is a sugar acid derived from glucose, with its sixth carbon atom oxidized to a carboxylic acid. In living beings, this primary oxidation occurs with UDP-α-D-glucose (UDPG), not with the free sugar. Glucuronic acid, like its precursor glucose, can exist as a linear (carboxo-)aldohexose (<1%), or as a cyclic ( or ) ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pentose
In chemistry, a pentose is a monosaccharide (simple sugar) with five carbon atoms. The chemical formula of many pentoses is , and their molecular weight is 150.13 g/mol.-Ribose . PubChem compound webpage, accessed on 2010-02-06. Pentoses are very important in . Ribose is a constituent of , and the related molecule, deoxyribose, is a constituent of DNA. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hexose
In chemistry, a hexose is a monosaccharide (simple sugar) with six carbon atoms. The chemical formula for all hexoses is , and their molecular weight is 180.156 g/mol. Hexoses exist in two forms, open-chain or cyclic, that easily convert into each other in aqueous solutions. The open-chain form of a hexose, which usually is favored in solutions, has the general structure , where ''n'' is 1, 2, 3, 4, 5. Namely, five of the carbons have one hydroxyl functional group () each, connected by a single bond, and one has an oxo group (), forming a carbonyl group (). The remaining bonds of the carbon atoms are satisfied by seven hydrogen atoms. The carbons are commonly numbered 1 to 6 starting at the end closest to the carbonyl. Hexoses are extremely important in biochemistry, both as isolated molecules (such as glucose and fructose) and as building blocks of other compounds such as starch, cellulose, and glycosides. Hexoses can form dihexose (like sucrose) by a condensation re ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
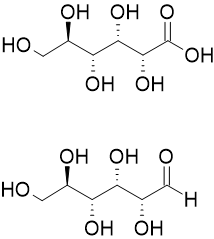
Aldonic Acid
Aldonic acids are sugar acids with the general chemical formula, HO2C(CHOH)nCH2OH. They are obtained by oxidizing the aldehyde (-CHO group) of an aldose to form a carboxylic acid (-COOH group). Aldonic acids are generally found in their ring form. However, these rings do not have a chiral carbon at the terminal end bearing the aldehyde, and they cannot form R−O−R′ linkages between different molecules. The nomenclature of aldonic acids and their lactones is based on replacing the suffix "-ose" with "onic acid" or "onolactone". Hence, D-glucose is oxidized to D-gluconic acid and D- gluconolactone. Inventory Sugar acids are white, water-soluble solids. They tend to dehydrate to the lactone derivative, often before they can be melted. All are chiral and, at least in nature, enantiopure. Synthesis Oxidation by bromine and water Aldonic acids are most commonly prepared by the oxidation of the sugar with bromine and water under neutral pH. Strecker reaction Alternatively ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |