|
Diazonium
Diazonium compounds or diazonium salts are a group of organic compounds sharing a common functional group where R can be any organic group, such as an alkyl or an aryl, and X is an inorganic or organic anion, such as a halide. The parent, compound where R is hydrogen, is diazenylium. Structure and general properties Arene derivatives According to X-ray crystallography the linkage is linear in typical diazonium salts. The bond distance in benzenediazonium tetrafluoroborate is 1.083(3) Å, which is almost identical to that for dinitrogen molecule (N≡N). The linear free energy constants σm and σp indicate that the diazonium group is strongly electron-withdrawing. Thus, the diazonio-substituted phenols and benzoic acids have greatly reduced p''K''a values compared to their unsubstituted counterparts. The p''K''a of phenolic proton of 4-hydroxybenzenediazonium is 3.4, versus 9.9 for phenol itself. In other words, the diazonium group raises the ionization constant ''K''a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Azo Coupling
In organic chemistry, an azo coupling is an organic reaction, reaction between a diazonium compound () and another aromatic compound that produces an azo compound (). In this electrophilic aromatic substitution reaction, the aryldiazonium cation is the electrophile, and the activating group, activated carbon (usually from an arene, which is called coupling agent), serves as a nucleophile. Classical coupling agents are phenols and naphthols. Usually the diazonium reagent attacks at the para position of the coupling agent. When the para position is occupied, coupling occurs at a ortho position, albeit at a slower rate. Uses of the reaction Aromatic azo compounds tend to be brightly colored due to their extended Conjugated system, conjugated systems. Many are useful dyes (see azo dye). Important azo dyes include methyl red and pigment red 170. Azo printing exploits this reaction as well. In this case, the diazonium ion is degraded by light, leaving a latent image in undegrade ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Benzenediazonium Cation
Benzenediazonium tetrafluoroborate is an organic compound with the formula 6H5N2F4. It is a salt of a diazonium cation and tetrafluoroborate. It exists as a colourless solid that is soluble in polar solvents. It is the parent member of the aryldiazonium compounds, which are widely used in organic chemistry. Synthesis Diazotization of aniline: : C6H5NH2 + HNO2 (from NaNO2 and HCl) + HCl → 6H5N2l + 2 H2O The tetrafluoroborate can be obtained from crude benzenediazonium chloride by salt metathesis using tetrafluoroboric acid. : 6H5N2l + HBF4 → 6H5N2F4 + HCl The tetrafluoroborate is more stable than the chloride. Properties The diazo group (N2) can be replaced by many other groups, usually anions, giving a variety of substituted phenyl derivatives: :C6H5N2+ + Nu− → C6H5Nu + N2 These transformations are associated with many named reactions including the Schiemann reaction, Sandmeyer reaction, and Gomberg–Bachmann reaction. A wide range of groups that can b ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Benzenediazonium Tetrafluoroborate
Benzenediazonium tetrafluoroborate is an organic compound with the formula 6H5N2F4. It is a salt of a diazonium cation and tetrafluoroborate. It exists as a colourless solid that is soluble in polar solvents. It is the parent member of the aryldiazonium compounds, which are widely used in organic chemistry. Synthesis Diazotization of aniline: : C6H5NH2 + HNO2 (from NaNO2 and HCl) + HCl → 6H5N2l + 2 H2O The tetrafluoroborate can be obtained from crude benzenediazonium chloride by salt metathesis using tetrafluoroboric acid. : 6H5N2l + HBF4 → 6H5N2F4 + HCl The tetrafluoroborate is more stable than the chloride. Properties The diazo group (N2) can be replaced by many other groups, usually anions, giving a variety of substituted phenyl derivatives: :C6H5N2+ + Nu− → C6H5Nu + N2 These transformations are associated with many named reactions including the Schiemann reaction, Sandmeyer reaction, and Gomberg–Bachmann reaction. A wide range of groups that can be us ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
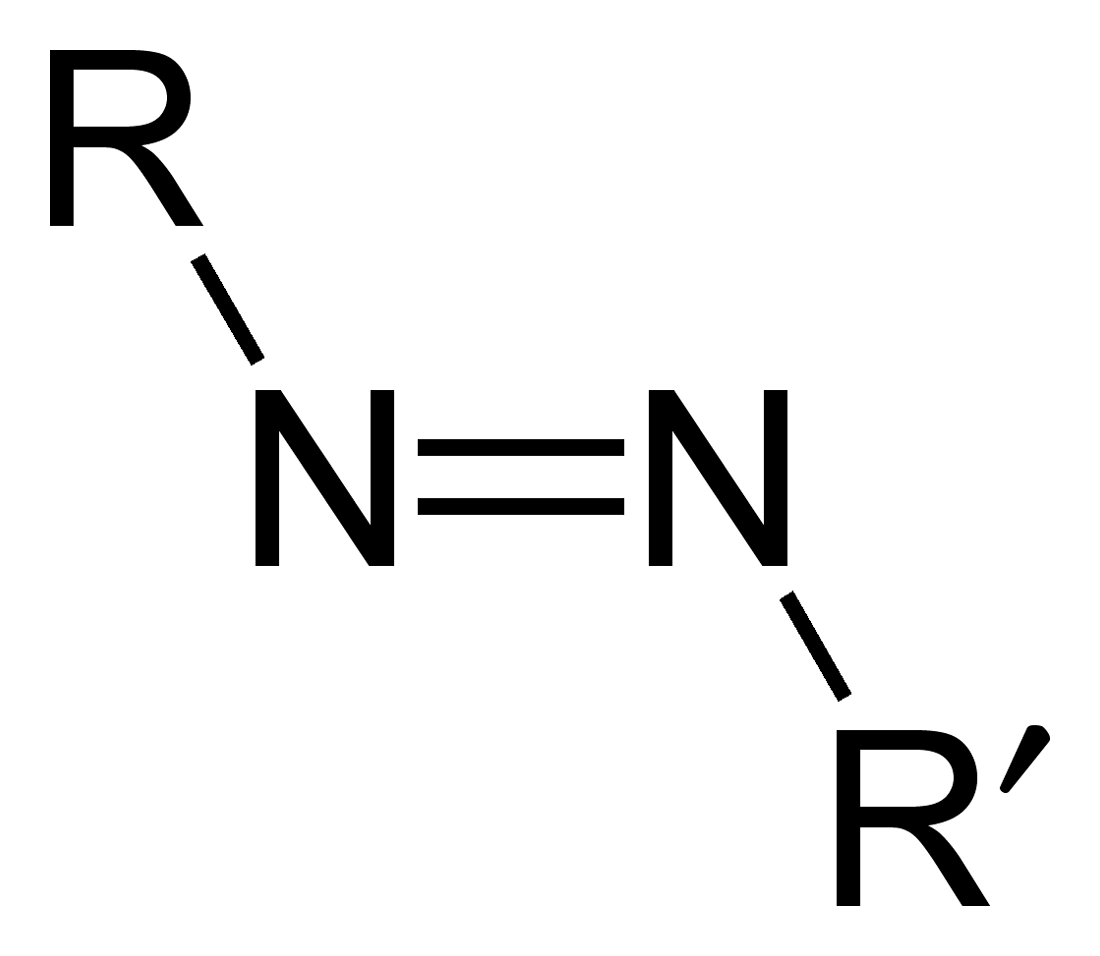
Azene
Azo compounds are organic compounds bearing the functional group diazenyl (, in which R and R′ can be either aryl or alkyl groups). IUPAC defines azo compounds as: "Derivatives of diazene (diimide), , wherein both hydrogens are substituted by hydrocarbyl groups, e.g. azobenzene or diphenyldiazene.", where Ph stands for phenyl group. The more stable derivatives contain two aryl groups. The group is called an ''azo group'' (, ). Many textile and leather articles are dyed with azo dyes and pigments. Aryl azo compounds urinary tract infections">Phenazopyridine, an aryl azo compound, is used to treat urinary tract infections">150px Aryl azo compounds are usually stable, crystalline species. Azobenzene is the prototypical aromatic azo compound. It exists mainly as the Cis-trans isomerism, ''trans'' isomer, but upon illumination, converts to the Cis-trans isomerism, ''cis'' isomer. Aromatic azo compounds can be synthesized by azo coupling, which entails an electrophilic subst ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nitrous Acid
Nitrous acid (molecular formula ) is a weak and monoprotic acid known only in solution, in the gas phase, and in the form of nitrite () salts. It was discovered by Carl Wilhelm Scheele, who called it " phlogisticated acid of niter". Nitrous acid is used to make diazonium salts from amines. The resulting diazonium salts are reagents in azo coupling reactions to give azo dyes. Structure In the gas phase, the planar nitrous acid molecule can adopt both a ''syn'' and an ''anti'' form. The ''anti'' form predominates at room temperature, and IR measurements indicate it is more stable by around 2.3 kJ/mol. p. 462. Image:Trans-nitrous-acid-2D-dimensions.png , Dimensions of the ''anti'' form(from the microwave spectrum) Image:Trans-nitrous-acid-3D-balls.png , Model of the ''anti'' form Image:Cis-nitrous-acid-3D-balls.png , ''syn'' form Preparation and decomposition Free, gaseous nitrous acid is unstable, rapidly disproportionating to nitric oxides: :2 HNO2 → NO2 + ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Methyldiazonium
Methyldiazonium is an organic compound consisting of a methyl group attached to a diazo group. This cation is the conjugate acid of diazomethane, with an estimated p''K''a<10. : 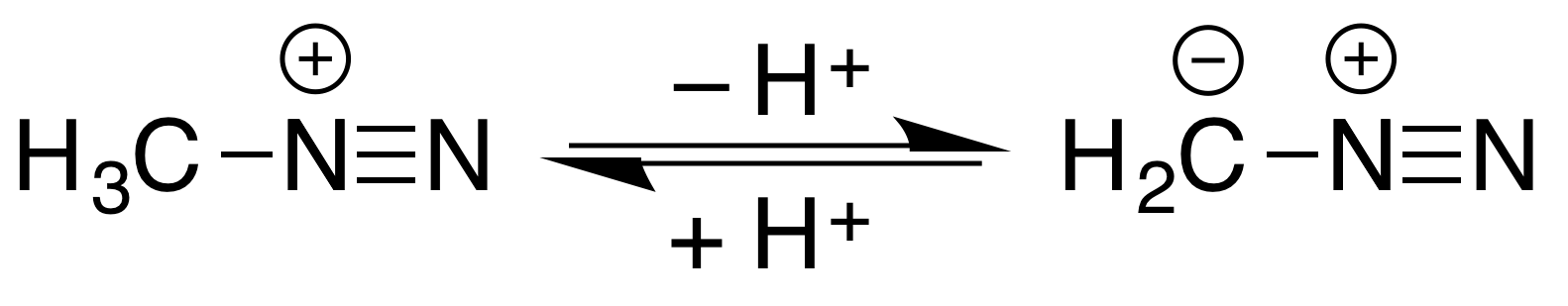 It is an intermediate in methylation reactions of diazomethane with acidic hydroxyl compounds, such as conversion of s to methyl esters and
It is an intermediate in methylation reactions of diazomethane with acidic hydroxyl compounds, such as conversion of s to methyl esters and
|
Aryl Radical
An aryl radical in organic chemistry is a reactive intermediate and an arene compound incorporating one free radical carbon atom as part of the ring structure. As such it is the radical counterpart of the arenium ion. The parent compound is the phenyl radical .''Minireview Intermolecular Olefin Functionalisation Involving Aryl Radicals Generated from Arenediazonium Salts'' Markus R. Heinrich Chemistry - A European Journal 2008 volume 15 issue 4, pages 820 - 833 Aryl radicals are intermediates in certain organic reactions. Synthesis Aryl radicals can be obtained via aryl diazonium salts. Alternatives for these salts are certain aryl triazenes and aryl hydrazines. Aryl bromides and iodides can be converted to aryl radicals via tributyltin hydride and related compounds and silyl hydrides. Aryl halides can also be converted via electrochemical cathodic reduction The mushroom ''Stephanospora caroticolor'' is suspected to generate an aryl radical as part of its biological chem ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Diazomethane
Diazomethane is an organic chemical compound with the formula CH2N2, discovered by German chemist Hans von Pechmann in 1894. It is the simplest diazo compound. In the pure form at room temperature, it is an extremely sensitive explosive yellow gas; thus, it is almost universally used as a solution in diethyl ether. The compound is a popular methylating agent in the laboratory, but it is too hazardous to be employed on an industrial scale without special precautions. Use of diazomethane has been significantly reduced by the introduction of the safer and equivalent reagent trimethylsilyldiazomethane. Use For safety and convenience diazomethane is always prepared as needed as a solution in diethyl ether, ether and used as such. It converts carboxylic acids to methyl esters and phenols into their methyl ethers. The reaction is thought to proceed via proton transfer from carboxylic acid to diazomethane to give a methyldiazonium cation, which reacts with the carboxylate ion to give th ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Azo Dye
Azo dyes are organic compounds bearing the functional group R−N=N−R′, in which R and R′ are usually aryl and substituted aryl groups. They are a commercially important family of azo compounds, i.e. compounds containing the C−N=N−C linkage. Azo dyes are synthetic dyes and do not occur naturally. Most azo dyes contain only one azo group but there are some that contain two or three azo groups, called "diazo dyes" and "triazo dyes" respectively. Azo dyes comprise 60–70% of all dyes used in food and textile industries. Azo dyes are widely used to treat textiles, leather articles, and some foods. Chemically related derivatives of azo dyes include azo pigments, which are insoluble in water and other solvents. Classes Many kinds of azo dyes are known, and several classification systems exist. Some classes include disperse dyes, metal-complex dyes, reactive dyes, and substantive dyes. Also called direct dyes, substantive dyes are employed for cellulose-based textil ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Peter Griess
Johann Peter Griess (6 September 1829 – 30 August 1888) was an industrial chemist and an early pioneer of organic chemistry. Griess was influential in the formation of modern dyes, first formulating the diazotization reaction of arylamines. Life After he finished at an agricultural private school, he joined the Hessian (soldier), Hessian cavalry, but left the military shortly after. He started his studies at the University of Jena in 1850, but changed to the University of Marburg in 1851. During his student life he was several times sentenced to the Karzer (campus jail) and was also banned from the city for one year, during which time he listened to lectures of Justus Liebig at the Ludwig Maximilian University of Munich. After most of the family possession had been spent, Griess had to start working at the chemical factory of Oehler in Offenbach am Main in 1856. This was only possible after the recommendation of Hermann Kolbe, who was head of the chemistry department in Marbu ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Angstrom
The angstrom (; ) is a unit of length equal to m; that is, one ten-billionth of a metre, a hundred-millionth of a centimetre, 0.1 nanometre, or 100 picometres. The unit is named after the Swedish physicist Anders Jonas Ångström (1814–1874). It was originally spelled with Swedish letters, as Ångström and later as ångström (). The latter spelling is still listed in some dictionaries, but is now rare in English texts. Some popular US dictionaries list only the spelling ''angstrom''. The unit's symbol is Å, which is a letter of the Swedish alphabet, regardless of how the unit is spelled. However, "A" or "A.U." may be used in less formal contexts or typographically limited media. The angstrom is often used in the natural sciences and technology to express sizes of atoms, molecules, microscopic biological structures, and lengths of chemical bonds, arrangement of atoms in crystals, wavelengths of electromagnetic radiation, and dimensions of integrated circuit part ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Supersaturation
In physical chemistry, supersaturation occurs with a solution (chemistry), solution when the concentration of a solute exceeds the concentration specified by the value of solubility at Solubility equilibrium, equilibrium. Most commonly the term is applied to a solution of a solid in a liquid, but it can also be applied to liquids and gases dissolved in a liquid. A supersaturated solution is in a metastable state; it may return to equilibrium by separation process, separation of the excess of solute from the solution, by dilution of the solution by adding solvent, or by increasing the solubility of the solute in the solvent. History Early studies of the phenomenon were conducted with sodium sulfate, also known as Glauber's Salt because, unusually, the solubility of this salt in water may decrease with increasing temperature. Early studies have been summarised by Tomlinson. It was shown that the crystallization of a supersaturated solution does not simply come from its agitation ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |