|
Metal Sulfate
Transition metal sulfate complexes or sulfato complexes are coordination complexes with one or more sulfate ligands. Being the conjugate base of a strong acid (sulfuric acid), sulfate is not basic. It is more commonly a counterion in coordination chemistry, not a ligand. Bonding modes Sulfate binds to metals through one, two, three, or all four oxygen atoms. Among the handful of complexes containing sulfate (or sulfato) ligands, most examples feature unidentate or chelating bidentate sulfate. Well characterized xamples are found with cobalt(III) ammines since these complexes are exchange inert. Monodentate sulfate is found in [Co(tren)(NH3)(SO4)]+ (tren = tris(2-aminoethyl)amine) Although is unknown, forms instead (en = ethylenediamine). Bidentate sulfate is observed crystallographically in . Sulfate function as a tridentate bridging ligand . All four oxygen atoms of sulfate bond to metals in some Dawson-type polyoxometalates, e.g. [S2Mo18O62]4-. Sulfate as a counterion ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chrome Alum
Chrome alum or Chromium(III) potassium sulfate is the potassium double sulfate of chromium. Its chemical formula is KCr(SO4)2 and it is commonly found in its dodecahydrate form as KCr(SO4)2·12(H2O). It is used in leather tanning. Production and properties Chromium alum is produced from chromate salts or from ferrochromium alloys. Concentrated aqueous solutions of potassium dichromate can be reduced, usually with sulfur dioxide but also with alcohols or formaldehyde, in the presence of sulfuric acid at temperatures <40 °C. Alternatively and less commonly, ferrochromium alloys can be dissolved in sulfuric acid and, after precipitation of the ferrous sulfate, the chrome alum crystallizes upon addition of potassium sulfate. Chromium alum crystallizes in regular octahedra with flattened cor ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ligands
In coordination chemistry, a ligand is an ion or molecule with a functional group that binds to a central metal atom to form a coordination complex. The bonding with the metal generally involves formal donation of one or more of the ligand's electron pairs, often through Lewis bases. The nature of metal–ligand bonding can range from covalent to ionic. Furthermore, the metal–ligand bond order can range from one to three. Ligands are viewed as Lewis bases, although rare cases are known to involve Lewis acidic "ligands". Metals and metalloids are bound to ligands in almost all circumstances, although gaseous "naked" metal ions can be generated in a high vacuum. Ligands in a complex dictate the reactivity of the central atom, including ligand substitution rates, the reactivity of the ligands themselves, and redox. Ligand selection requires critical consideration in many practical areas, including bioinorganic and medicinal chemistry, homogeneous catalysis, and environme ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
1,10-Phenanthroline
1,10-Phenanthroline (phen) is a heterocyclic organic compound. It is a white solid that is soluble in organic solvents. The 1,10 refers to the location of the nitrogen atoms that replace CH's in the hydrocarbon called phenanthrene. Abbreviated "phen", or sometimes "o-phen" for ortho-phenanthroline, it is used as a ligand in coordination chemistry, forming strong complexes with most metal ions.Luman, C.R. and Castellano, F.N. (2003) "Phenanthroline Ligands" in Comprehensive Coordination Chemistry II. Elsevier. . It is often sold as the monohydrate. Synthesis Phenanthroline can be prepared by two successive Skraup reactions of glycerol with ''o''-phenylenediamine, catalyzed by sulfuric acid, and an oxidizing agent, traditionally aqueous arsenic acid or nitrobenzene. Dehydration of glycerol gives acrolein which condenses with the amine followed by a cyclization. Reactions Oxidation of 1,10-phenanthroline with a mixture of nitric and sulfuric acids gives 1,10-phenanthroline-5,6-d ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
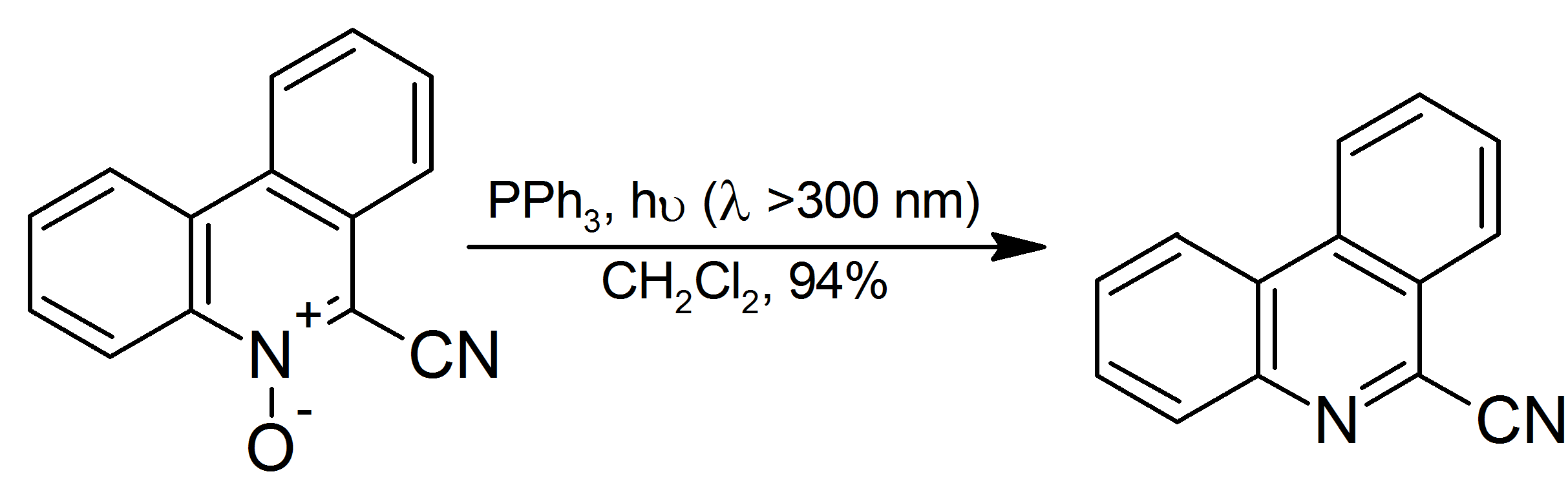
Triphenylphosphine
Triphenylphosphine (IUPAC name: triphenylphosphane) is a common organophosphorus compound with the formula P(C6H5)3 and often abbreviated to P Ph3 or Ph3P. It is versatile compound that is widely used as a reagent in organic synthesis and as a ligand for transition metal complexes, including ones that serve as catalysts in organometallic chemistry. PPh3 exists as relatively air stable, colorless crystals at room temperature. It dissolves in non-polar organic solvents such as benzene and diethyl ether. Preparation and structure Triphenylphosphine can be prepared in the laboratory by treatment of phosphorus trichloride with phenylmagnesium bromide or phenyllithium. The industrial synthesis involves the reaction between phosphorus trichloride, chlorobenzene, and sodium: :PCl3 + 3 PhCl + 6 Na → PPh3 + 6 NaCl Triphenylphosphine crystallizes in triclinic and monoclinic modification. In both cases, the molecule adopts a pyramidal structure with propeller-like arrangement of the thre ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sulfur Dioxide
Sulfur dioxide (IUPAC-recommended spelling) or sulphur dioxide (traditional Commonwealth English) is the chemical compound with the formula . It is a colorless gas with a pungent smell that is responsible for the odor of burnt matches. It is released naturally by volcanic activity and is produced as a by-product of metals refining and the burning of Sour gas, sulfur-Sour crude oil, bearing fossil fuels. Sulfur dioxide is somewhat toxic to humans, although only when inhaled in relatively large quantities for a period of several minutes or more. It was known to medieval alchemy, alchemists as "volatile spirit of sulfur". Structure and bonding SO2 is a bent molecule with ''C''2v Point groups in three dimensions, symmetry point group. A valence bond theory approach considering just ''s'' and ''p'' orbitals would describe the bonding in terms of resonance (chemistry), resonance between two resonance structures. The sulfur–oxygen bond has a bond order of 1.5. There is support f ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
CSD CIF HUXMIX
CSD may refer to: Finance * Central securities depository * Confederate States Dollar * Serbian dinar, by previous ISO 4217 code Organizations Education * California School for the Deaf (other), several institutions * Canyons School District, in Utah, US * Cheltenham School District, in Pennsylvania, US * Christina School District, in Delaware, US * Cleveland School District, in Mississippi, US * Cordova School District, in Alaska, US Other organizations * Canteen Stores Department (India), a chain of stores operated by the Indian Ministry of Defence at military bases * CSD Pakistan (Canteen Stores Department), a chain of stores operated by the Pakistani Ministry of Defence * Chartered Society of Designers, a British learned society for various kinds of design work * Commission on Sustainable Development (1992–2013), a former UN agency * Communication Service for the Deaf, an American non-profit company providing ASL services * Congress of Democratic Trade Uni ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Copper(II) Sulfate
Copper(II) sulfate is an inorganic compound with the chemical formula . It forms hydrates , where ''n'' can range from 1 to 7. The pentahydrate (''n'' = 5), a bright blue crystal, is the most commonly encountered hydrate of copper(II) sulfate, while its anhydrous form is white. Older names for the pentahydrate include blue vitriol, bluestone, vitriol of copper,Antoine-François de Fourcroy, tr. by Robert Heron (1796) "Elements of Chemistry, and Natural History: To which is Prefixed the Philosophy of Chemistry". J. Murray and others, Edinburgh. Page 348. and Roman vitriol.Oxford University Press,Roman vitriol, Oxford Living Dictionaries. Accessed on 2016-11-13 It exothermically dissolves in water to give the aquo complex , which has octahedral molecular geometry. The structure of the solid pentahydrate reveals a polymeric structure wherein copper is again octahedral but bound to four water ligands. The centers are interconnected by sulfate anions to form chains. Preparation and ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Aquo Complex
In chemistry, metal aquo complexes are coordination compounds containing metal ions with only water as a ligand. These complexes are the predominant species in aqueous solutions of many metal salts, such as metal nitrates, sulfates, and perchlorates. They have the general stoichiometry . Their behavior underpins many aspects of environmental, biological, and industrial chemistry. This article focuses on complexes where water is the only ligand (" homoleptic aquo complexes"), but of course many complexes are known to consist of a mix of aquo and other ligands. Stoichiometry and structure Hexa-aquo complexes Most aquo complexes are mono-nuclear, with the general formula , with or 3; they have an octahedral structure. The water molecules function as Lewis bases, donating a pair of electrons to the metal ion and forming a dative covalent bond with it. Typical examples are listed in the following table. Tutton's salts are crystalline compounds with the generic formula (where ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
X-ray Crystallography
X-ray crystallography is the experimental science of determining the atomic and molecular structure of a crystal, in which the crystalline structure causes a beam of incident X-rays to Diffraction, diffract in specific directions. By measuring the angles and intensities of the X-ray diffraction, a crystallography, crystallographer can produce a three-dimensional picture of the density of electrons within the crystal and the positions of the atoms, as well as their chemical bonds, crystallographic disorder, and other information. X-ray crystallography has been fundamental in the development of many scientific fields. In its first decades of use, this method determined the size of atoms, the lengths and types of chemical bonds, and the atomic-scale differences between various materials, especially minerals and alloys. The method has also revealed the structure and function of many biological molecules, including vitamins, drugs, proteins and nucleic acids such as DNA. X-ray crystall ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Alum
An alum () is a type of chemical compound, usually a hydrated double salt, double sulfate salt (chemistry), salt of aluminium with the general chemical formula, formula , such that is a valence (chemistry), monovalent cation such as potassium or ammonium. By itself, "alum" often refers to potassium alum, with the formula . Other alums are named after the monovalent ion, such as sodium alum and ammonium alum. The name "alum" is also used, more generally, for salts with the same formula and structure, except that aluminium is replaced by another Valence (chemistry), trivalent metal ion like chromium#Chromium(III), chromium, or sulfur is replaced by another chalcogen like selenium. The most common of these analogs is chrome alum . In most industries, the name "alum" (or "papermaker's alum") is used to refer to aluminium sulfate, , which is used for most industrial flocculation (the variable is an integer whose size depends on the amount of water absorbed into the alum). For medi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Coordination Complex
A coordination complex is a chemical compound consisting of a central atom or ion, which is usually metallic and is called the ''coordination centre'', and a surrounding array of chemical bond, bound molecules or ions, that are in turn known as ''ligands'' or complexing agents. Many metal-containing chemical compound, compounds, especially those that include transition metals (elements like titanium that belong to the periodic table's d-block), are coordination complexes. Nomenclature and terminology Coordination complexes are so pervasive that their structures and reactions are described in many ways, sometimes confusingly. The atom within a ligand that is bonded to the central metal atom or ion is called the donor atom. In a typical complex, a metal ion is bonded to several donor atoms, which can be the same or different. A Ligand#Polydentate and polyhapto ligand motifs and nomenclature, polydentate (multiple bonded) ligand is a molecule or ion that bonds to the central atom ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |