Xenon(II) Compounds on:
[Wikipedia]
[Google]
[Amazon]
Xenon is a
 Xenon and the other noble gases were for a long time considered to be completely chemically inert and not able to form compounds. However, while teaching at the
Xenon and the other noble gases were for a long time considered to be completely chemically inert and not able to form compounds. However, while teaching at the

 Xenon has
Xenon has  Liquid or solid xenon nanoparticles can be formed at room temperature by implanting Xe+ ions into a solid matrix. Many solids have lattice constants smaller than solid Xe. This results in compression of the implanted Xe to pressures that may be sufficient for its liquefaction or solidification.
Xenon is a member of the zero-
Liquid or solid xenon nanoparticles can be formed at room temperature by implanting Xe+ ions into a solid matrix. Many solids have lattice constants smaller than solid Xe. This results in compression of the implanted Xe to pressures that may be sufficient for its liquefaction or solidification.
Xenon is a member of the zero-
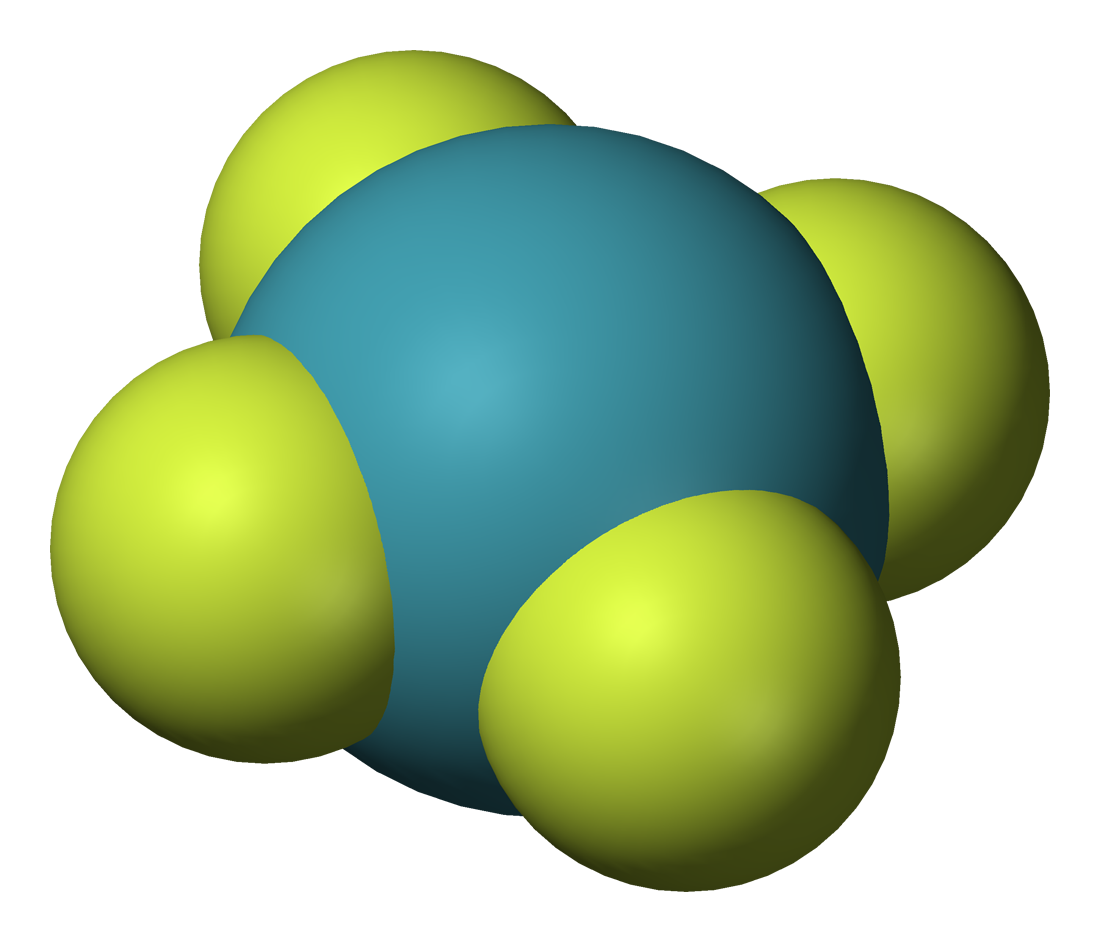
 Three fluorides are known: xenon difluoride, , xenon tetrafluoride, , and xenon hexafluoride, . XeF is theorized to be unstable. These are the starting points for the synthesis of almost all xenon compounds.
The solid, crystalline difluoride is formed when a mixture of fluorine and xenon gases is exposed to ultraviolet light. The ultraviolet component of ordinary daylight is sufficient. Long-term heating of at high temperatures under an catalyst yields . Pyrolysis of in the presence of sodium fluoride, NaF yields high-purity .
The xenon fluorides behave as both fluoride acceptors and fluoride donors, forming salts that contain such cations as and , and anions such as , , and . The green, paramagnetic is formed by the reduction of by xenon gas.
also forms complex (chemistry), coordination complexes with transition metal ions. More than 30 such complexes have been synthesized and characterized.
Whereas the xenon fluorides are well characterized, the other halides are not known, with the exception of Xenon dichloride, XeCl2 and Xenon tetrachloride, XeCl4. Xenon dichloride, formed by the high-frequency irradiation of a mixture of xenon, fluorine, and silicon tetrachloride, silicon or carbon tetrachloride, is reported to be an endothermic, colorless, crystalline compound that decomposes into the elements at 80 °C. However, may be merely a van der Waals molecule of weakly bound Xe atoms and molecules and not a real compound. Theoretical calculations indicate that the linear molecule is less stable than the van der Waals complex. Xenon tetrachloride is more unstable that it cannot be synthesized by chemical reactions. It was created by Radioactive decay, radioactive decay.
Three fluorides are known: xenon difluoride, , xenon tetrafluoride, , and xenon hexafluoride, . XeF is theorized to be unstable. These are the starting points for the synthesis of almost all xenon compounds.
The solid, crystalline difluoride is formed when a mixture of fluorine and xenon gases is exposed to ultraviolet light. The ultraviolet component of ordinary daylight is sufficient. Long-term heating of at high temperatures under an catalyst yields . Pyrolysis of in the presence of sodium fluoride, NaF yields high-purity .
The xenon fluorides behave as both fluoride acceptors and fluoride donors, forming salts that contain such cations as and , and anions such as , , and . The green, paramagnetic is formed by the reduction of by xenon gas.
also forms complex (chemistry), coordination complexes with transition metal ions. More than 30 such complexes have been synthesized and characterized.
Whereas the xenon fluorides are well characterized, the other halides are not known, with the exception of Xenon dichloride, XeCl2 and Xenon tetrachloride, XeCl4. Xenon dichloride, formed by the high-frequency irradiation of a mixture of xenon, fluorine, and silicon tetrachloride, silicon or carbon tetrachloride, is reported to be an endothermic, colorless, crystalline compound that decomposes into the elements at 80 °C. However, may be merely a van der Waals molecule of weakly bound Xe atoms and molecules and not a real compound. Theoretical calculations indicate that the linear molecule is less stable than the van der Waals complex. Xenon tetrachloride is more unstable that it cannot be synthesized by chemical reactions. It was created by Radioactive decay, radioactive decay.


 Continuous, short-arc, high pressure xenon arc lamps have a color temperature closely approximating noon sunlight and are used in Solar Simulator, solar simulators. That is, the chromaticity of these lamps closely approximates a heated black body radiator at the temperature of the Sun. First introduced in the 1940s, these lamps replaced the shorter-lived carbon arc lamps in movie projectors. They are also employed in typical 35mm movie film, 35mm, IMAX, and digital projectors, digital Movie projector, film projection systems. They are an excellent source of short wavelength ultraviolet radiation and have intense emissions in the near infrared used in some night vision systems. Xenon is used as a starter gas in Metal-halide lamp, metal halide lamps for HID Headlight, automotive HID headlights, and high-end tactical light, "tactical" flashlights.
The individual cells in a plasma display contain a mixture of xenon and neon ionized with electrodes. The interaction of this plasma with the electrodes generates ultraviolet photons, which then excite the phosphor coating on the front of the display.
Xenon is used as a "starter gas" in Sodium vapor lamp, high pressure sodium lamps. It has the lowest thermal conductivity and lowest ionization potential of all the non-radioactive noble gases. As a noble gas, it does not interfere with the chemical reactions occurring in the operating lamp. The low thermal conductivity minimizes thermal losses in the lamp while in the operating state, and the low ionization potential causes the breakdown voltage of the gas to be relatively low in the cold state, which allows the lamp to be more easily started.
Continuous, short-arc, high pressure xenon arc lamps have a color temperature closely approximating noon sunlight and are used in Solar Simulator, solar simulators. That is, the chromaticity of these lamps closely approximates a heated black body radiator at the temperature of the Sun. First introduced in the 1940s, these lamps replaced the shorter-lived carbon arc lamps in movie projectors. They are also employed in typical 35mm movie film, 35mm, IMAX, and digital projectors, digital Movie projector, film projection systems. They are an excellent source of short wavelength ultraviolet radiation and have intense emissions in the near infrared used in some night vision systems. Xenon is used as a starter gas in Metal-halide lamp, metal halide lamps for HID Headlight, automotive HID headlights, and high-end tactical light, "tactical" flashlights.
The individual cells in a plasma display contain a mixture of xenon and neon ionized with electrodes. The interaction of this plasma with the electrodes generates ultraviolet photons, which then excite the phosphor coating on the front of the display.
Xenon is used as a "starter gas" in Sodium vapor lamp, high pressure sodium lamps. It has the lowest thermal conductivity and lowest ionization potential of all the non-radioactive noble gases. As a noble gas, it does not interfere with the chemical reactions occurring in the operating lamp. The low thermal conductivity minimizes thermal losses in the lamp while in the operating state, and the low ionization potential causes the breakdown voltage of the gas to be relatively low in the cold state, which allows the lamp to be more easily started.
 Liquid xenon is used in Calorimeter (particle physics), calorimeters to measure gamma rays, and as a detector of hypothetical
Liquid xenon is used in Calorimeter (particle physics), calorimeters to measure gamma rays, and as a detector of hypothetical
Xenon
at ''The Periodic Table of Videos'' (University of Nottingham)
USGS Periodic Table – Xenon
{{Authority control Xenon, Chemical elements Noble gases Dissociative drugs General anesthetics Glycine receptor agonists Industrial gases Nicotinic antagonists NMDA receptor antagonists 5-HT3 antagonists ATPase inhibitors Rocket propellants
chemical element
A chemical element is a species of atoms that have a given number of protons in their nuclei, including the pure substance consisting only of that species. Unlike chemical compounds, chemical elements cannot be broken down into simpler sub ...
with the symbol
A symbol is a mark, sign, or word that indicates, signifies, or is understood as representing an idea, object, or relationship. Symbols allow people to go beyond what is known or seen by creating linkages between otherwise very different conc ...
Xe and atomic number
The atomic number or nuclear charge number (symbol ''Z'') of a chemical element is the charge number of an atomic nucleus. For ordinary nuclei, this is equal to the proton number (''n''p) or the number of protons found in the nucleus of every ...
54. It is a dense, colorless, odorless noble gas
The noble gases (historically also the inert gases; sometimes referred to as aerogens) make up a class of chemical elements with similar properties; under standard conditions, they are all odorless, colorless, monatomic gases with very low che ...
found in Earth's atmosphere
The atmosphere of Earth is the layer of gases, known collectively as air, retained by Earth's gravity that surrounds the planet and forms its planetary atmosphere. The atmosphere of Earth protects life on Earth by creating pressure allowing f ...
in trace amounts. Although generally unreactive, it can undergo a few chemical reaction
A chemical reaction is a process that leads to the chemical transformation of one set of chemical substances to another. Classically, chemical reactions encompass changes that only involve the positions of electrons in the forming and break ...
s such as the formation of xenon hexafluoroplatinate, the first noble gas compound
In chemistry, noble gas compounds are chemical compounds that include an element from the noble gases, group 18 of the periodic table. Although the noble gases are generally unreactive elements, many such compounds have been observed, particula ...
to be synthesized.
Xenon is used in flash lamps and arc lamps, and as a general anesthetic. The first excimer laser
An excimer laser, sometimes more correctly called an exciplex laser, is a form of ultraviolet laser which is commonly used in the production of microelectronic devices, semiconductor based integrated circuits or "chips", eye surgery, and micr ...
design used a xenon dimer molecule (Xe2) as the lasing medium, and the earliest laser
A laser is a device that emits light through a process of optical amplification based on the stimulated emission of electromagnetic radiation. The word "laser" is an acronym for "light amplification by stimulated emission of radiation". The firs ...
designs used xenon flash lamps as pumps
A pump is a device that moves fluids ( liquids or gases), or sometimes slurries, by mechanical action, typically converted from electrical energy into hydraulic energy. Pumps can be classified into three major groups according to the method the ...
. Xenon is also used to search for hypothetical weakly interacting massive particles
Weakly interacting massive particles (WIMPs) are hypothetical particles that are one of the proposed candidates for dark matter.
There exists no formal definition of a WIMP, but broadly, a WIMP is a new elementary particle which interacts via gra ...
and as a propellant
A propellant (or propellent) is a mass that is expelled or expanded in such a way as to create a thrust or other motive force in accordance with Newton's third law of motion, and "propel" a vehicle, projectile, or fluid payload. In vehicles, the ...
for ion thruster
An ion thruster, ion drive, or ion engine is a form of electric propulsion used for spacecraft propulsion. It creates thrust by accelerating ions using electricity.
An ion thruster ionizes a neutral gas by extracting some electrons out o ...
s in spacecraft.
Naturally occurring xenon consists of seven stable isotopes and two long-lived radioactive isotopes. More than 40 unstable xenon isotopes undergo radioactive decay
Radioactive decay (also known as nuclear decay, radioactivity, radioactive disintegration, or nuclear disintegration) is the process by which an unstable atomic nucleus loses energy by radiation. A material containing unstable nuclei is consid ...
, and the isotope ratios of xenon are an important tool for studying the early history of the Solar System
The Solar System Capitalization of the name varies. The International Astronomical Union, the authoritative body regarding astronomical nomenclature, specifies capitalizing the names of all individual astronomical objects but uses mixed "Solar ...
. Radioactive xenon-135 is produced by beta decay
In nuclear physics, beta decay (β-decay) is a type of radioactive decay in which a beta particle (fast energetic electron or positron) is emitted from an atomic nucleus, transforming the original nuclide to an isobar of that nuclide. For e ...
from iodine-135 (a product of nuclear fission
Nuclear fission is a nuclear reaction, reaction in which the atomic nucleus, nucleus of an atom splits into two or more smaller atomic nucleus, nuclei. The fission process often produces gamma ray, gamma photons, and releases a very large ...
), and is the most significant (and unwanted) neutron absorber in nuclear reactor
A nuclear reactor is a device used to initiate and control a fission nuclear chain reaction or nuclear fusion reactions. Nuclear reactors are used at nuclear power plants for electricity generation and in nuclear marine propulsion. Heat from nu ...
s.
History
Xenon was discovered in England by the Scottish chemist William Ramsay and English chemist Morris Travers in September 1898, shortly after their discovery of the elementskrypton
Krypton (from grc, κρυπτός, translit=kryptos 'the hidden one') is a chemical element with the symbol Kr and atomic number 36. It is a colorless, odorless, tasteless noble gas that occurs in trace amounts in the atmosphere and is often ...
and neon
Neon is a chemical element with the symbol Ne and atomic number 10. It is a noble gas. Neon is a colorless, odorless, inert monatomic gas under standard conditions, with about two-thirds the density of air. It was discovered (along with krypt ...
. They found xenon in the residue left over from evaporating components of liquid air. Ramsay suggested the name ''xenon'' for this gas from the Greek
Greek may refer to:
Greece
Anything of, from, or related to Greece, a country in Southern Europe:
*Greeks, an ethnic group.
*Greek language, a branch of the Indo-European language family.
**Proto-Greek language, the assumed last common ancestor ...
word ξένον ''xénon'', neuter singular form of ξένος ''xénos'', meaning 'foreign(er)', 'strange(r)', or 'guest'. In 1902, Ramsay estimated the proportion of xenon in the Earth's atmosphere to be one part in 20 million.
During the 1930s, American engineer Harold Edgerton
Harold Eugene "Doc" Edgerton (April 6, 1903 – January 4, 1990), also known as Papa Flash, was an American scientist and researcher, a professor of electrical engineering at the Massachusetts Institute of Technology. He is largely credited with ...
began exploring strobe light
A strobe light or stroboscopic lamp, commonly called a strobe, is a device used to produce regular flashes of light. It is one of a number of devices that can be used as a stroboscope. The word originated from the Ancient Greek ('), meaning ...
technology for high speed photography
High-speed photography is the science of taking pictures of very fast phenomena. In 1948, the Society of Motion Picture and Television Engineers (SMPTE) defined high-speed photography as any set of photographs captured by a camera capable of 69 ...
. This led him to the invention of the xenon flash lamp
The electric flash-lamp uses electric current to start flash powder burning, to provide a brief sudden burst of bright light. It was principally used for flash photography in the early 20th century but had other uses as well. Previously, photog ...
in which light is generated by passing brief electric current through a tube filled with xenon gas. In 1934, Edgerton was able to generate flashes as brief as one microsecond
A microsecond is a unit of time in the International System of Units (SI) equal to one millionth (0.000001 or 10−6 or ) of a second. Its symbol is μs, sometimes simplified to us when Unicode is not available.
A microsecond is equal to 100 ...
with this method.
In 1939, American physician Albert R. Behnke Jr. began exploring the causes of "drunkenness" in deep-sea divers. He tested the effects of varying the breathing mixtures on his subjects, and discovered that this caused the divers to perceive a change in depth. From his results, he deduced that xenon gas could serve as an anesthetic
An anesthetic (American English) or anaesthetic (British English; see spelling differences) is a drug used to induce anesthesia — in other words, to result in a temporary loss of sensation or awareness. They may be divided into tw ...
. Although Russian toxicologist Nikolay V. Lazarev apparently studied xenon anesthesia in 1941, the first published report confirming xenon anesthesia was in 1946 by American medical researcher John H. Lawrence, who experimented on mice. Xenon was first used as a surgical anesthetic in 1951 by American anesthesiologist Stuart C. Cullen, who successfully used it with two patients.
University of British Columbia
The University of British Columbia (UBC) is a public university, public research university with campuses near Vancouver and in Kelowna, British Columbia. Established in 1908, it is British Columbia's oldest university. The university ranks a ...
, Neil Bartlett discovered that the gas platinum hexafluoride (PtF6) was a powerful oxidizing
Redox (reduction–oxidation, , ) is a type of chemical reaction in which the oxidation states of substrate change. Oxidation is the loss of electrons or an increase in the oxidation state, while reduction is the gain of electrons or a d ...
agent that could oxidize oxygen gas (O2) to form dioxygenyl hexafluoroplatinate (). Since O2(1165 kJ/mol) and xenon (1170 kJ/mol) have almost the same first ionization potential
Ionization, or Ionisation is the process by which an atom or a molecule acquires a negative or positive charge by gaining or losing electrons, often in conjunction with other chemical changes. The resulting electrically charged atom or molecule ...
, Bartlett realized that platinum hexafluoride might also be able to oxidize xenon. On March 23, 1962, he mixed the two gases and produced the first known compound of a noble gas, xenon hexafluoroplatinate.
Bartlett thought its composition to be Xe+ tF6sup>−, but later work revealed that it was probably a mixture of various xenon-containing salts. Since then, many other xenon compounds have been discovered, in addition to some compounds of the noble gases argon
Argon is a chemical element with the symbol Ar and atomic number 18. It is in group 18 of the periodic table and is a noble gas. Argon is the third-most abundant gas in Earth's atmosphere, at 0.934% (9340 ppmv). It is more than twice as a ...
, krypton
Krypton (from grc, κρυπτός, translit=kryptos 'the hidden one') is a chemical element with the symbol Kr and atomic number 36. It is a colorless, odorless, tasteless noble gas that occurs in trace amounts in the atmosphere and is often ...
, and radon
Radon is a chemical element with the symbol Rn and atomic number 86. It is a radioactive, colourless, odourless, tasteless noble gas. It occurs naturally in minute quantities as an intermediate step in the normal radioactive decay chains through ...
, including argon fluorohydride (HArF), krypton difluoride (KrF2), and radon fluoride. By 1971, more than 80 xenon compounds were known.
In November 1989, IBM scientists demonstrated a technology capable of manipulating individual atom
Every atom is composed of a nucleus and one or more electrons bound to the nucleus. The nucleus is made of one or more protons and a number of neutrons. Only the most common variety of hydrogen has no neutrons.
Every solid, liquid, gas ...
s. The program, called IBM in atoms, used a scanning tunneling microscope
A scanning tunneling microscope (STM) is a type of microscope used for imaging surfaces at the atomic level. Its development in 1981 earned its inventors, Gerd Binnig and Heinrich Rohrer, then at IBM Zürich, the Nobel Prize in Physics in 1986 ...
to arrange 35 individual xenon atoms on a substrate of chilled crystal of nickel
Nickel is a chemical element with symbol Ni and atomic number 28. It is a silvery-white lustrous metal with a slight golden tinge. Nickel is a hard and ductile transition metal. Pure nickel is chemically reactive but large pieces are slow ...
to spell out the three letter company initialism. It was the first time atoms had been precisely positioned on a flat surface.
Characteristics

 Xenon has
Xenon has atomic number
The atomic number or nuclear charge number (symbol ''Z'') of a chemical element is the charge number of an atomic nucleus. For ordinary nuclei, this is equal to the proton number (''n''p) or the number of protons found in the nucleus of every ...
54; that is, its nucleus contains 54 protons. At standard temperature and pressure
Standard temperature and pressure (STP) are standard sets of conditions for experimental measurements to be established to allow comparisons to be made between different sets of data. The most used standards are those of the International Union ...
, pure xenon gas has a density of 5.894 kg/m3, about 4.5 times the density of the Earth's atmosphere at sea level, 1.217 kg/m3. As a liquid, xenon has a density of up to 3.100 g/mL, with the density maximum occurring at the triple point. Liquid xenon has a high polarizability due to its large atomic volume, and thus is an excellent solvent. It can dissolve hydrocarbons, biological molecules, and even water. Under the same conditions, the density of solid xenon, 3.640 g/cm3, is greater than the average density of granite
Granite () is a coarse-grained ( phaneritic) intrusive igneous rock composed mostly of quartz, alkali feldspar, and plagioclase. It forms from magma with a high content of silica and alkali metal oxides that slowly cools and solidifies und ...
, 2.75 g/cm3. Under gigapascals of pressure
Pressure (symbol: ''p'' or ''P'') is the force applied perpendicular to the surface of an object per unit area over which that force is distributed. Gauge pressure (also spelled ''gage'' pressure)The preferred spelling varies by country a ...
, xenon forms a metallic phase.
Solid xenon changes from face-centered cubic
In crystallography, the cubic (or isometric) crystal system is a crystal system where the unit cell is in the shape of a cube. This is one of the most common and simplest shapes found in crystals and minerals.
There are three main varieties o ...
(fcc) to hexagonal close packed (hcp) crystal phase under pressure and begins to turn metallic at about 140 GPa, with no noticeable volume change in the hcp phase. It is completely metallic at 155 GPa. When metallized, xenon appears sky blue because it absorbs red light and transmits other visible frequencies. Such behavior is unusual for a metal and is explained by the relatively small width of the electron bands in that state.
 Liquid or solid xenon nanoparticles can be formed at room temperature by implanting Xe+ ions into a solid matrix. Many solids have lattice constants smaller than solid Xe. This results in compression of the implanted Xe to pressures that may be sufficient for its liquefaction or solidification.
Xenon is a member of the zero-
Liquid or solid xenon nanoparticles can be formed at room temperature by implanting Xe+ ions into a solid matrix. Many solids have lattice constants smaller than solid Xe. This results in compression of the implanted Xe to pressures that may be sufficient for its liquefaction or solidification.
Xenon is a member of the zero-valence
Valence or valency may refer to:
Science
* Valence (chemistry), a measure of an element's combining power with other atoms
* Degree (graph theory), also called the valency of a vertex in graph theory
* Valency (linguistics), aspect of verbs rel ...
elements that are called noble or inert gas
An inert gas is a gas that does not readily undergo chemical reactions with other chemical substances and therefore does not readily form chemical compounds. The noble gases often do not react with many substances and were historically referred to ...
es. It is inert to most common chemical reactions (such as combustion, for example) because the outer valence shell contains eight electrons. This produces a stable, minimum energy configuration in which the outer electrons are tightly bound.
In a gas-filled tube
A gas-filled tube, also commonly known as a discharge tube or formerly as a Plücker tube, is an arrangement of electrodes in a gas within an insulating, temperature-resistant envelope. Gas-filled tubes exploit phenomena related to electric ...
, xenon emits a blue
Blue is one of the three primary colours in the RYB colour model (traditional colour theory), as well as in the RGB (additive) colour model. It lies between violet and cyan on the spectrum of visible light. The eye perceives blue when ...
or lavenderish glow when excited by electrical discharge
An electric discharge is the release and transmission of electricity in an applied electric field through a medium such as a gas (ie., an outgoing flow of electric current through a non-metal medium).American Geophysical Union, National Research ...
. Xenon emits a band of emission lines that span the visual spectrum, but the most intense lines occur in the region of blue light, producing the coloration.
Occurrence and production
Xenon is atrace gas
Trace gases are gases that are present in small amounts within an environment such as a planet's atmosphere. Trace gases in Earth's atmosphere are gases other than nitrogen (78.1%), oxygen (20.9%), and argon (0.934%) which, in combination, make u ...
in Earth's atmosphere
The atmosphere of Earth is the layer of gases, known collectively as air, retained by Earth's gravity that surrounds the planet and forms its planetary atmosphere. The atmosphere of Earth protects life on Earth by creating pressure allowing f ...
, occurring at a volume fraction of (parts per billion
In science and engineering, the parts-per notation is a set of pseudo-units to describe small values of miscellaneous dimensionless quantities, e.g. mole fraction or mass fraction. Since these fractions are quantity-per-quantity measures, th ...
), or approximately 1 part per 11.5 million. It is also found as a component of gases emitted from some mineral spring
Mineral springs are naturally occurring springs that produces hard water, water that contains dissolved minerals. Salts, sulfur compounds, and gases are among the substances that can be dissolved in the spring water during its passage unde ...
s. Given a total mass of the atmosphere of , the atmosphere contains on the order of of xenon in total when taking the average molar mass of the atmosphere as 28.96 g/mol which is equivalent to some 394 mass ppb.
Commercial
Xenon is obtained commercially as a by-product of the separation of air intooxygen
Oxygen is the chemical element with the symbol O and atomic number 8. It is a member of the chalcogen group in the periodic table, a highly reactive nonmetal, and an oxidizing agent that readily forms oxides with most elements as we ...
and nitrogen
Nitrogen is the chemical element with the symbol N and atomic number 7. Nitrogen is a nonmetal and the lightest member of group 15 of the periodic table, often called the pnictogens. It is a common element in the universe, estimated at seve ...
. After this separation, generally performed by fractional distillation
Fractional distillation is the separation of a mixture into its component parts, or fractions. Chemical compounds are separated by heating them to a temperature at which one or more fractions of the mixture will vaporize. It uses distillation to ...
in a double-column plant, the liquid oxygen
Liquid oxygen—abbreviated LOx, LOX or Lox in the aerospace, submarine and gas industries—is the liquid form of molecular oxygen. It was used as the oxidizer in the first liquid-fueled rocket invented in 1926 by Robert H. Goddard, an a ...
produced will contain small quantities of krypton
Krypton (from grc, κρυπτός, translit=kryptos 'the hidden one') is a chemical element with the symbol Kr and atomic number 36. It is a colorless, odorless, tasteless noble gas that occurs in trace amounts in the atmosphere and is often ...
and xenon. By additional fractional distillation, the liquid oxygen may be enriched to contain 0.1–0.2% of a krypton/xenon mixture, which is extracted either by adsorption
Adsorption is the adhesion of atoms, ions or molecules from a gas, liquid or dissolved solid to a surface. This process creates a film of the ''adsorbate'' on the surface of the ''adsorbent''. This process differs from absorption, in which ...
onto silica gel
Silica gel is an amorphous and porous form of silicon dioxide (silica), consisting of an irregular tridimensional framework of alternating silicon and oxygen atoms with nanometer-scale voids and pores. The voids may contain water or some other ...
or by distillation. Finally, the krypton/xenon mixture may be separated into krypton and xenon by further distillation.
Worldwide production of xenon in 1998 was estimated at . At a density of this is equivalent to roughly . Because of its scarcity, xenon is much more expensive than the lighter noble gases—approximate prices for the purchase of small quantities in Europe in 1999 were 10 €/L (=~1.7€/g) for xenon, 1 €/L (=~0.27€/g) for krypton, and 0.20 €/L (=~0.22€/g) for neon, while the much more plentiful argon, which makes up over 1% by volume of earth's atmosphere, costs less than a cent per liter.
Solar system
Within the Solar System, thenucleon
In physics and chemistry, a nucleon is either a proton or a neutron, considered in its role as a component of an atomic nucleus. The number of nucleons in a nucleus defines the atom's mass number (nucleon number).
Until the 1960s, nucleons w ...
fraction of xenon is , for an abundance of approximately one part in 630 thousand of the total mass. Xenon is relatively rare in the Sun's atmosphere, on Earth
Earth is the third planet from the Sun and the only astronomical object known to harbor life. While large volumes of water can be found throughout the Solar System, only Earth sustains liquid surface water. About 71% of Earth's surf ...
, and in asteroid
An asteroid is a minor planet of the Solar System#Inner solar system, inner Solar System. Sizes and shapes of asteroids vary significantly, ranging from 1-meter rocks to a dwarf planet almost 1000 km in diameter; they are rocky, metallic o ...
s and comet
A comet is an icy, small Solar System body that, when passing close to the Sun, warms and begins to release gases, a process that is called outgassing. This produces a visible atmosphere or Coma (cometary), coma, and sometimes also a Comet ta ...
s. The abundance of xenon in the atmosphere of planet Jupiter
Jupiter is the fifth planet from the Sun and the largest in the Solar System. It is a gas giant with a mass more than two and a half times that of all the other planets in the Solar System combined, but slightly less than one-thousandt ...
is unusually high, about 2.6 times that of the Sun. This abundance remains unexplained, but may have been caused by an early and rapid buildup of planetesimal
Planetesimals are solid objects thought to exist in protoplanetary disks and debris disks. Per the Chamberlin–Moulton planetesimal hypothesis, they are believed to form out of cosmic dust grains. Believed to have formed in the Solar System ...
s—small, subplanetary bodies—before the heating of the presolar disk. (Otherwise, xenon would not have been trapped in the planetesimal ices.) The problem of the low terrestrial xenon may be explained by covalent bonding of xenon to oxygen within quartz
Quartz is a hard, crystalline mineral composed of silica ( silicon dioxide). The atoms are linked in a continuous framework of SiO4 silicon-oxygen tetrahedra, with each oxygen being shared between two tetrahedra, giving an overall chemical ...
, reducing the outgassing
Outgassing (sometimes called offgassing, particularly when in reference to indoor air quality) is the release of a gas that was dissolved, trapped, frozen, or absorbed in some material. Outgassing can include sublimation and evaporation (which ...
of xenon into the atmosphere.
Stellar
Unlike the lower-mass noble gases, the normalstellar nucleosynthesis
Stellar nucleosynthesis is the creation (nucleosynthesis) of chemical elements by nuclear fusion reactions within stars. Stellar nucleosynthesis has occurred since the original creation of hydrogen, helium and lithium during the Big Bang. As a ...
process inside a star does not form xenon. Elements more massive than iron-56
Iron-56 (56Fe) is the most common isotope of iron. About 91.754% of all iron is iron-56.
Of all nuclides, iron-56 has the lowest mass per nucleon. With 8.8 MeV binding energy per nucleon, iron-56 is one of the most tightly bound nuclei.
...
consume energy through fusion, and the synthesis of xenon represents no energy gain for a star. Instead, xenon is formed during supernova explosions, in classical nova
A nova (plural novae or novas) is a transient astronomical event that causes the sudden appearance of a bright, apparently "new" star (hence the name "nova", which is Latin for "new") that slowly fades over weeks or months. Causes of the dramati ...
explosions, by the slow neutron-capture process ( s-process) in red giant
A red giant is a luminous giant star of low or intermediate mass (roughly 0.3–8 solar masses ()) in a late phase of stellar evolution. The outer atmosphere is inflated and tenuous, making the radius large and the surface temperature around or ...
stars that have exhausted their core hydrogen and entered the asymptotic giant branch
The asymptotic giant branch (AGB) is a region of the Hertzsprung–Russell diagram populated by evolved cool luminous stars. This is a period of stellar evolution undertaken by all low- to intermediate-mass stars (about 0.5 to 8 solar masses) lat ...
, and from radioactive decay, for example by beta decay
In nuclear physics, beta decay (β-decay) is a type of radioactive decay in which a beta particle (fast energetic electron or positron) is emitted from an atomic nucleus, transforming the original nuclide to an isobar of that nuclide. For e ...
of extinct iodine-129
Iodine-129 (129I) is a long-lived radioisotope of iodine which occurs naturally, but also is of special interest in the monitoring and effects of man-made nuclear fission products, where it serves as both tracer and potential radiological contami ...
and spontaneous fission
Spontaneous fission (SF) is a form of radioactive decay that is found only in very heavy chemical elements. The nuclear binding energy of the elements reaches its maximum at an atomic mass number of about 56 (e.g., iron-56); spontaneous breakd ...
of thorium
Thorium is a weakly radioactive metallic chemical element with the symbol Th and atomic number 90. Thorium is silvery and tarnishes black when it is exposed to air, forming thorium dioxide; it is moderately soft and malleable and has a high ...
, uranium
Uranium is a chemical element with the symbol U and atomic number 92. It is a silvery-grey metal in the actinide series of the periodic table. A uranium atom has 92 protons and 92 electrons, of which 6 are valence electrons. Uranium is weakly ...
, and plutonium
Plutonium is a radioactive chemical element with the symbol Pu and atomic number 94. It is an actinide metal of silvery-gray appearance that tarnishes when exposed to air, and forms a dull coating when oxidized. The element normally exhib ...
.
Nuclear fission
Xenon-135 is a notable neutron poison with a high fission product yield. As it is relatively short lived, it decays at the same rate it is produced during ''steady'' operation of a nuclear reactor. However, if power is reduced or the reactor isscram
A scram or SCRAM is an emergency shutdown of a nuclear reactor effected by immediately terminating the fission reaction. It is also the name that is given to the manually operated kill switch that initiates the shutdown. In commercial reactor ...
ed, less xenon is destroyed than is produced from the beta decay of its parent nuclides. This phenomenon called xenon poisoning
The iodine pit, also called the iodine hole or xenon pit, is a temporary disabling of a nuclear reactor due to buildup of short- lived nuclear poisons in the reactor core. The main isotope responsible is 135Xe, mainly produced by natural decay of ...
can cause significant problems in restarting a reactor after a scram or increasing power after it had been reduced and it was one of several contributing factors in the Chernobyl nuclear accident.
Stable or extremely long lived isotopes of xenon are also produced in appreciable quantities in nuclear fission. Xenon-136 is produced when xenon-135 undergoes neutron capture
Neutron capture is a nuclear reaction in which an atomic nucleus and one or more neutrons collide and merge to form a heavier nucleus. Since neutrons have no electric charge, they can enter a nucleus more easily than positively charged protons, ...
before it can decay. The ratio of xenon-136 to xenon-135 (or its decay products) can give hints as to the power history of a given reactor and the absence of xenon-136 is a "fingerprint" for nuclear explosions, as xenon-135 is not produced directly but as a product of successive beta decays and thus it can't absorb any neutrons in a nuclear explosion which occurs in fractions of a second.
The stable isotope xenon-132 has a fission product yield of over 4% in the thermal neutron fission of which means that stable or nearly stable xenon isotopes have a higher mass fraction in spent nuclear fuel
Spent nuclear fuel, occasionally called used nuclear fuel, is nuclear fuel that has been irradiated in a nuclear reactor (usually at a nuclear power plant). It is no longer useful in sustaining a nuclear reaction in an ordinary thermal reactor an ...
(which is about 3% fission products) than it does in air. However, there is as of 2022 no commercial effort to extract xenon from spent fuel during nuclear reprocessing
Nuclear reprocessing is the chemical separation of fission products and actinides from spent nuclear fuel. Originally, reprocessing was used solely to extract plutonium for producing nuclear weapons. With commercialization of nuclear power, ...
.
Isotopes
Naturally occurring xenon is composed of seven stableisotope
Isotopes are two or more types of atoms that have the same atomic number (number of protons in their nuclei) and position in the periodic table (and hence belong to the same chemical element), and that differ in nucleon numbers ( mass number ...
s: 126Xe, 128–132Xe, and 134Xe. The isotopes 126Xe and 134Xe are predicted by theory to undergo double beta decay, but this has never been observed so they are considered stable. In addition, more than 40 unstable isotopes have been studied. The longest lived of these isotopes are the primordial
Primordial may refer to:
* Primordial era, an era after the Big Bang. See Chronology of the universe
* Primordial sea (a.k.a. primordial ocean, ooze or soup). See Abiogenesis
* Primordial nuclide, nuclides, a few radioactive, that formed before ...
124Xe, which undergoes double electron capture with a half-life of , and 136Xe, which undergoes double beta decay with a half-life of . 129Xe is produced by beta decay
In nuclear physics, beta decay (β-decay) is a type of radioactive decay in which a beta particle (fast energetic electron or positron) is emitted from an atomic nucleus, transforming the original nuclide to an isobar of that nuclide. For e ...
of 129 I, which has a half-life
Half-life (symbol ) is the time required for a quantity (of substance) to reduce to half of its initial value. The term is commonly used in nuclear physics to describe how quickly unstable atoms undergo radioactive decay or how long stable at ...
of 16 million years. 131mXe, 133Xe, 133mXe, and 135Xe are some of the fission
Fission, a splitting of something into two or more parts, may refer to:
* Fission (biology), the division of a single entity into two or more parts and the regeneration of those parts into separate entities resembling the original
* Nuclear fissio ...
products of 235 U and 239 Pu, and are used to detect and monitor nuclear explosions.
Nuclear spin
Nuclei of two of the stable isotopes of xenon, 129Xe and 131Xe, have non-zero intrinsic angular momenta ( nuclear spins, suitable fornuclear magnetic resonance
Nuclear magnetic resonance (NMR) is a physical phenomenon in which nuclei in a strong constant magnetic field are perturbed by a weak oscillating magnetic field (in the near field) and respond by producing an electromagnetic signal with a ...
). The nuclear spins can be aligned beyond ordinary polarization levels by means of circularly polarized light and rubidium
Rubidium is the chemical element with the symbol Rb and atomic number 37. It is a very soft, whitish-grey solid in the alkali metal group, similar to potassium and caesium. Rubidium is the first alkali metal in the group to have a density hig ...
vapor. The resulting spin polarization
Spin polarization is the degree to which the spin, i.e., the intrinsic angular momentum of elementary particles, is aligned with a given direction. This property may pertain to the spin, hence to the magnetic moment, of conduction electrons in fer ...
of xenon nuclei can surpass 50% of its maximum possible value, greatly exceeding the thermal equilibrium value dictated by paramagnetic
Paramagnetism is a form of magnetism whereby some materials are weakly attracted by an externally applied magnetic field, and form internal, induced magnetic fields in the direction of the applied magnetic field. In contrast with this behavior, ...
statistics (typically 0.001% of the maximum value at room temperature
Colloquially, "room temperature" is a range of air temperatures that most people prefer for indoor settings. It feels comfortable to a person when they are wearing typical indoor clothing. Human comfort can extend beyond this range depending on ...
, even in the strongest magnet
A magnet is a material or object that produces a magnetic field. This magnetic field is invisible but is responsible for the most notable property of a magnet: a force that pulls on other ferromagnetic materials, such as iron, steel, nic ...
s). Such non-equilibrium alignment of spins is a temporary condition, and is called '' hyperpolarization''. The process of hyperpolarizing the xenon is called ''optical pumping'' (although the process is different from pumping a laser).
Because a 129Xe nucleus has a spin of 1/2, and therefore a zero electric quadrupole moment, the 129Xe nucleus does not experience any quadrupolar interactions during collisions with other atoms, and the hyperpolarization persists for long periods even after the engendering light and vapor have been removed. Spin polarization of 129Xe can persist from several seconds for xenon atoms dissolved in blood
Blood is a body fluid in the circulatory system of humans and other vertebrates that delivers necessary substances such as nutrients and oxygen to the cells, and transports metabolic waste products away from those same cells. Blood in th ...
to several hours in the gas phase and several days in deeply frozen solid xenon. In contrast, 131Xe has a nuclear spin value of and a nonzero quadrupole moment, and has t1 relaxation times in the millisecond
A millisecond (from '' milli-'' and second; symbol: ms) is a unit of time in the International System of Units (SI) equal to one thousandth (0.001 or 10−3 or 1/1000) of a second and to 1000 microseconds.
A unit of 10 milliseconds may be ca ...
and second ranges.
From fission
Some radioactive isotopes of xenon (for example, 133Xe and 135Xe) are produced byneutron
The neutron is a subatomic particle, symbol or , which has a neutral (not positive or negative) charge, and a mass slightly greater than that of a proton. Protons and neutrons constitute the nuclei of atoms. Since protons and neutrons behav ...
irradiation of fissionable material within nuclear reactor
A nuclear reactor is a device used to initiate and control a fission nuclear chain reaction or nuclear fusion reactions. Nuclear reactors are used at nuclear power plants for electricity generation and in nuclear marine propulsion. Heat from nu ...
s. 135Xe is of considerable significance in the operation of nuclear fission reactors. 135Xe has a huge cross section for thermal neutrons, 2.6×106 barns, and operates as a neutron absorber or "poison
Poison is a chemical substance that has a detrimental effect to life. The term is used in a wide range of scientific fields and industries, where it is often specifically defined. It may also be applied colloquially or figuratively, with a broa ...
" that can slow or stop the chain reaction after a period of operation. This was discovered in the earliest nuclear reactors built by the American Manhattan Project
The Manhattan Project was a research and development undertaking during World War II that produced the first nuclear weapons. It was led by the United States with the support of the United Kingdom and Canada. From 1942 to 1946, the project w ...
for plutonium
Plutonium is a radioactive chemical element with the symbol Pu and atomic number 94. It is an actinide metal of silvery-gray appearance that tarnishes when exposed to air, and forms a dull coating when oxidized. The element normally exhib ...
production. However, the designers had made provisions in the design to increase the reactor's reactivity (the number of neutrons per fission that go on to fission other atoms of nuclear fuel
Nuclear fuel is material used in nuclear power stations to produce heat to power turbines. Heat is created when nuclear fuel undergoes nuclear fission.
Most nuclear fuels contain heavy fissile actinide elements that are capable of undergoi ...
).
135Xe reactor poisoning was a major factor in the Chernobyl disaster
The Chernobyl disaster was a nuclear accident that occurred on 26 April 1986 at the No. 4 nuclear reactor, reactor in the Chernobyl Nuclear Power Plant, near the city of Pripyat in the north of the Ukrainian Soviet Socialist Republic, Ukrainia ...
. A shutdown or decrease of power of a reactor can result in buildup of 135Xe, with reactor operation going into a condition known as the iodine pit. Under adverse conditions, relatively high concentrations of radioactive xenon isotopes may emanate from cracked fuel rod
Nuclear fuel is material used in nuclear power stations to produce heat to power turbines. Heat is created when nuclear fuel undergoes nuclear fission.
Most nuclear fuels contain heavy fissile actinide elements that are capable of undergoing ...
s, or fissioning of uranium in Water cooling, cooling water.
Isotope ratios of xenon produced in natural nuclear fission reactors at Oklo in Gabon reveal the reactor properties during chain reaction that took place about 2 billion years ago.
Cosmic processes
Because xenon is a tracer for two parent isotopes, xenon isotope ratios in meteorites are a powerful tool for studying the formation of the Solar System. The Iodine–xenon dating, iodine–xenon method of Radiometric dating, dating gives the time elapsed between nucleosynthesis and the condensation of a solid object from the solar nebula. In 1960, physicist John Reynolds (physicist), John H. Reynolds discovered that certain meteorites contained an isotopic anomaly in the form of an overabundance of xenon-129. He inferred that this was a decay product of radioactiveiodine-129
Iodine-129 (129I) is a long-lived radioisotope of iodine which occurs naturally, but also is of special interest in the monitoring and effects of man-made nuclear fission products, where it serves as both tracer and potential radiological contami ...
. This isotope is produced slowly by cosmic ray spallation and nuclear fission
Nuclear fission is a nuclear reaction, reaction in which the atomic nucleus, nucleus of an atom splits into two or more smaller atomic nucleus, nuclei. The fission process often produces gamma ray, gamma photons, and releases a very large ...
, but is produced in quantity only in supernova explosions.
Because the half-life of 129I is comparatively short on a cosmological time scale (16 million years), this demonstrated that only a short time had passed between the supernova and the time the meteorites had solidified and trapped the 129I. These two events (supernova and solidification of gas cloud) were inferred to have happened during the early history of the Solar System
The Solar System Capitalization of the name varies. The International Astronomical Union, the authoritative body regarding astronomical nomenclature, specifies capitalizing the names of all individual astronomical objects but uses mixed "Solar ...
, because the 129I isotope was likely generated shortly before the Solar System was formed, seeding the solar gas cloud with isotopes from a second source. This supernova source may also have caused collapse of the solar gas cloud.
In a similar way, xenon isotopic ratios such as 129Xe/130Xe and 136Xe/130Xe are a powerful tool for understanding planetary differentiation and early outgassing. For example, the atmosphere of Mars shows a xenon abundance similar to that of Earth (0.08 parts per million) but Mars shows a greater abundance of 129Xe than the Earth or the Sun. Since this isotope is generated by radioactive decay, the result may indicate that Mars lost most of its primordial atmosphere, possibly within the first 100 million years after the planet was formed. In another example, excess 129Xe found in carbon dioxide well gases from New Mexico is believed to be from the decay of Mantle (geology), mantle-derived gases from soon after Earth's formation.
Compounds
After Neil Bartlett's discovery in 1962 that xenon can form chemical compounds, a large number of xenon compounds have been discovered and described. Almost all known xenon compounds contain the electronegative atoms fluorine or oxygen. The chemistry of xenon in each oxidation state is analogous to that of the neighboring element iodine in the immediately lower oxidation state.Halides

Oxides and oxohalides
Three oxides of xenon are known: xenon trioxide () and xenon tetroxide (), both of which are dangerously explosive and powerful oxidizing agents, and xenon dioxide (XeO2), which was reported in 2011 with a coordination number of four. XeO2 forms when xenon tetrafluoride is poured over ice. Its crystal structure may allow it to replace silicon in silicate minerals. The XeOO+ cation has been identified by infrared spectroscopy in solidargon
Argon is a chemical element with the symbol Ar and atomic number 18. It is in group 18 of the periodic table and is a noble gas. Argon is the third-most abundant gas in Earth's atmosphere, at 0.934% (9340 ppmv). It is more than twice as a ...
.
Xenon does not react with oxygen directly; the trioxide is formed by the hydrolysis of :
: + 3 → + 6 HF
is weakly acidic, dissolving in alkali to form unstable ''xenate'' salts containing the anion. These unstable salts easily disproportionation, disproportionate into xenon gas and perxenate salts, containing the anion.
Barium perxenate, when treated with concentrated sulfuric acid, yields gaseous xenon tetroxide:
: + 2 → 2 + 2 +
To prevent decomposition, the xenon tetroxide thus formed is quickly cooled into a pale-yellow solid. It explodes above −35.9 °C into xenon and oxygen gas, but is otherwise stable.
A number of xenon oxyfluorides are known, including , xenon oxytetrafluoride, , , and . is formed by reacting oxygen difluoride, with xenon gas at low temperatures. It may also be obtained by partial hydrolysis of . It disproportionates at −20 °C into and . is formed by the partial hydrolysis of , or the reaction of with sodium perxenate, . The latter reaction also produces a small amount of . reacts with caesium fluoride, CsF to form the anion, while XeOF3 reacts with the alkali metal fluorides potassium fluoride, KF, rubidium fluoride, RbF and CsF to form the anion.
Other compounds
Xenon can be directly bonded to a less electronegative element than fluorine or oxygen, particularly carbon. Electron-withdrawing groups, such as groups with fluorine substitution, are necessary to stabilize these compounds. Numerous such compounds have been characterized, including: * , where C6F5 is the pentafluorophenyl group. * * * * * * * * Other compounds containing xenon bonded to a less electronegative element include and . The latter is synthesized from dioxygenyl tetrafluoroborate, , at −100 °C. An unusual ion containing xenon is the tetraxenonogold(II) cation, , which contains Xe–Au bonds. This ion occurs in the compound , and is remarkable in having direct chemical bonds between two notoriously unreactive atoms, xenon and gold, with xenon acting as a transition metal ligand. The compound contains a Xe–Xe bond, the longest element-element bond known (308.71 pm = 3.0871 Angstrom, Å). In 1995, M. Räsänen and co-workers, scientists at the University of Helsinki in Finland, announced the preparation of xenon dihydride (HXeH), and later xenon hydride-hydroxide (HXeOH), hydroxenoacetylene (HXeCCH), and other Xe-containing molecules. In 2008, Khriachtchev ''et al.'' reported the preparation of HXeOXeH by the photolysis of water within a cryogenic xenon matrix. Deuterium, Deuterated molecules, HXeOD and DXeOH, have also been produced.Clathrates and excimers
In addition to compounds where xenon forms a chemical bond, xenon can form clathrates—substances where xenon atoms or pairs are trapped by the Crystal structure, crystalline lattice of another compound. One example is xenon hydrate (Xe·H2O), where xenon atoms occupy vacancies in a lattice of water molecules. This clathrate has a melting point of 24 °C. The deuterated version of this hydrate has also been produced. Another example is xenon hydride (Xe(H2)8), in which xenon pairs (Dimer (chemistry), dimers) are trapped inside solid hydrogen. Such clathrate hydrates can occur naturally under conditions of high pressure, such as in Lake Vostok underneath the Antarctica, Antarctic ice sheet. Clathrate formation can be used to fractionally distill xenon, argon and krypton. Xenon can also form endohedral fullerene compounds, where a xenon atom is trapped inside a fullerene molecule. The xenon atom trapped in the fullerene can be observed by 129Xenuclear magnetic resonance
Nuclear magnetic resonance (NMR) is a physical phenomenon in which nuclei in a strong constant magnetic field are perturbed by a weak oscillating magnetic field (in the near field) and respond by producing an electromagnetic signal with a ...
(NMR) spectroscopy. Through the sensitive chemical shift of the xenon atom to its environment, chemical reactions on the fullerene molecule can be analyzed. These observations are not without caveat, however, because the xenon atom has an electronic influence on the reactivity of the fullerene.
When xenon atoms are in the stationary state, ground energy state, they repel each other and will not form a bond. When xenon atoms becomes energized, however, they can form an excimer (excited dimer) until the electrons return to the ground state. This entity is formed because the xenon atom tends to complete the outermost Electron shell, electronic shell by adding an electron from a neighboring xenon atom. The typical lifetime of a xenon excimer is 1–5 nanoseconds, and the decay releases photons with wavelengths of about 150 and 173 Nanometre, nm. Xenon can also form excimers with other elements, such as the halogens bromine, chlorine, and fluorine.
Applications
Although xenon is rare and relatively expensive to extract from the atmosphere of Earth, Earth's atmosphere, it has a number of applications.Illumination and optics
Gas-discharge lamps
Xenon is used in light-emitting devices called xenon flash lamps, used in Flash (photography), photographic flashes and stroboscopic lamps; to excite the active laser medium, active medium inlaser
A laser is a device that emits light through a process of optical amplification based on the stimulated emission of electromagnetic radiation. The word "laser" is an acronym for "light amplification by stimulated emission of radiation". The firs ...
s which then generate coherent light; and, occasionally, in Bactericide, bactericidal lamps. The first solid-state laser
A laser is a device that emits light through a process of optical amplification based on the stimulated emission of electromagnetic radiation. The word "laser" is an acronym for "light amplification by stimulated emission of radiation". The firs ...
, invented in 1960, was pumped by a xenon flash lamp, and lasers used to power inertial confinement fusion are also pumped by xenon flash lamps.


 Continuous, short-arc, high pressure xenon arc lamps have a color temperature closely approximating noon sunlight and are used in Solar Simulator, solar simulators. That is, the chromaticity of these lamps closely approximates a heated black body radiator at the temperature of the Sun. First introduced in the 1940s, these lamps replaced the shorter-lived carbon arc lamps in movie projectors. They are also employed in typical 35mm movie film, 35mm, IMAX, and digital projectors, digital Movie projector, film projection systems. They are an excellent source of short wavelength ultraviolet radiation and have intense emissions in the near infrared used in some night vision systems. Xenon is used as a starter gas in Metal-halide lamp, metal halide lamps for HID Headlight, automotive HID headlights, and high-end tactical light, "tactical" flashlights.
The individual cells in a plasma display contain a mixture of xenon and neon ionized with electrodes. The interaction of this plasma with the electrodes generates ultraviolet photons, which then excite the phosphor coating on the front of the display.
Xenon is used as a "starter gas" in Sodium vapor lamp, high pressure sodium lamps. It has the lowest thermal conductivity and lowest ionization potential of all the non-radioactive noble gases. As a noble gas, it does not interfere with the chemical reactions occurring in the operating lamp. The low thermal conductivity minimizes thermal losses in the lamp while in the operating state, and the low ionization potential causes the breakdown voltage of the gas to be relatively low in the cold state, which allows the lamp to be more easily started.
Continuous, short-arc, high pressure xenon arc lamps have a color temperature closely approximating noon sunlight and are used in Solar Simulator, solar simulators. That is, the chromaticity of these lamps closely approximates a heated black body radiator at the temperature of the Sun. First introduced in the 1940s, these lamps replaced the shorter-lived carbon arc lamps in movie projectors. They are also employed in typical 35mm movie film, 35mm, IMAX, and digital projectors, digital Movie projector, film projection systems. They are an excellent source of short wavelength ultraviolet radiation and have intense emissions in the near infrared used in some night vision systems. Xenon is used as a starter gas in Metal-halide lamp, metal halide lamps for HID Headlight, automotive HID headlights, and high-end tactical light, "tactical" flashlights.
The individual cells in a plasma display contain a mixture of xenon and neon ionized with electrodes. The interaction of this plasma with the electrodes generates ultraviolet photons, which then excite the phosphor coating on the front of the display.
Xenon is used as a "starter gas" in Sodium vapor lamp, high pressure sodium lamps. It has the lowest thermal conductivity and lowest ionization potential of all the non-radioactive noble gases. As a noble gas, it does not interfere with the chemical reactions occurring in the operating lamp. The low thermal conductivity minimizes thermal losses in the lamp while in the operating state, and the low ionization potential causes the breakdown voltage of the gas to be relatively low in the cold state, which allows the lamp to be more easily started.
Lasers
In 1962, a group of researchers at Bell Labs, Bell Laboratories discovered laser action in xenon, and later found that the laser gain was improved by adding helium to the lasing medium. The firstexcimer laser
An excimer laser, sometimes more correctly called an exciplex laser, is a form of ultraviolet laser which is commonly used in the production of microelectronic devices, semiconductor based integrated circuits or "chips", eye surgery, and micr ...
used a xenon dimer (Xe2) energized by a beam of electrons to produce stimulated emission at an ultraviolet wavelength of 176 nanometre, nm.
Xenon chloride and xenon fluoride have also been used in excimer (or, more accurately, exciplex) lasers.
Medical
Anesthesia
Xenon has been used as a general anesthetic, but it is more expensive than conventional anesthetics. Xenon interacts with many different receptors and ion channels, and like many theoretically multi-modal inhalation anesthetics, these interactions are likely complementary. Xenon is a high-affinity glycine-site NMDA receptor antagonist. However, xenon is different from certain other NMDA receptor antagonists in that it is not neurotoxicity, neurotoxic and it inhibits the neurotoxicity of ketamine and nitrous oxide (N2O), while actually producing neuroprotection, neuroprotective effects. Unlike ketamine and nitrous oxide, xenon does not stimulate a dopamine efflux in the nucleus accumbens. Like nitrous oxide and cyclopropane, xenon activates the two-pore domain potassium channel KCNK2, TREK-1. A related channel KCNK9, TASK-3 also implicated in the actions of inhalation anesthetics is insensitive to xenon. Xenon inhibits nicotinic acetylcholine Alpha-4 beta-2 nicotinic receptor, α4β2 receptors which contribute to spinally mediated analgesia. Xenon is an effective inhibitor of Plasma membrane Ca2+ ATPase, plasma membrane Ca2+ ATPase. Xenon inhibits Ca2+ ATPase by binding to a hydrophobic pore within the enzyme and preventing the enzyme from assuming active conformations. Xenon is a competitive inhibitor of the serotonin 5-HT3 receptor, 5-HT3 receptor. While neither anesthetic nor antinociceptive, this reduces anesthesia-emergent nausea and vomiting. Xenon has a minimum alveolar concentration (MAC) of 72% at age 40, making it 44% more potent than N2O as an anesthetic. Thus, it can be used with oxygen in concentrations that have a lower risk of Hypoxia (medical), hypoxia. Unlike nitrous oxide, xenon is not a greenhouse gas and is viewed as environmentally friendly. Though recycled in modern systems, xenon vented to the atmosphere is only returning to its original source, without environmental impact.Neuroprotectant
Xenon induces robust cardioprotection and neuroprotection through a variety of mechanisms. Through its influence on Ca2+, K+, ATP-sensitive potassium channel, KATP\HIF, and NMDA antagonism, xenon is neuroprotective when administered before, during and after Ischemia, ischemic insults. Xenon is a high affinity antagonist at the NMDA receptor glycine site. Xenon is cardioprotective in ischemia-reperfusion conditions by inducing Pharmacology, pharmacologic non-ischemic preconditioning. Xenon is cardioprotective by activating PKC-epsilon and downstream p38-MAPK. Xenon mimics neuronal ischemic preconditioning by activating ATP sensitive potassium channels. Xenon allosterically reduces ATP mediated channel activation inhibition independently of the sulfonylurea receptor1 subunit, increasing KATP open-channel time and frequency.Sports doping
Inhaling a xenon/oxygen mixture activates production of the transcription factor HIF1A, HIF-1-alpha, which may lead to increased production of erythropoietin. The latter hormone is known to increase red blood cell production and athletic performance. Reportedly, doping with xenon inhalation has been used in Russia since 2004 and perhaps earlier. On August 31, 2014, the World Anti Doping Agency (WADA) added xenon (andargon
Argon is a chemical element with the symbol Ar and atomic number 18. It is in group 18 of the periodic table and is a noble gas. Argon is the third-most abundant gas in Earth's atmosphere, at 0.934% (9340 ppmv). It is more than twice as a ...
) to the list of prohibited substances and methods, although no reliable doping tests for these gases have yet been developed. In addition, effects of xenon on erythropoietin production in humans have not been demonstrated, so far.
Imaging
gamma ray, Gamma emission from the radioisotope 133Xe of xenon can be used to image the heart, lungs, and brain, for example, by means of single photon emission computed tomography. 133Xe has also been used to measure blood flow. Xenon, particularly hyperpolarized 129Xe, is a useful contrast agent for MRI, magnetic resonance imaging (MRI). In the gas phase, it can image cavities in a porous sample, alveoli in lungs, or the flow of gases within the lungs. Because xenon is soluble both in water and in hydrophobic solvents, it can image various soft living tissues. Xenon-129 is currently being used as a visualization agent in MRI scans. When a patient inhales hyperpolarized xenon-129 ventilation and gas exchange in the lungs can be imaged and quantified. Unlike xenon-133, xenon-129 is non-ionizing and is safe to be inhaled with no adverse effects.Surgery
The xenon chlorideexcimer laser
An excimer laser, sometimes more correctly called an exciplex laser, is a form of ultraviolet laser which is commonly used in the production of microelectronic devices, semiconductor based integrated circuits or "chips", eye surgery, and micr ...
has certain dermatological uses.
NMR spectroscopy
Because of the xenon atom's large, flexible outer electron shell, the nuclear magnetic resonance, NMR spectrum changes in response to surrounding conditions and can be used to monitor the surrounding chemical circumstances. For instance, xenon dissolved in water, xenon dissolved in hydrophobic solvent, and xenon associated with certain proteins can be distinguished by NMR. Hyperpolarized xenon can be used by Surface science, surface chemists. Normally, it is difficult to characterize surfaces with NMR because signals from a surface are overwhelmed by signals from the atomic nuclei in the bulk of the sample, which are much more numerous than surface nuclei. However, nuclear spins on solid surfaces can be selectively polarized by Proton Enhanced Nuclear Induction Spectroscopy, transferring spin polarization to them from hyperpolarized xenon gas. This makes the surface signals strong enough to measure and distinguish from bulk signals.Other
In Nuclear physics, nuclear energy studies, xenon is used in bubble chambers, probes, and in other areas where a high Molecular mass, molecular weight and inert chemistry is desirable. A by-product of nuclear weapon testing is the release of radioactive isotopes of xenon, xenon-133 and xenon-135. These isotopes are monitored to ensure compliance with nuclear Test Ban Treaty (disambiguation), test ban treaties, and to confirm nuclear tests by states such as North Korea. Liquid xenon is used in Calorimeter (particle physics), calorimeters to measure gamma rays, and as a detector of hypothetical
Liquid xenon is used in Calorimeter (particle physics), calorimeters to measure gamma rays, and as a detector of hypothetical weakly interacting massive particles
Weakly interacting massive particles (WIMPs) are hypothetical particles that are one of the proposed candidates for dark matter.
There exists no formal definition of a WIMP, but broadly, a WIMP is a new elementary particle which interacts via gra ...
, or WIMPs. When a WIMP collides with a xenon nucleus, theory predicts it will impart enough energy to cause ionization and Scintillation (physics), scintillation. Liquid xenon is useful for these experiments because its density makes dark matter interaction more likely and it permits a quiet detector through self-shielding.
Xenon is the preferred propellant
A propellant (or propellent) is a mass that is expelled or expanded in such a way as to create a thrust or other motive force in accordance with Newton's third law of motion, and "propel" a vehicle, projectile, or fluid payload. In vehicles, the ...
for ion propulsion of spacecraft because it has low ionization potential per Atomic mass, atomic weight and can be stored as a liquid at near room temperature
Colloquially, "room temperature" is a range of air temperatures that most people prefer for indoor settings. It feels comfortable to a person when they are wearing typical indoor clothing. Human comfort can extend beyond this range depending on ...
(under high pressure), yet easily evaporated to feed the engine. Xenon is inert, environmentally friendly, and less corrosive to an ion engine than other fuels such as Mercury (element), mercury or caesium. Xenon was first used for satellite ion engines during the 1970s. It was later employed as a propellant for JPL's Deep Space 1 probe, Europe's SMART-1 spacecraft and for the three ion propulsion engines on NASA's Dawn Spacecraft.
Chemically, the perxenate compounds are used as oxidizing agents in analytical chemistry. Xenon difluoride is used as an etchant for silicon, particularly in the production of microelectromechanical systems (MEMS). The anticancer drug Fluorouracil, 5-fluorouracil can be produced by reacting xenon difluoride with uracil. Xenon is also used in X-ray crystallography, protein crystallography. Applied at pressures from 0.5 to 5 Pascal (unit), MPa (5 to 50 atmosphere (unit), atm) to a protein crystal, xenon atoms bind in predominantly Hydrophobe, hydrophobic cavities, often creating a high-quality, isomorphous, heavy-atom derivative that can be used for solving the phase problem.
Precautions
Xenon gas can be safely kept in normal sealed glass or metal containers atstandard temperature and pressure
Standard temperature and pressure (STP) are standard sets of conditions for experimental measurements to be established to allow comparisons to be made between different sets of data. The most used standards are those of the International Union ...
. However, it readily dissolves in most plastics and rubber, and will gradually escape from a container sealed with such materials. Xenon is non-toxic, although it does dissolve in blood and belongs to a select group of substances that penetrate the blood–brain barrier, causing mild to full surgical anesthesia when inhaled in high concentrations with oxygen.
The speed of sound in xenon gas (169 m/s) is less than that in air because the average velocity of the heavy xenon atoms is less than that of nitrogen and oxygen molecules in air. Hence, xenon vibrates more slowly in the vocal tract, vocal cords when exhaled and produces lowered voice tones (low-frequency-enhanced sounds, but the fundamental frequency or Pitch (music), pitch doesn't change), an effect opposite to the high-toned voice produced in helium. Specifically, when the vocal tract is filled with xenon gas, its natural resonant frequency becomes lower than when it's filled with air. Thus, the low frequencies of the sound wave produced by the same direct vibration of the vocal cords would be enhanced, resulting in a change of the timbre of the sound amplified by the vocal tract. Like helium, xenon does not satisfy the body's need for oxygen, and it is both a simple asphyxiant gas, asphyxiant and an anesthetic more powerful than nitrous oxide; consequently, and because xenon is expensive, many universities have prohibited the voice stunt as a general chemistry demonstration. The gas sulfur hexafluoride is similar to xenon in molecular weight (146 versus 131), less expensive, and though an asphyxiant, not toxic or anesthetic; it is often substituted in these demonstrations.
Dense gases such as xenon and sulfur hexafluoride can be breathed safely when mixed with at least 20% oxygen. Xenon at 80% concentration along with 20% oxygen rapidly produces the unconsciousness of general anesthesia (and has been used for this, as discussed above). Breathing mixes gases of different densities very effectively and rapidly so that heavier gases are purged along with the oxygen, and do not accumulate at the bottom of the lungs. There is, however, a danger associated with any heavy gas in large quantities: it may sit invisibly in a container, and a person who enters an area filled with an odorless, colorless gas may be asphyxiated without warning. Xenon is rarely used in large enough quantities for this to be a concern, though the potential for danger exists any time a tank or container of xenon is kept in an unventilated space.
Water-soluble xenon compounds such as monosodium xenate are moderately toxic, but have a very short half-life of the body — intravenously injected xenate is reduced to elemental xenon in about a minute.
See also
* Buoyant levitation * Noble gases * Penning mixtureReferences
External links
Xenon
at ''The Periodic Table of Videos'' (University of Nottingham)
USGS Periodic Table – Xenon
{{Authority control Xenon, Chemical elements Noble gases Dissociative drugs General anesthetics Glycine receptor agonists Industrial gases Nicotinic antagonists NMDA receptor antagonists 5-HT3 antagonists ATPase inhibitors Rocket propellants