|
Triphenylphosphine
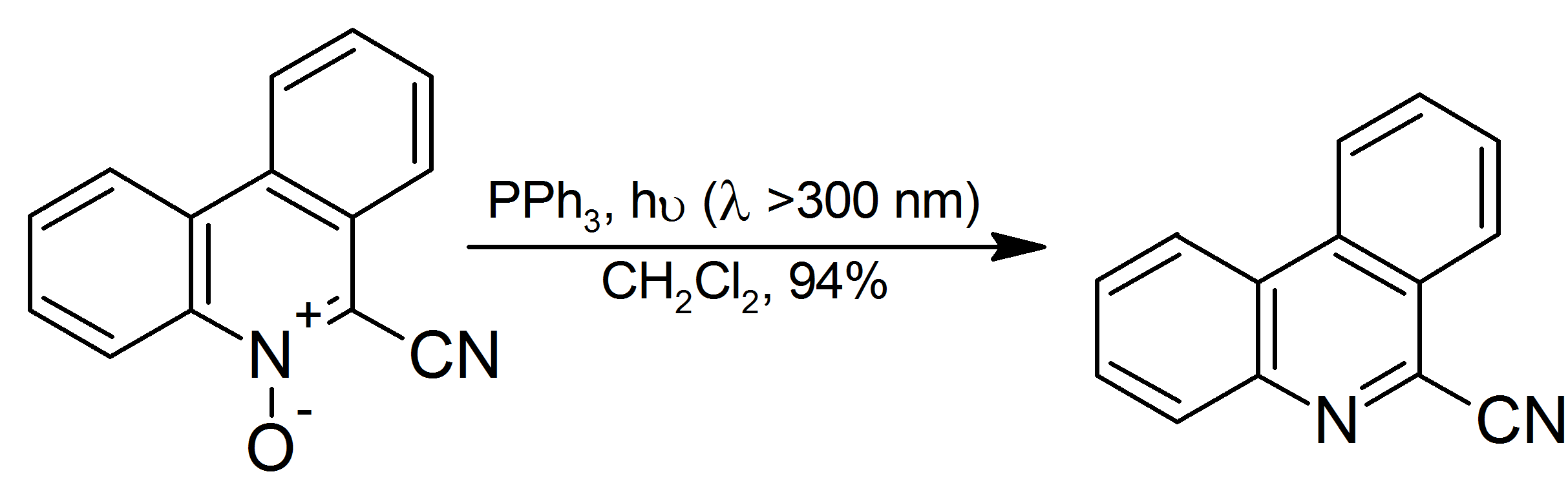
Triphenylphosphine (IUPAC name: triphenylphosphane) is a common organophosphorus compound with the formula P(C6H5)3 and often abbreviated to P Ph3 or Ph3P. It is versatile compound that is widely used as a reagent in organic synthesis and as a ligand for transition metal complexes, including ones that serve as catalysts in organometallic chemistry. PPh3 exists as relatively air stable, colorless crystals at room temperature. It dissolves in non-polar organic solvents such as benzene and diethyl ether. Preparation and structure Triphenylphosphine can be prepared in the laboratory by treatment of phosphorus trichloride with phenylmagnesium bromide or phenyllithium. The industrial synthesis involves the reaction between phosphorus trichloride, chlorobenzene, and sodium: :PCl3 + 3 PhCl + 6 Na → PPh3 + 6 NaCl Triphenylphosphine crystallizes in triclinic and monoclinic modification. In both cases, the molecule adopts a pyramidal structure with propeller-like arrangement of the thre ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tetrakis(triphenylphosphine)palladium(0)
Tetrakis(triphenylphosphine)palladium(0) (sometimes called quatrotriphenylphosphine palladium) is the chemical compound d(P(C6H5)3)4 often abbreviated Pd( PPh3)4, or rarely PdP4. It is a bright yellow crystalline solid that becomes brown upon decomposition in air. Structure and properties The four phosphorus atoms are at the corners of a tetrahedron surrounding the palladium(0) center. This structure is typical for four-coordinate 18 e− complexes. The corresponding complexes Ni(PPh3)4 and Pt(PPh3)4 are also well known. Such complexes reversibly dissociate PPh3 ligands in solution, so reactions attributed to Pd(PPh3)4 often in fact arise from Pd(PPh3)3 or even Pd(PPh3)2. Preparation Tetrakis(triphenylphosphine)palladium(0) was first prepared by Lamberto Malatesta et al. in the 1950s by reduction of sodium chloropalladate with hydrazine in the presence of the phosphine. It is commercially available, but can be prepared in two steps from Pd(II) precursors: :PdCl2 + 2 PPh3 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Triphenylphosphine Dichloride
Triphenylphosphine dichloride is an organophosphorus compound with the chemical formula , often abbreviated as , where Ph is phenyl. It is a chlorinating agent widely used in organic chemistry. Applications include the conversion of alcohols and ethers to alkyl chlorides, the cleavage of epoxides to vicinal dichlorides and the chlorination of carboxylic acids to acyl chlorides. Structure In polar solvents such as acetonitrile, adopts an ionic phosphonium salt structure, (chlorotriphenylphosphonium chloride), whereas in non-polar solvents like diethyl ether it exists as a non-solvated trigonal bipyramidal molecule. Two species can also adopt an unusual dinuclear ionic structure—both interacting with a via long Cl–Cl contacts. File:Chlorotriphenylphosphonium-chloride-2D.png File:Chlorotriphenylphosphonium-chloride-DCM-solvate-from-xtal-3D-balls.png File:Chlorotriphenylphosphonium-chloride-DCM-solvate-from-xtal-3D-vdW.png Synthesis Triphenylphosphine dichloride ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tertiary Phosphine
Organophosphines are organophosphorus compounds with the formula PR''n''H3−''n'', where R is an organic substituent. These compounds can be classified according to the value of ''n'': primary phosphines (''n'' = 1), secondary phosphines (''n'' = 2), tertiary phosphines (''n'' = 3). All adopt pyramidal structures. Organophosphines are generally colorless, lipophilic liquids or solids. The parent of the organophosphines is phosphine (PH3). Annette Schier and Hubert Schmidbaur"P-Donor Ligands" in Encyclopedia of Inorganic Chemistry 2006, Wiley-VCH, Weinheim. 1° vs 2° vs 3° phosphines Organophophines are classified according to the number of organic substituents. Primary phosphines Primary (1°) phosphines, with the formula RPH2, in principle are derived by alkylation of phosphine. Some simple alkyl derivatives such as methylphosphine (CH3PH2) can be prepared by alkylation of phosphine in the presence of base: : (M = Li, Na, K) A more common syn ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Triphenylphosphine Oxide
Triphenylphosphine oxide (often abbreviated TPPO) is the organophosphorus compound with the formula , also written as or (Ph = ). It is one of the more common phosphine oxides. This colourless crystalline compound is a common but potentially useful waste product in reactions involving triphenylphosphine. It is a popular reagent to induce the crystallizing of chemical compounds. Structure and properties is structurally related to . As established by X-ray crystallography, the geometry around P is tetrahedral, and the P-O distance is 1.48 Å. Other modifications of have been found: For example, a monoclinic form crystalizes in the space group ''P''21/''c'' with Z = 4 and a = 15.066(1) Å, b = 9.037(2) Å, c = 11.296(3) Å, and β = 98.47(1)°.The orthorhombic modification crystallizes in the space group ''Pbca'' with Z = 4 and 29.089(3) Å, b = 9.1347(9), c = 11.261(1) Å. The oxygen center is relatively basic. The rigidity of the backbone and the basicity of the oxygen ce ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Organophosphorus Compound
Organophosphorus chemistry is the scientific study of the synthesis and properties of organophosphorus compounds, which are organic compounds containing phosphorus. They are used primarily in pest control as an alternative to chlorinated hydrocarbons that persist in the environment. Some organophosphorus compounds are highly effective insecticides, although some are extremely toxic to humans, including sarin and VX (nerve agent), VX nerve agents. Phosphorus, like nitrogen, is in pnictogen, group 15 of the periodic table, and thus phosphorus compounds and nitrogen compounds have many similar properties. The definition of organophosphorus compounds is variable, which can lead to confusion. In industrial and environmental chemistry, an organophosphorus compound need contain only an organic substituent, but need not have a direct phosphorus-carbon (P-C) bond. Thus a large proportion of pesticides (e.g., malathion), are often included in this class of compounds. Phosphorus can adopt a v ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phosphorus Trichloride
Phosphorus trichloride is an inorganic compound with the chemical formula PCl3. A colorless liquid when pure, it is an important industrial chemical, being used for the manufacture of phosphites and other organophosphorus compounds. It is toxic and reacts readily with water to release hydrogen chloride. History Phosphorus trichloride was first prepared in 1808 by the French chemists Joseph Louis Gay-Lussac and Louis Jacques Thénard by heating calomel (Hg2Cl2) with phosphorus. Later during the same year, the English chemist Humphry Davy produced phosphorus trichloride by burning phosphorus in chlorine gas. Preparation World production exceeds one-third of a million tonnes. Phosphorus trichloride is prepared industrially by the reaction of chlorine with white phosphorus, using phosphorus trichloride as the solvent. In this continuous process PCl3 is removed as it is formed in order to avoid the formation of PCl5. :P4 + 6 Cl2 → 4 PCl3 Structure and spectroscopy It has a t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Triphenylphosphine Selenide
Triphenylphosphine selenide is an organophosphorus compound with the formula (C6H5)3PSe. It is a white solid which is soluble in most organic solvents. The compound is used in the preparation of other selenium compounds and is itself prepared by the reaction of triphenylphosphine with potassium selenocyanate. Single crystals have been isolated with both monoclinic and triclinic structures (space groups: P21/c and P{{overline, 1 respectively); in both cases the geometry at phosphorus is tetrahedral. See also * Triphenylphosphine oxide * Triphenylphosphine sulfide Triphenylphosphine sulfide (IUPAC name: triphenyl-''λ''5-phosphanethione) is the organophosphorus compound with the formula , usually written (where Ph = phenyl). It is a colourless solid, which is soluble in a variety of organic solvents. Struc ... * Trioctylphosphine selenide References Organophosphanes Selenides Phenyl compounds ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Triphenylphosphine Sulfide
Triphenylphosphine sulfide (IUPAC name: triphenyl-''λ''5-phosphanethione) is the organophosphorus compound with the formula , usually written (where Ph = phenyl). It is a colourless solid, which is soluble in a variety of organic solvents. Structurally, the molecule resembles the corresponding oxide, with idealized C3 point group symmetry. It is weakly nucleophilic at the sulfur atom. Applications Organic synthesis Triphenylphosphine sulfide is useful for the conversion of epoxides to the corresponding episulfides: : It also reacts with ketenes to form thioketenes: : Analytical chemistry In analytical chemistry, triphenylphosphine is used for the analysis of certain kinds of sulfur compounds. Elemental sulfur (), as occurs in some oils, and labile organosulfur compounds, such as organic trisulfides, react with triphenylphosphine to give , which can be detected by gas chromatography Gas chromatography (GC) is a common type of chromatography used in analytical chemistry ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Organometallic Chemistry
Organometallic chemistry is the study of organometallic compounds, chemical compounds containing at least one chemical bond between a carbon atom of an organic molecule and a metal, including alkali, alkaline earth, and transition metals, and sometimes broadened to include metalloids like boron, silicon, and selenium, as well. Aside from bonds to organyl fragments or molecules, bonds to 'inorganic' carbon, like carbon monoxide (Metal carbonyl, metal carbonyls), cyanide, or carbide, are generally considered to be organometallic as well. Some related compounds such as transition metal hydrides and metal phosphine complexes are often included in discussions of organometallic compounds, though strictly speaking, they are not necessarily organometallic. The related but distinct term "metalorganics, metalorganic compound" refers to metal-containing compounds lacking direct metal-carbon bonds but which contain organic ligands. Metal β-diketonates, alkoxides, dialkylamides, and metal p ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phosphorus
Phosphorus is a chemical element; it has Chemical symbol, symbol P and atomic number 15. All elemental forms of phosphorus are highly Reactivity (chemistry), reactive and are therefore never found in nature. They can nevertheless be prepared artificially, the two most common allotropes being white phosphorus and red phosphorus. With as its only stable isotope, phosphorus has an occurrence in Earth's crust of about 0.1%, generally as phosphate rock. A member of the pnictogen family, phosphorus readily forms a wide variety of organic compound, organic and inorganic compound, inorganic compounds, with as its main oxidation states +5, +3 and −3. The isolation of white phosphorus in 1669 by Hennig Brand marked the scientific community's first discovery since Antiquity of an element. The name phosphorus is a reference to the Phosphorus (morning star), god of the Morning star in Greek mythology, inspired by the faint glow of white phosphorus when exposed to oxygen. This property is ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phenylmagnesium Bromide
Phenylmagnesium bromide, with the simplified formula , is a magnesium-containing organometallic compound. It forms colorless crystals. It is commercially available as a solution in diethyl ether or tetrahydrofuran (THF). Phenylmagnesium bromide is a Grignard reagent. It is often used as a synthetic equivalent for the phenyl "Ph−" synthon. Preparation Phenylmagnesium bromide is commercially available as solutions of diethyl ether or THF. Laboratory preparation involves treating bromobenzene with magnesium metal, usually in the form of turnings. A small amount of iodine may be used to activate the magnesium to initiate the reaction. Coordinating solvents such as ether or THF, are required to solvate (complex) the magnesium(II) center. The solvent must be aprotic since alcohols and water contain an acidic proton and thus react with phenylmagnesium bromide to give benzene. Carbonyl-containing solvents, such as acetone and ethyl acetate, are also incompatible with the reagent. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |