|
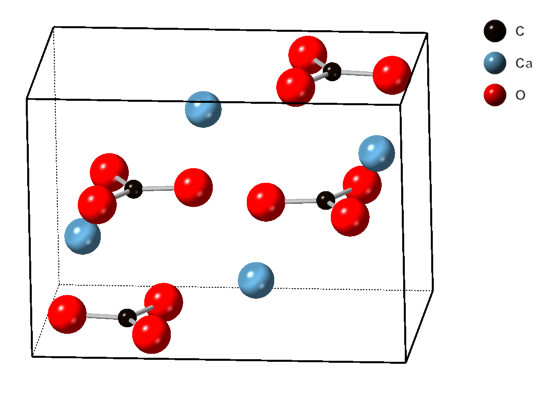
Vaterite
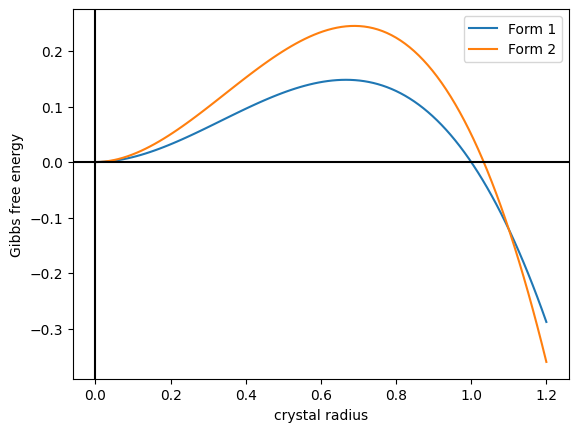
Vaterite is a mineral, a polymorphism (materials science), polymorph of calcium carbonate (calcium, Cacarbon, Coxygen, O3). It was named after the German mineralogist Heinrich Vater. It is also known as mu-calcium carbonate (μ-CaCO3). Vaterite belongs to the hexagonal crystal system, whereas calcite is trigonal and aragonite is orthorhombic. Vaterite, like aragonite, is a metastable phase of calcium carbonate at ambient conditions at the surface of the Earth. As it is less stable than either calcite, the most stable polymorph, or aragonite, vaterite has a higher solubility than either of these phases. Therefore, once vaterite is exposed to water, it converts to calcite (at low temperature) or aragonite (at high temperature: ~60 °C). At 37 °C for example a solution-mediated transition from vaterite to calcite occurs, where the vaterite dissolves and subsequently precipitates as calcite assisted by an Ostwald ripening process. However, vaterite does occur naturally in ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Calcium Carbonate
Calcium carbonate is a chemical compound with the chemical formula . It is a common substance found in Rock (geology), rocks as the minerals calcite and aragonite, most notably in chalk and limestone, eggshells, gastropod shells, shellfish skeletons and pearls. Materials containing much calcium carbonate or resembling it are described as calcareous. Calcium carbonate is the active ingredient in agricultural lime and is produced when calcium ions in hard water react with carbonate ions to form limescale. It has medical use as a calcium supplement or as an antacid, but excessive consumption can be hazardous and cause hypercalcemia and digestive issues. Chemistry Calcium carbonate shares the typical properties of other carbonates. Notably, it: *reacts with acids, releasing carbonic acid which quickly disintegrates into carbon dioxide and water: : *releases carbon dioxide upon heating, called a thermal decomposition reaction, or calcination (to above 840 °C in the case of ), t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Calcite
Calcite is a Carbonate minerals, carbonate mineral and the most stable Polymorphism (materials science), polymorph of calcium carbonate (CaCO3). It is a very common mineral, particularly as a component of limestone. Calcite defines hardness 3 on the Mohs scale of mineral hardness, based on Scratch hardness, scratch hardness comparison. Large calcite crystals are used in optical equipment, and limestone composed mostly of calcite has numerous uses. Other polymorphs of calcium carbonate are the minerals aragonite and vaterite. Aragonite will change to calcite over timescales of days or less at temperatures exceeding 300 °C, and vaterite is even less stable. Etymology Calcite is derived from the German , a term from the 19th century that came from the Latin word for Lime (material), lime, (genitive ) with the suffix ''-ite'' used to name minerals. It is thus a Doublet (linguistics), doublet of the word ''wikt:chalk, chalk''. When applied by archaeology, archaeologists and ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Aragonite
Aragonite is a carbonate mineral and one of the three most common naturally occurring crystal forms of calcium carbonate (), the others being calcite and vaterite. It is formed by biological and physical processes, including precipitation from marine and freshwater environments. The crystal lattice of aragonite differs from that of calcite, resulting in a different crystal shape, an orthorhombic crystal system with acicular crystal. Repeated twinning results in pseudo-hexagonal forms. Aragonite may be columnar or fibrous, occasionally in branching helictitic forms called ''flos-ferri'' ("flowers of iron") from their association with the ores at the Carinthian iron mines. Occurrence The type location for aragonite is Molina de Aragón in the Province of Guadalajara in Castilla-La Mancha, Spain, for which it was named in 1797. Aragonite is found in this locality as cyclic twins inside gypsum and marls of the Keuper facies of the Triassic. This type of aragoni ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Polymorphism (materials Science)
In crystallography, polymorphism is the phenomenon where a compound or element can crystallize into more than one crystal structure. The preceding definition has evolved over many years and is still under discussion today. Discussion of the defining characteristics of polymorphism involves distinguishing among types of transitions and structural changes occurring in polymorphism versus those in other phenomena. Overview Phase transitions (phase changes) that help describe polymorphism include polymorphic transitions as well as melting and vaporization transitions. According to IUPAC, a polymorphic transition is "A reversible transition of a solid crystalline phase at a certain temperature and pressure (the inversion point) to another phase of the same chemical composition with a different crystal structure." Additionally, Walter McCrone described the phases in polymorphic matter as "different in crystal structure but identical in the liquid or vapor states." McCrone also def ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ikaite
Ikaite is the mineral name for the hexahydrate of calcium carbonate, . Ikaite tends to form very steep or spiky pyramidal crystals, often radially arranged, of varied sizes from thumbnail size aggregates to gigantic salient spurs. It is only found in a metastable state and decomposes rapidly by losing most of its water content once removed from near-freezing water. This "melting mineral" is more commonly known through its pseudomorphs. Distribution It is usually considered a rare mineral, but this is likely due to difficulty in preserving samples. It was first discovered in nature by the Danish mineralogist Pauly in the Ikka Fjord in southwest Greenland, close to Ivittuut, the locality of the famous cryolite deposit. Here ikaite occurs in truly spectacular towers or columns (up to tall) growing out of the fjord floor towards the surface water, where they are naturally truncated by waves, or unnaturally by the occasional boat. At the Ikka Fjord, it is supposed that the ikai ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Monohydrocalcite
Monohydrocalcite is a mineral that is a hydrous form of calcium carbonate, CaCO3·H2O. It was formerly also known by the name hydrocalcite, which is now discredited by the IMA. It is a trigonal mineral which is white when pure. Monohydrocalcite is not a common rock-forming mineral, but is frequently associated with other calcium and magnesium carbonate minerals, such as calcite, aragonite, lansfordite, and nesquehonite. Monohydrocalcite has been observed in air conditioning systems, and in moonmilk deposits in caves, both probably formed from spray of carbonate rich fluids. It is well known in Robe on the Limestone Coast of South Australia as a component of beach sands of Lake Fellmongery and Lake Butler, where it is believed to be formed from algal spume. Other lacustrine deposits include Lake Issyk-Kul, Kyrgyzstan, Lake Kivu, Democratic Republic of the Congo, and Solar Lake, Sinai. It has been reported as a significant component of the decomposition of ikaite in th ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Metal
A metal () is a material that, when polished or fractured, shows a lustrous appearance, and conducts electrical resistivity and conductivity, electricity and thermal conductivity, heat relatively well. These properties are all associated with having electrons available at the Fermi level, as against nonmetallic materials which do not. Metals are typically ductile (can be drawn into a wire) and malleable (can be shaped via hammering or pressing). A metal may be a chemical element such as iron; an alloy such as stainless steel; or a molecular compound such as polythiazyl, polymeric sulfur nitride. The general science of metals is called metallurgy, a subtopic of materials science; aspects of the electronic and thermal properties are also within the scope of condensed matter physics and solid-state chemistry, it is a multidisciplinary topic. In colloquial use materials such as steel alloys are referred to as metals, while others such as polymers, wood or ceramics are nonmetallic ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Saxifraga
''Saxifraga'' is the largest genus in the family Saxifragaceae, containing about 473 species of holarctic perennial plants, known as saxifrages or rockfoils. The Latin word ''saxifraga'' means literally "stone-breaker", from Latin ' ("rock" or "stone") + ' ("to break"). It is usually thought to indicate a medicinal use for treatment of urinary calculi (known as kidney or bladder stones), rather than breaking rocks apart. Description Most saxifrages are small perennial, biennial (e.g. '' S. adscendens'') or annual (e.g. '' S. tridactylites'') herbaceous plants whose basal or cauline leaves grow close to the ground, often in a rosette. The leaves typically have a more or less incised margin; they may be succulent, needle-like and/or hairy, reducing evaporation. The inflorescence or single flower clusters rise above the main plant body on naked stalks. The small actinomorphic hermaphrodite flowers have five petals and sepals and are usually white, but red to yellow in some spe ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cambridge University Botanic Garden
The Cambridge University Botanic Garden is a botanical garden located in Cambridge, England, associated with the university Department of Plant Sciences, University of Cambridge, Department of Plant Sciences (formerly Botany School). It lies between Trumpington Road to the west, Bateman Street to the north and Hills Road, Cambridge, Hills Road to the east. The garden covers an area of 16 hectares (40 acres). The site is almost entirely on level ground and in addition to its scientific value, the garden is highly rated by gardening enthusiasts. It holds a plant collection of over 8,000 plant species from all over the world to facilitate teaching and research. The garden was created for the University of Cambridge in 1831 by Professor John Stevens Henslow (Charles Darwin's mentor) and was opened to the public in 1846. The United Kingdom weather records, second-highest temperature recorded in the UK, 38.7 °C (101.7 °F), was recorded on 2019 European heatwaves, 25 July ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Carbonate Minerals
Carbonate minerals are those minerals containing the carbonate ion, . Carbonate divisions Anhydrous carbonates *Calcite group: trigonal **Calcite CaCO3 **Gaspéite (Ni,Mg,Fe2+)CO3 **Magnesite MgCO3 **Otavite CdCO3 **Rhodochrosite MnCO3 **Siderite FeCO3 **Smithsonite ZnCO3 **Spherocobaltite CoCO3 *Aragonite group: orthorhombic **Aragonite CaCO3 **Cerussite PbCO3 **Strontianite SrCO3 **Witherite BaCO3 **Rutherfordine UO2CO3 **Natrite Na2CO3 Anhydrous carbonates with compound formulas *Dolomite group: trigonal **Ankerite CaFe(CO3)2 **Dolomite (mineral), Dolomite CaMg(CO3)2 **Huntite Mg3Ca(CO3)4 **Minrecordite CaZn(CO3)2 **Barytocalcite BaCa(CO3)2 Carbonates with hydroxyl or halogen *Carbonate with hydroxide: monoclinic **Azurite Cu3(CO3)2(OH)2 **Hydrocerussite Pb3(CO3)2(OH)2 **Malachite Cu2CO3(OH)2 **Rosasite (Cu,Zn)2CO3(OH)2 **Phosgenite Pb2(CO3)Cl2 **Hydrozincite Zn5(CO3)2(OH)6 **Aurichalcite (Zn,Cu)5(CO3)2(OH)6 Hydrated carbonates *Hydromagnesite Mg5(CO3)4(OH)2.4H2O *Ikaite ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Corals
Corals are colonial marine invertebrates within the subphylum Anthozoa of the phylum Cnidaria. They typically form compact Colony (biology), colonies of many identical individual polyp (zoology), polyps. Coral species include the important Coral reef, reef builders that inhabit tropical oceans and secrete calcium carbonate to form a hard skeleton. A coral "group" is a colony of very many cloning, genetically identical polyps. Each polyp is a sac-like animal typically only a few millimeters in diameter and a few centimeters in height. A set of tentacles surround a central mouth opening. Each polyp excretes an exoskeleton near the base. Over many generations, the colony thus creates a skeleton characteristic of the species which can measure up to several meters in size. Individual colonies grow by asexual reproduction of polyps. Corals also breed sexually by spawning: polyps of the same species release gametes simultaneously overnight, often around a full moon. Fertilized eggs form ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |