|
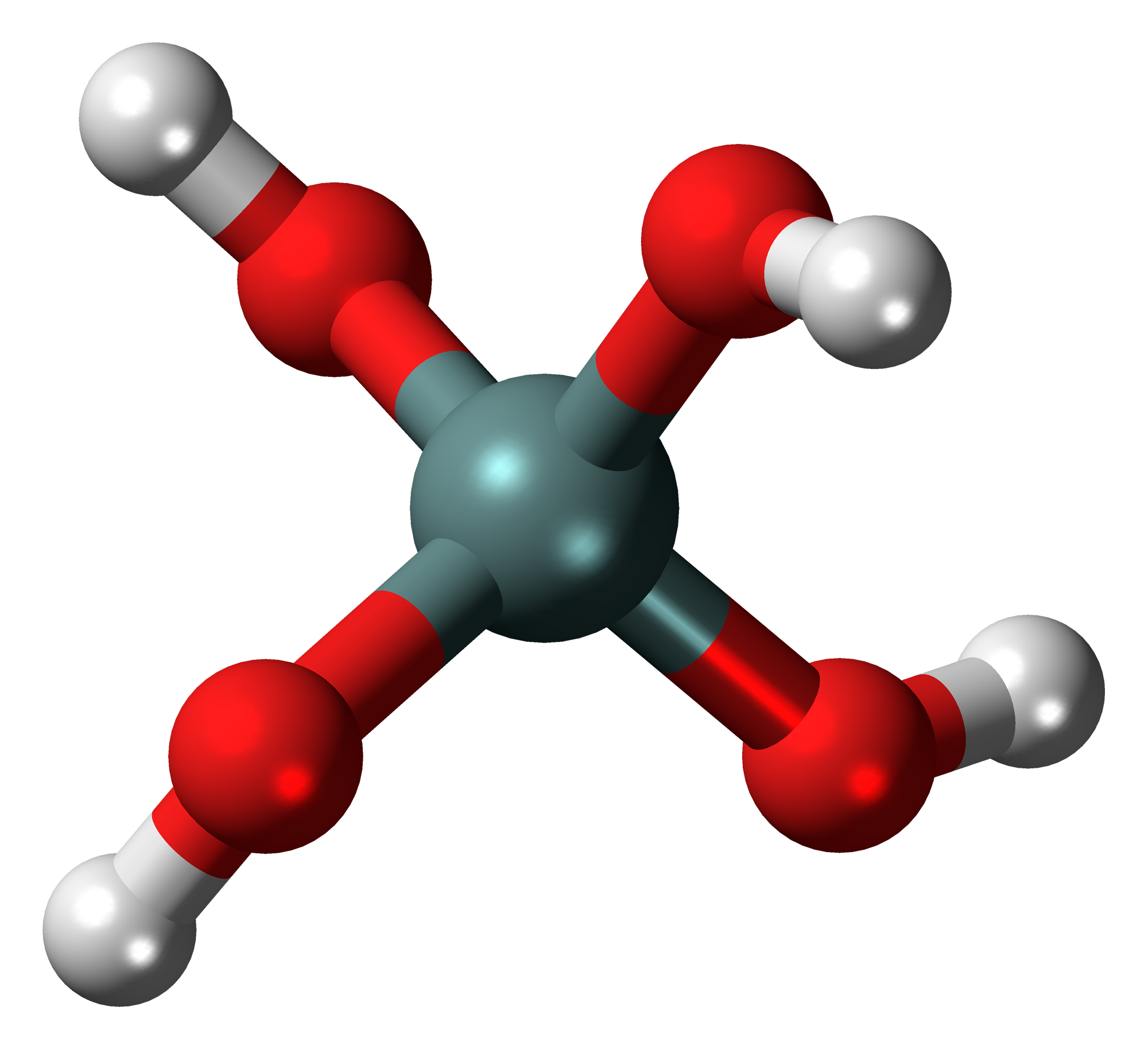
Pyrosilicic Acid
Pyrosilicic acid is the chemical compound with formula or . It is one of the silicic acids and has pyrosilicate as its conjugate base. It was synthesized, using nonaqueous solutions, in 2017. : Pyrosilicic acid may be present in sea water and other natural waters at very low concentration.Katsumi Goto (1956): "Effect of pH on Polymerization of Silicic Acid". ''Journal of Physical Chemistry'', volume 60, issue 7, pages 1007–1008. M. F. Bechtold (1955): "Polymerization and Properties of Dilute Aqueous Silicic Acid from Cation Exchange". ''Journal of Physical Chemistry'', volume 59, issue 6, pages 532–541. Compounds formally derived from it, such as sodium pyrosilicate, are found in the sorosilicate minerals. References {{reflist Oxoacids Silicon compounds ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pyrosilicate
A pyrosilicate is a type of chemical compound; either an ionic compound that contains the pyrosilicate anion , or an organic compound with the hexavalent ≡-O-≡ group. The anion is also called disilicateViktor Renman (2017): "Structural and Electrochemical Relations in Electrode Materials for Rechargeable Batteries", Doctoral Thesis, Uppsala University, Department of Chemistry. ORCID: 0000-0001-8739-4054 Structure The pyrosilicate anion can be described as two tetrahedra that share a vertex (an oxygen atom). The vertices that are not shared carry a negative charge each. The structure of solid sodium pyrosilicate was described by Volker Kahlenberg and others in 2010.Volker Kahlenberg, Thomas Langreiter, and Erik Arroyabe (2010): " – The Missing Structural Link among Alkali Pyrosilicates". ''Zeitschrift für anorganishe und allgemeine Chemie'' (''Journal for Inorganic and General Chemistry''), volume 636, issue 11, pages 1974-1979. Yuri Smolin and Yuri Shepelev determi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chemical Compound
A chemical compound is a chemical substance composed of many identical molecules (or molecular entities) containing atoms from more than one chemical element held together by chemical bonds. A molecule consisting of atoms of only one element is therefore not a compound. A compound can be transformed into a different substance by a chemical reaction, which may involve interactions with other substances. In this process, bonds between atoms may be broken or new bonds formed or both. There are four major types of compounds, distinguished by how the constituent atoms are bonded together. Molecular compounds are held together by covalent bonds; ionic compounds are held together by ionic bonds; intermetallic compounds are held together by metallic bonds; coordination complexes are held together by coordinate covalent bonds. Non-stoichiometric compounds form a disputed marginal case. A chemical formula specifies the number of atoms of each element in a compound molecule, usin ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Silicic Acid
In chemistry, a silicic acid () is any chemical compound containing the element silicon attached to oxide () and hydroxyl () groups, with the general formula or, equivalently, . Orthosilicic acid is a representative example. Silicic acids are rarely observed in isolation, but are thought to exist in aqueous solutions, including seawater, and play a role in biomineralization. They are typically colorless weak acids that are sparingly soluble in water. Like the silicate anions, which are their better known conjugate bases, silicic acids are proposed to be oligomeric or polymeric. No Monomer, simple silicic acid has ever been identified, since these species are primarily of theoretical interest. Depending on the number of silicon atoms present, there are mono- and polysilicic (di-, tri-, tetrasilicic, etc.) acids. Well defined silicic acids have not been obtained in a form that has been characterized by X-ray crystallography. Examples Reactions Silicic acids can be seen as hy ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pyrosilicate
A pyrosilicate is a type of chemical compound; either an ionic compound that contains the pyrosilicate anion , or an organic compound with the hexavalent ≡-O-≡ group. The anion is also called disilicateViktor Renman (2017): "Structural and Electrochemical Relations in Electrode Materials for Rechargeable Batteries", Doctoral Thesis, Uppsala University, Department of Chemistry. ORCID: 0000-0001-8739-4054 Structure The pyrosilicate anion can be described as two tetrahedra that share a vertex (an oxygen atom). The vertices that are not shared carry a negative charge each. The structure of solid sodium pyrosilicate was described by Volker Kahlenberg and others in 2010.Volker Kahlenberg, Thomas Langreiter, and Erik Arroyabe (2010): " – The Missing Structural Link among Alkali Pyrosilicates". ''Zeitschrift für anorganishe und allgemeine Chemie'' (''Journal for Inorganic and General Chemistry''), volume 636, issue 11, pages 1974-1979. Yuri Smolin and Yuri Shepelev determi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Conjugate Base
A conjugate acid, within the Brønsted–Lowry acid–base theory, is a chemical compound formed when an acid gives a proton () to a base—in other words, it is a base with a hydrogen ion added to it, as it loses a hydrogen ion in the reverse reaction. On the other hand, a conjugate base is what remains after an acid has donated a proton during a chemical reaction. Hence, a conjugate base is a substance formed by the removal of a proton from an acid, as it can gain a hydrogen ion in the reverse reaction. Because some acids can give multiple protons, the conjugate base of an acid may itself be acidic. In summary, this can be represented as the following chemical reaction: \text + \text \; \ce \; \text + \text Johannes Nicolaus Brønsted and Martin Lowry introduced the Brønsted–Lowry theory, which said that any compound that can give a proton to another compound is an acid, and the compound that receives the proton is a base. A proton is a subatomic particle in the ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
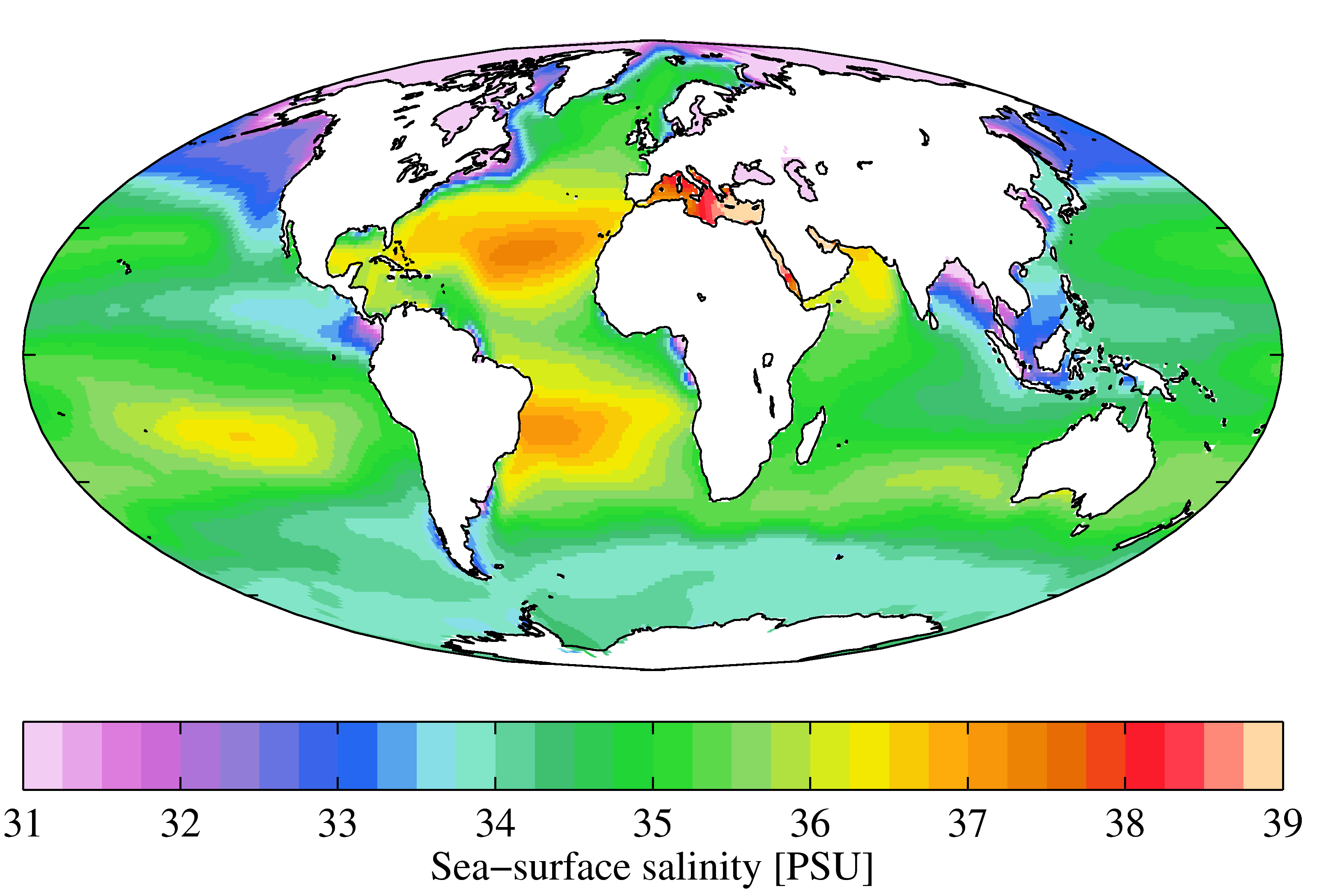
Sea Water
Seawater, or sea water, is water from a sea or ocean. On average, seawater in the world's oceans has a salinity of about 3.5% (35 g/L, 35 ppt, 600 mM). This means that every kilogram (roughly one liter by volume) of seawater has approximately of dissolved salts (predominantly sodium () and chloride () ions). The average density at the surface is 1.025 kg/L. Seawater is denser than both fresh water and pure water (density 1.0 kg/L at ) because the dissolved salts increase the mass by a larger proportion than the volume. The freezing point of seawater decreases as salt concentration increases. At typical salinity, it freezes at about . The coldest seawater still in the liquid state ever recorded was found in 2010, in a stream under an Antarctic glacier: the measured temperature was . Seawater pH is typically limited to a range between 7.5 and 8.4. However, there is no universally accepted reference pH-scale for seawater and the difference between measurements b ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sodium Pyrosilicate
Sodium pyrosilicate is the chemical compound . It is one of the sodium silicates, specifically a pyrosilicate, formally a salt of the unstable pyrosilicic acid .Myron C Waddell (1932): "Process of purifying technical sodium pyrosilicate hydrates". US patent US1931364A.J. F. Schairer and N. L. Bowen (1956): "The system ——". ''American Journal of Science'', volume 254, issue 3, pages 129-195 Structure The anhydrous solid has the triclinic crystal structure, with space group P (a = 5.8007(8) Å, b = 11.5811(15) Å, c = 23.157(3) Å, α = 89.709(10)°, β = 88.915(11)°, γ = 89.004(11)°, V = 1555.1(4) Å3, Z = 8, Dx = 2.615 g·cm−3, μ(Mo‐Kα) = 7.94 cm−1). The anions are arranged in layers parallel to the (100) plane, with the sodium cations distributed in 24 distinct crystallographic positions, coordinated by 4 to 6 near oxygen atoms. Some of the 4-coordinated sodium atoms can be interpreted as parallel columns of edge-sharing tetrahedra. The columnar arrangem ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sorosilicate
Silicate minerals are rock-forming minerals made up of silicate groups. They are the largest and most important class of minerals and make up approximately 90 percent of Earth's crust. In mineralogy, the crystalline forms of silica (silicon dioxide, ) are usually considered to be tectosilicates, and they are classified as such in the Dana system (75.1). However, the Nickel-Strunz system classifies them as oxide minerals (4.DA). Silica is found in nature as the mineral quartz, and its polymorphs. On Earth, a wide variety of silicate minerals occur in an even wider range of combinations as a result of the processes that have been forming and re-working the crust for billions of years. These processes include partial melting, crystallization, fractionation, metamorphism, weathering, and diagenesis. Living organisms also contribute to this geologic cycle. For example, a type of plankton known as diatoms construct their exoskeletons ("frustules") from silica extracted from seawat ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Oxoacids
An oxyacid, oxoacid, or ternary acid is an acid that contains oxygen. Specifically, it is a compound that contains hydrogen, oxygen, and at least one other element, with at least one hydrogen atom bonded to oxygen that can dissociate to produce the H+ cation and the anion of the acid. Description Under Lavoisier's original theory, all acids contained oxygen, which was named from . It was later discovered that some acids, notably hydrochloric acid, did not contain oxygen and so acids were divided into oxo-acids and these new hydroacids. All oxyacids have the acidic hydrogen bound to an oxygen atom, so bond strength (length) is not a factor, as it is with binary nonmetal hydrides. Rather, the electronegativity of the central atom and the number of oxygen atoms determine oxyacid acidity. For oxyacids with the same central atom, acid strength increases with the number of oxygen atoms attached to it. With the same number of oxygen atoms attached to it, acid strength increases with ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |