|
Nitrogen Halides
Nitrogen is a chemical element; it has symbol N and atomic number 7. Nitrogen is a nonmetal and the lightest member of group 15 of the periodic table, often called the pnictogens. It is a common element in the universe, estimated at seventh in total abundance in the Milky Way and the Solar System. At standard temperature and pressure, two atoms of the element bond to form N2, a colourless and odourless diatomic gas. N2 forms about 78% of Earth's atmosphere, making it the most abundant chemical species in air. Because of the volatility of nitrogen compounds, nitrogen is relatively rare in the solid parts of the Earth. It was first discovered and isolated by Scottish physician Daniel Rutherford in 1772 and independently by Carl Wilhelm Scheele and Henry Cavendish at about the same time. The name was suggested by French chemist Jean-Antoine-Claude Chaptal in 1790 when it was found that nitrogen was present in nitric acid and nitrates. Antoine Lavoisier suggested instead the n ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chemical Element
A chemical element is a chemical substance whose atoms all have the same number of protons. The number of protons is called the atomic number of that element. For example, oxygen has an atomic number of 8: each oxygen atom has 8 protons in its atomic nucleus, nucleus. Atoms of the same element can have different numbers of neutrons in their nuclei, known as isotopes of the element. Two or more atoms can combine to form molecules. Some elements form Homonuclear molecule, molecules of atoms of said element only: e.g. atoms of hydrogen (H) form Diatomic molecule, diatomic molecules (H). Chemical compounds are substances made of atoms of different elements; they can have molecular or non-molecular structure. Mixtures are materials containing different chemical substances; that means (in case of molecular substances) that they contain different types of molecules. Atoms of one element can be transformed into atoms of a different element in nuclear reactions, which change an atom's at ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Henry Cavendish
Henry Cavendish ( ; 10 October 1731 – 24 February 1810) was an English experimental and theoretical chemist and physicist. He is noted for his discovery of hydrogen, which he termed "inflammable air". He described the density of inflammable air, which formed water on combustion, in a 1766 paper, ''On Factitious Airs''. Antoine Lavoisier later reproduced Cavendish's experiment and gave the element its name. A shy man, Cavendish was distinguished for great accuracy and precision in his researches into the composition of atmospheric air, the properties of different gases, the synthesis of water, the law governing electrical attraction and repulsion, a mechanical theory of heat, and calculations of the density (and hence the mass) of the Earth. His experiment to measure the density of the Earth (which, in turn, allows the gravitational constant to be calculated) has come to be known as the Cavendish experiment. Early life Henry Cavendish was born on 10 October 1731 in Nice, whe ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Propellant
A propellant (or propellent) is a mass that is expelled or expanded in such a way as to create a thrust or another motive force in accordance with Newton's third law of motion, and "propel" a vehicle, projectile, or fluid payload. In vehicles, the engine that expels the propellant is called a reaction engine. Although technically a propellant is the reaction mass used to create thrust, the term "propellant" is often used to describe a substance which contains both the reaction mass and the fuel that holds the energy used to accelerate the reaction mass. For example, the term "propellant" is often used in chemical rocket design to describe a combined fuel/propellant, although the propellants should not be confused with the fuel that is used by an engine to produce the energy that expels the propellant. Even though the byproducts of substances used as fuel are also often used as a reaction mass to create the thrust, such as with a chemical rocket engine, propellant and fuel a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ammonia
Ammonia is an inorganic chemical compound of nitrogen and hydrogen with the chemical formula, formula . A Binary compounds of hydrogen, stable binary hydride and the simplest pnictogen hydride, ammonia is a colourless gas with a distinctive pungent smell. It is widely used in fertilizers, refrigerants, explosives, cleaning agents, and is a precursor for numeous chemicals. Biologically, it is a common nitrogenous waste, and it contributes significantly to the nutritional needs of terrestrial organisms by serving as a precursor to fertilisers. Around 70% of ammonia produced industrially is used to make fertilisers in various forms and composition, such as urea and diammonium phosphate. Ammonia in pure form is also applied directly into the soil. Ammonia, either directly or indirectly, is also a building block for the synthesis of many chemicals. In many countries, it is classified as an List of extremely hazardous substances, extremely hazardous substance. Ammonia is toxic, cau ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cryogenic
In physics, cryogenics is the production and behaviour of materials at very low temperatures. The 13th International Institute of Refrigeration's (IIR) International Congress of Refrigeration (held in Washington, DC in 1971) endorsed a universal definition of "cryogenics" and "cryogenic" by accepting a threshold of to distinguish these terms from conventional refrigeration. This is a logical dividing line, since the normal boiling points of the so-called permanent gases (such as helium, hydrogen, neon, nitrogen, oxygen, and normal air) lie below 120 K, while the Freon refrigerants, hydrocarbons, and other common refrigerants have boiling points above 120 K. Discovery of superconducting materials with critical temperatures significantly above the boiling point of nitrogen has provided new interest in reliable, low-cost methods of producing high-temperature cryogenic refrigeration. The term "high temperature cryogenic" describes temperatures ranging from above the ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Liquid Nitrogen
Liquid nitrogen (LN2) is nitrogen in a liquid state at cryogenics, low temperature. Liquid nitrogen has a boiling point of about . It is produced industrially by fractional distillation of liquid air. It is a colorless, mobile liquid whose viscosity is about one-tenth that of acetone (i.e. roughly one-thirtieth that of water at room temperature). Liquid nitrogen is widely used as a coolant. Physical properties The diatomic character of the N2 molecule is retained after liquefaction. The weak van der Waals interaction between the N2 molecules results in little interatomic attraction. This is the cause of nitrogen's unusually low boiling point. The temperature of liquid nitrogen can readily be reduced to its freezing point by placing it in a vacuum chamber pumped by a vacuum pump. Liquid nitrogen's efficiency as a coolant is limited by the fact that it boils immediately on contact with a warmer object, enveloping the object in an insulating layer of nitrogen gas bubbles. Thi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pressure Swing Adsorption
Pressure swing adsorption (PSA) is a technique used to separate some gas species from a mixture of gases (typically air) under pressure according to the species' molecular characteristics and affinity for an adsorbent material. It operates at near-ambient temperature and significantly differs from the cryogenic distillation commonly used to separate gases. Selective adsorbent materials (e.g., zeolites, (aka molecular sieves), activated carbon, etc.) are used as trapping material, preferentially adsorbing the target gas species at high pressure. The process then swings to low pressure to desorb the adsorbed gas. Process The pressure swing adsorption (PSA) process is based on the phenomenon that under high pressure, gases tend to be trapped onto solid surfaces, ''i.e.'' to be "adsorbed". The higher the pressure, the more gas is adsorbed. When the pressure is dropped, the gas is released, or desorbed. PSA can be used to separate gases in a mixture because different gases are adsor ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Azo Compound
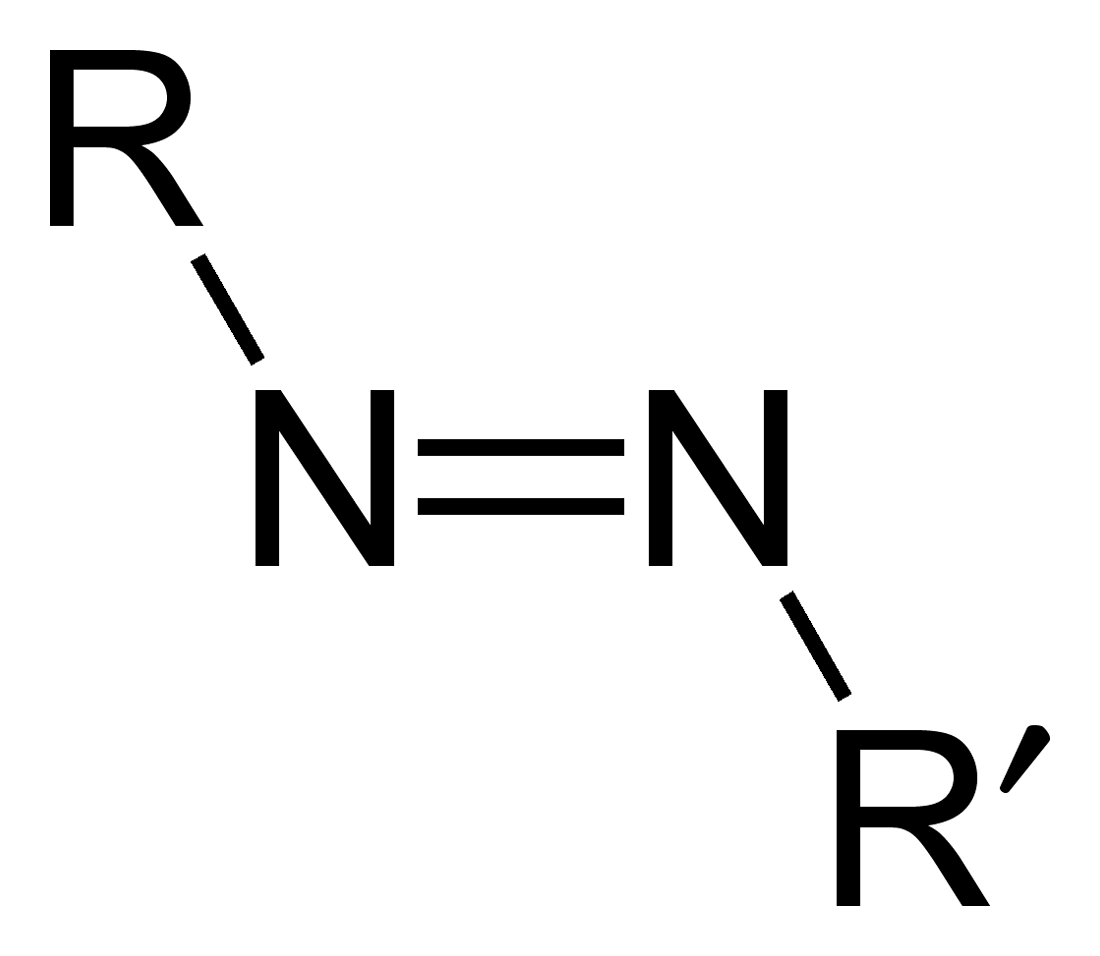
Azo compounds are organic compounds bearing the functional group diazenyl (, in which R and R′ can be either aryl or alkyl groups). IUPAC defines azo compounds as: "Derivatives of diazene (diimide), , wherein both hydrogens are substituted by hydrocarbyl groups, e.g. azobenzene or diphenyldiazene.", where Ph stands for phenyl group. The more stable derivatives contain two aryl groups. The group is called an ''azo group'' (, ). Many textile and leather articles are dyed with azo dyes and pigments. Aryl azo compounds urinary tract infections">Phenazopyridine, an aryl azo compound, is used to treat urinary tract infections">150px Aryl azo compounds are usually stable, crystalline species. Azobenzene is the prototypical aromatic azo compound. It exists mainly as the Cis-trans isomerism, ''trans'' isomer, but upon illumination, converts to the Cis-trans isomerism, ''cis'' isomer. Aromatic azo compounds can be synthesized by azo coupling, which entails an electrophilic ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Azide
In chemistry, azide (, ) is a linear, polyatomic anion with the formula and structure . It is the conjugate base of hydrazoic acid . Organic azides are organic compounds with the formula , containing the azide functional group. The dominant application of azides is as a propellant in air bags. Preparation Sodium azide is made industrially by the reaction of nitrous oxide, with sodium amide in liquid ammonia as solvent: : Many inorganic azides can be prepared directly or indirectly from sodium azide. For example, lead azide, used in detonators, may be prepared from the metathesis reaction between lead nitrate and sodium azide. An alternative route is direct reaction of the metal with silver azide dissolved in liquid ammonia. Some azides are produced by treating the carbonate salts with hydrazoic acid. Bonding Azide is isoelectronic with carbon dioxide , cyanate , nitrous oxide , nitronium ion , molecular beryllium fluoride and cyanogen fluoride FCN. Per ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrazine
Hydrazine is an inorganic compound with the chemical formula . It is a simple pnictogen hydride, and is a colourless flammable liquid with an ammonia-like odour. Hydrazine is highly hazardous unless handled in solution as, for example, hydrazine hydrate (). Hydrazine is mainly used as a foaming agent in preparing Polymeric foam, polymer foams, but applications also include its uses as a precursor (chemistry), precursor to pharmaceuticals and agrochemicals, as well as a long-term storable propellant for in-outer space, space spacecraft propulsion. Additionally, hydrazine is used in various rocket propellant, rocket fuels and to prepare the gas precursors used in airbags. Hydrazine is used within both nuclear and conventional electrical power plant steam cycles as an oxygen scavenger to control concentrations of dissolved oxygen in an effort to reduce corrosion. , approximately 120,000 tons of hydrazine hydrate (corresponding to a 64% solution of hydrazine in water by weight) we ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Asphyxiant Gas
An asphyxiant gas, also known as a simple asphyxiant, is a nontoxic or minimally toxic gas which reduces or displaces the normal oxygen concentration in breathing air. Breathing of oxygen-depleted air can lead to death by asphyxiation (suffocation). Because asphyxiant gases are relatively inert and odorless, their presence in high concentration may not be noticed, except in the case of carbon dioxide ( hypercapnia). Toxic gases, by contrast, cause death by other mechanisms, such as competing with oxygen on the cellular level (e.g. carbon monoxide) or directly damaging the respiratory system (e.g. phosgene). Far smaller quantities of these are deadly. Notable examples of asphyxiant gases are methane, nitrogen, argon, helium, butane and propane. Along with trace gases such as carbon dioxide and ozone, these compose 79% of Earth's atmosphere. Asphyxia hazard Asphyxiant gases in the breathing air are normally not hazardous. Only where elevated concentrations of asphyxiant g ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Antoine Lavoisier
Antoine-Laurent de Lavoisier ( ; ; 26 August 17438 May 1794), When reduced without charcoal, it gave off an air which supported respiration and combustion in an enhanced way. He concluded that this was just a pure form of common air and that it was the air itself "undivided, without alteration, without decomposition" which combined with metals on calcination. After returning from Paris, Priestley took up once again his investigation of the air from mercury calx. His results now showed that this air was not just an especially pure form of common air but was "five or six times better than common air, for the purpose of respiration, inflammation, and ... every other use of common air". He called the air dephlogisticated air, as he thought it was common air deprived of its phlogiston. Since it was therefore in a state to absorb a much greater quantity of phlogiston given off by burning bodies and respiring animals, the greatly enhanced combustion of substances and the greater ease ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |