|
Timothy P. Lodge
Timothy P. Lodge (born 11 April 1954) is an American polymer scientist. Lodge is a Regents Professor (2013–present), an Institute of Technology Distinguished Professor (2004–present), and a Distinguished McKnight University Professor (2001–present) in the Department of Chemistry and the Department of Chemical Engineering and Materials Science at the University of Minnesota, twin cities, Twin Cities, Minneapolis-Saint Paul, Minnesota where he has been a faculty member since 1982. He served as the Editor-in-Chief of the American Chemical Society journal Macromolecules (journal), ''Macromolecules'' for 17 years (2001–2017) and as the founding editor of ''ACS Macro Letters'' (2011–2018). He is recognized for his research in polymer science, especially fundamental explorations of polymer chain dynamics in miscibility, miscible blends, block polymers, multicompartment micelles and polymers in ionic liquids. According to ''Web of Science'', he has produced 331 published works ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Manchester
Manchester () is a city in Greater Manchester, England. It had a population of 552,000 in 2021. It is bordered by the Cheshire Plain to the south, the Pennines to the north and east, and the neighbouring city of City of Salford, Salford to the west. The two cities and the surrounding towns form one of the United Kingdom's most populous conurbations, the Greater Manchester Built-up Area, which has a population of 2.87 million. The history of Manchester began with the civilian settlement associated with the Roman Britain, Roman fort (''castra'') of ''Mamucium'' or ''Mancunium'', established in about AD 79 on a sandstone bluff near the confluence of the rivers River Medlock, Medlock and River Irwell, Irwell. Historic counties of England, Historically part of Lancashire, areas of Cheshire south of the River Mersey were incorporated into Manchester in the 20th century, including Wythenshawe in 1931. Throughout the Middle Ages Manchester remained a manorialism, manorial Township ( ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Arthur S
Arthur is a common male given name of Brythonic origin. Its popularity derives from it being the name of the legendary hero King Arthur. The etymology is disputed. It may derive from the Celtic ''Artos'' meaning “Bear”. Another theory, more widely believed, is that the name is derived from the Roman clan '' Artorius'' who lived in Roman Britain for centuries. A common spelling variant used in many Slavic, Romance, and Germanic languages is Artur. In Spanish and Italian it is Arturo. Etymology The earliest datable attestation of the name Arthur is in the early 9th century Welsh-Latin text ''Historia Brittonum'', where it refers to a circa 5th to 6th-century Briton general who fought against the invading Saxons, and who later gave rise to the famous King Arthur of medieval legend and literature. A possible earlier mention of the same man is to be found in the epic Welsh poem '' Y Gododdin'' by Aneirin, which some scholars assign to the late 6th century, though this is still ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Fireproofing
Fireproofing is rendering something ( structures, materials, etc.) resistant to fire, or incombustible; or material for use in making anything fire-proof. It is a passive fire protection measure. "Fireproof" or "fireproofing" can be used as a noun, verb or adjective; it may be hyphenated ("fire-proof"). Applying a certification listed fireproofing system to certain structures allows them to have a fire-resistance rating. The term "fireproofing" may be used in conjunction with standards, as reflected in common North American construction specifications. An item classed as fireproof is resistant in specified circumstances, and may burn or be rendered inoperable by fire exceeding the intensity or duration that it is designed to withstand. Markets * Commercial construction * Residential construction * Industrial construction * Marine ( ships) * Offshore construction * Aerodynamics * Tunnel concrete walls and ceilings or linings * Under- and above-ground mining operations Ap ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Vapor Pressure
Vapor pressure (or vapour pressure in English-speaking countries other than the US; see spelling differences) or equilibrium vapor pressure is defined as the pressure exerted by a vapor in thermodynamic equilibrium with its condensed phases (solid or liquid) at a given temperature in a closed system. The equilibrium vapor pressure is an indication of a liquid's evaporation rate. It relates to the tendency of particles to escape from the liquid (or a solid). A substance with a high vapor pressure at normal temperatures is often referred to as '' volatile''. The pressure exhibited by vapor present above a liquid surface is known as vapor pressure. As the temperature of a liquid increases, the kinetic energy of its molecules also increases. As the kinetic energy of the molecules increases, the number of molecules transitioning into a vapor also increases, thereby increasing the vapor pressure. The vapor pressure of any substance increases non-linearly with temperature accord ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
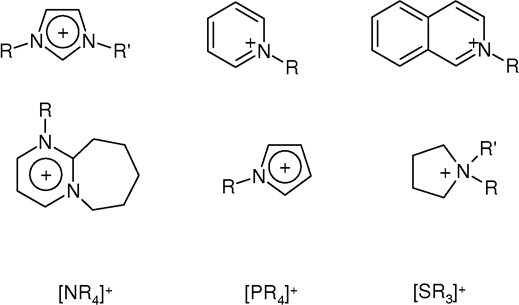
Ionic Liquids
An ionic liquid (IL) is a salt in the liquid state. In some contexts, the term has been restricted to salts whose melting point is below a specific temperature, such as . While ordinary liquids such as water and gasoline are predominantly made of electrically neutral molecules, ionic liquids are largely made of ions. These substances are variously called liquid electrolytes, ionic melts, ionic fluids, fused salts, liquid salts, or ionic glasses. Ionic liquids have many potential applications. They are powerful solvents and can be used as electrolytes. Salts that are liquid at near-ambient temperature are important for electric battery applications, and have been considered as sealants due to their very low vapor pressure. Any salt that melts without decomposing or vaporizing usually yields an ionic liquid. Sodium chloride (NaCl), for example, melts at into a liquid that consists largely of sodium cations () and chloride anions (). Conversely, when an ionic liquid is coole ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chromophore
A chromophore is the part of a molecule responsible for its color. The color that is seen by our eyes is the one not absorbed by the reflecting object within a certain wavelength spectrum of visible light. The chromophore is a region in the molecule where the energy difference between two separate molecular orbitals falls within the range of the visible spectrum. Visible light that hits the chromophore can thus be absorbed by exciting an electron from its ground state into an excited state. In biological molecules that serve to capture or detect light energy, the chromophore is the moiety that causes a conformational change in the molecule when hit by light. Conjugated pi-bond system chromophores Just like how two adjacent p-orbitals in a molecule will form a pi-bond, three or more adjacent p-orbitals in a molecule can form a conjugated pi-system. In a conjugated pi-system, electrons are able to capture certain photons as the electrons resonate along a certain distanc ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrophile
A hydrophile is a molecule or other molecular entity that is attracted to water molecules and tends to be dissolved by water.Liddell, H.G. & Scott, R. (1940). ''A Greek-English Lexicon'' Oxford: Clarendon Press. In contrast, hydrophobes are not attracted to water and may seem to be repelled by it. Hygroscopics ''are'' attracted to water, but are not dissolved by water. Molecules A hydrophilic molecule or portion of a molecule is one whose interactions with water and other polar substances are more thermodynamically favorable than their interactions with oil or other hydrophobic solvents. They are typically charge-polarized and capable of hydrogen bonding. This makes these molecules soluble not only in water but also in other polar solvents. Hydrophilic molecules (and portions of molecules) can be contrasted with hydrophobic molecules (and portions of molecules). In some cases, both hydrophilic and hydrophobic properties occur in a single molecule. An example of these amphip ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Epitaxy
Epitaxy refers to a type of crystal growth or material deposition in which new crystalline layers are formed with one or more well-defined orientations with respect to the crystalline seed layer. The deposited crystalline film is called an epitaxial film or epitaxial layer. The relative orientation(s) of the epitaxial layer to the seed layer is defined in terms of the orientation of the crystal lattice of each material. For most epitaxial growths, the new layer is usually crystalline and each crystallographic domain of the overlayer must have a well-defined orientation relative to the substrate crystal structure. Epitaxy can involve single-crystal structures, although grain-to-grain epitaxy has been observed in granular films. For most technological applications, single domain epitaxy, which is the growth of an overlayer crystal with one well-defined orientation with respect to the substrate crystal, is preferred. Epitaxy can also play an important role while growing superlatti ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Polymorphism (materials Science)
In materials science, polymorphism describes the existence of a solid material in more than one form or crystal structure. Polymorphism is a form of isomerism. Any crystalline material can exhibit the phenomenon. Allotropy refers to polymorphism for chemical elements. Polymorphism is of practical relevance to pharmaceuticals, agrochemicals, pigments, dyestuffs, foods, and explosives. According to IUPAC, a polymorphic transition is "A reversible transition of a solid crystalline phase at a certain temperature and pressure (the inversion point) to another phase of the same chemical composition with a different crystal structure." According to McCrone, polymorphs are "different in crystal structure but identical in the liquid or vapor states." Materials with two polymorphs are called dimorphic, with three polymorphs, trimorphic, etc. Examples Many compounds exhibit polymorphism. It has been claimed that "every compound has different polymorphic forms, and that, in general, the nu ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chemical Kinetics
Chemical kinetics, also known as reaction kinetics, is the branch of physical chemistry that is concerned with understanding the rates of chemical reactions. It is to be contrasted with chemical thermodynamics, which deals with the direction in which a reaction occurs but in itself tells nothing about its rate. Chemical kinetics includes investigations of how experimental conditions influence the speed of a chemical reaction and yield information about the reaction's mechanism and transition states, as well as the construction of mathematical models that also can describe the characteristics of a chemical reaction. History In 1864, Peter Waage and Cato Guldberg pioneered the development of chemical kinetics by formulating the law of mass action, which states that the speed of a chemical reaction is proportional to the quantity of the reacting substances.C.M. Guldberg and P. Waage,"Studies Concerning Affinity" ''Forhandlinger i Videnskabs-Selskabet i Christiania'' (1864), 3 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Thermodynamics
Thermodynamics is a branch of physics that deals with heat, work, and temperature, and their relation to energy, entropy, and the physical properties of matter and radiation. The behavior of these quantities is governed by the four laws of thermodynamics which convey a quantitative description using measurable macroscopic physical quantities, but may be explained in terms of microscopic constituents by statistical mechanics. Thermodynamics applies to a wide variety of topics in science and engineering, especially physical chemistry, biochemistry, chemical engineering and mechanical engineering, but also in other complex fields such as meteorology. Historically, thermodynamics developed out of a desire to increase the efficiency of early steam engines, particularly through the work of French physicist Sadi Carnot (1824) who believed that engine efficiency was the key that could help France win the Napoleonic Wars. Scots-Irish physicist Lord Kelvin was the first to ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Copolymers
In polymer chemistry, a copolymer is a polymer derived from more than one species of monomer. The polymerization of monomers into copolymers is called copolymerization. Copolymers obtained from the copolymerization of two monomer species are sometimes called ''bipolymers''. Those obtained from three and four monomers are called ''terpolymers'' and ''quaterpolymers'', respectively. Copolymers can be characterized by a variety of techniques such as NMR spectroscopy and size-exclusion chromatography to determine the molecular size, weight, properties, and composition of the material. Commercial copolymers include acrylonitrile butadiene styrene (ABS), styrene/butadiene co-polymer (SBR), nitrile rubber, styrene-acrylonitrile, styrene-isoprene-styrene (SIS) and ethylene-vinyl acetate, all of which are formed by chain-growth polymerization. Another production mechanism is step-growth polymerization, which is used to produce the nylon-12/6/66 copolymer of nylon 12, nylon 6 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |


.jpeg/1200px-Fall_Leaves_(199582361).jpeg)


