|
Copper(I) Bromide
Copper(I) bromide is the chemical compound with the formula CuBr. This white diamagnetic solid adopts a polymeric structure akin to that for zinc sulfide. The compound is widely used in the synthesis of organic compounds and as a lasing medium in copper bromide lasers. Preparation, basic properties, structure The compound is white, although samples are often colored due to the presence of copper(II) impurities. The copper(I) ion also oxidizes easily in air. It is commonly prepared by the reduction of cupric salts with sulfite in the presence of bromide.This report gives a procedure for generating CuBr: For example, the reduction of copper(II) bromide with sulfite yields copper(I) bromide and hydrogen bromide: :2 CuBr2 + H2O + → 2 CuBr + + 2 HBr CuBr is insoluble in most solvents due to its polymeric structure, which features four-coordinated, tetrahedral Cu centers interconnected by bromide ligands (ZnS structure). Upon treatment with Lewis bases, CuBr con ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrochloric Acid
Hydrochloric acid, also known as muriatic acid or spirits of salt, is an aqueous solution of hydrogen chloride (HCl). It is a colorless solution with a distinctive pungency, pungent smell. It is classified as a acid strength, strong acid. It is a component of the gastric acid in the digestive systems of most animal species, including humans. Hydrochloric acid is an important laboratory reagent and industrial chemical. Etymology Because it was produced from halite, rock salt according to the methods of Johann Rudolph Glauber, hydrochloric acid was historically called by European alchemists ''spirits of salt'' or ''acidum salis'' (salt acid). Both names are still used, especially in other languages, such as , , , , , , , , , , (''ensan''), zh, 盐酸 (''yánsuān''), and (''yeomsan''). Gaseous HCl was called ''marine acid air''. The name ''muriatic acid'' has the same origin (''muriatic'' means "pertaining to brine or salt", hence ''muriate'' means hydrochloride), and this ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Copper(II) Bromide
Copper(II) bromide ( Cu Br2) is a chemical compound that forms an unstable tetrahydrate CuBr2·4H2O. It is used in photographic processing as an intensifier and as a brominating agent in organic synthesis. It is also used in the copper vapor laser, a class of laser where the medium is copper bromide vapour formed in-situ from hydrogen bromide reacting with the copper discharge tube. Producing yellow or green light, it is used in dermatological applications. Synthesis Copper(II) bromide can be obtained by combining copper oxide and hydrobromic acid: :CuO + 2HBr → CuBr2 + H2O. The tetrahydrate can be produced by recrystallization of solutions of copper(II) bromide at 0 °C. If heated above 18 °C, it releases water to produce the anhydrous form. Purification Copper(II) bromide is purified by crystallization twice from water, filtration to remove any CuBr and concentration under vacuum. This product is dehydrated using phosphorus pentoxide. Molecular and cry ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Catalyst
Catalysis () is the increase in rate of a chemical reaction due to an added substance known as a catalyst (). Catalysts are not consumed by the reaction and remain unchanged after it. If the reaction is rapid and the catalyst recycles quickly, very small amounts of catalyst often suffice; mixing, surface area, and temperature are important factors in reaction rate. Catalysts generally react with one or more reactants to form intermediates that subsequently give the final reaction product, in the process of regenerating the catalyst. The rate increase occurs because the catalyst allows the reaction to occur by an alternative mechanism which may be much faster than the noncatalyzed mechanism. However the noncatalyzed mechanism does remain possible, so that the total rate (catalyzed plus noncatalyzed) can only increase in the presence of the catalyst and never decrease. Catalysis may be classified as either homogeneous, whose components are dispersed in the same phase (usual ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Organocopper
Organocopper chemistry is the study of the physical properties, reactions, and synthesis of organocopper compounds, which are organometallic compounds containing a carbon to copper chemical bond. They are reagents in organic chemistry. The first organocopper compound, the explosive copper(I) acetylide (), was synthesized by Rudolf Christian Böttger in 1859 by passing acetylene gas through a solution of copper(I) chloride: : Structure and bonding Organocopper compounds are diverse in structure and reactivity, but almost all are based on copper with an oxidation state of +1, sometimes denoted Cu(I) or . With 10 electrons in its valence shell, the bonding behavior of Cu(I) is similar to Ni(0), but owing to its higher oxidation state, it engages in less Pi backbonding, pi-backbonding. Organic derivatives of copper's higher oxidation states of +2 and +3 are sometimes encountered as reaction intermediates, but rarely isolated or even observed. Organocopper compounds form comp ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Aryl
In organic chemistry, an aryl is any functional group or substituent derived from an aromatic ring, usually an aromatic hydrocarbon, such as phenyl and naphthyl. "Aryl" is used for the sake of abbreviation or generalization, and "Ar" is used as a placeholder for the aryl group in chemical structure diagrams, analogous to “R” used for any organic substituent. “Ar” is not to be confused with the elemental symbol for argon. A simple aryl group is phenyl (), a group derived from benzene. Examples of other aryl groups consist of: * The tolyl group () which is derived from toluene (methylbenzene) * The xylyl group (), which is derived from xylene (dimethylbenzene) * The naphthyl group (), which is derived from naphthalene Arylation is the process in which an aryl group is attached to a substituent. It is typically achieved by cross-coupling reactions. Nomenclature The simplest aryl group is phenyl, which is made up of a benzene ring with one of its hydrogen atom ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sandmeyer Reaction
The Sandmeyer reaction is a chemical reaction used to synthesize aryl halides from aryl diazonium salts using copper salts as reagents or catalysts. It is an example of a radical-nucleophilic aromatic substitution. The Sandmeyer reaction provides a method through which one can perform unique transformations on benzene, such as halogenation, cyanation, trifluoromethylation, and hydroxylation. The reaction was discovered in 1884 by Swiss chemist Traugott Sandmeyer, when he attempted to synthesize phenylacetylene from benzenediazonium chloride and copper(I) acetylide. Instead, the main product he isolated was chlorobenzene. In modern times, the Sandmeyer reaction refers to any method for substitution of an aromatic amino group via preparation of its diazonium salt followed by its displacement with a nucleophile in the presence of catalytic copper(I) salts. : The most commonly employed Sandmeyer reactions are the chlorination, bromination, cyanation, and hydroxylation reaction ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pyrotechnic
Pyrotechnics is the science and craft of creating fireworks, but also includes safety matches, oxygen candles, explosive bolts (and other fasteners), parts of automotive airbags, as well as gas-pressure blasting in mining, quarrying, and demolition. This trade relies upon self-contained and self-sustained exothermic chemical reactions to make heat, light, gas, smoke and/or sound. The name comes from the Greek words ''pyr'' (πυρ; 'fire') and ''technikós'' (τεχνικός; 'artistic'). Improper use of pyrotechnics could lead to pyrotechnic accidents. People responsible for the safe storage, handling, and functioning of pyrotechnic devices are known as pyrotechnicians. Proximate pyrotechnics Explosions, flashes, smoke, flames, fireworks and other pyrotechnic-driven effects used in the entertainment industry are referred to as proximate pyrotechnics. Proximate refers to the pyrotechnic device's location relative to an audience. In the majority of jurisdictions, special tr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Copper(I) Chloride
Copper(I) chloride, commonly called cuprous chloride, is the lower chloride of copper, with the formula CuCl. The substance is a white solid sparingly soluble in water, but very soluble in concentrated hydrochloric acid. Impure samples appear green due to the presence of copper(II) chloride (CuCl2). History Copper(I) chloride was first prepared by Robert Boyle and designated rosin of copper in the mid-seventeenth century from mercury(II) chloride ("Venetian sublimate") and copper metal: :HgCl2 + 2 Cu → 2 CuCl + Hg In 1799, Joseph Proust first differentiated two different chlorides of copper. He prepared CuCl (which he called white muriate of copper) by heating CuCl2 at red heat in the absence of air, causing it to lose half of its combined chlorine followed by removing residual CuCl2 by washing with water. An acidic solution of CuCl was formerly used to analyze carbon monoxide content in gases, for example in Hempel's gas apparatus where the CuCl absorbs the carbon ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Colorfulness
Colorfulness, chroma and saturation are attributes of perceived color relating to chromatic intensity. As defined formally by the International Commission on Illumination (CIE) they respectively describe three different aspects of chromatic intensity, but the terms are often used loosely and interchangeably in contexts where these aspects are not clearly distinguished. The precise meanings of the terms vary by what other functions they are dependent on. * Colorfulness is the "attribute of a visual perception according to which the perceived color of an area appears to be more or less chromatic (Any color that is absent of white, grey, or black)"., page 87. The colorfulness evoked by an object depends not only on its spectral reflectance but also on the strength of the illumination, and increases with the latter unless the brightness is very high (Hunt effect (color), Hunt effect). * Chroma is the "colorfulness of an area judged as a proportion of the brightness of a similarl ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
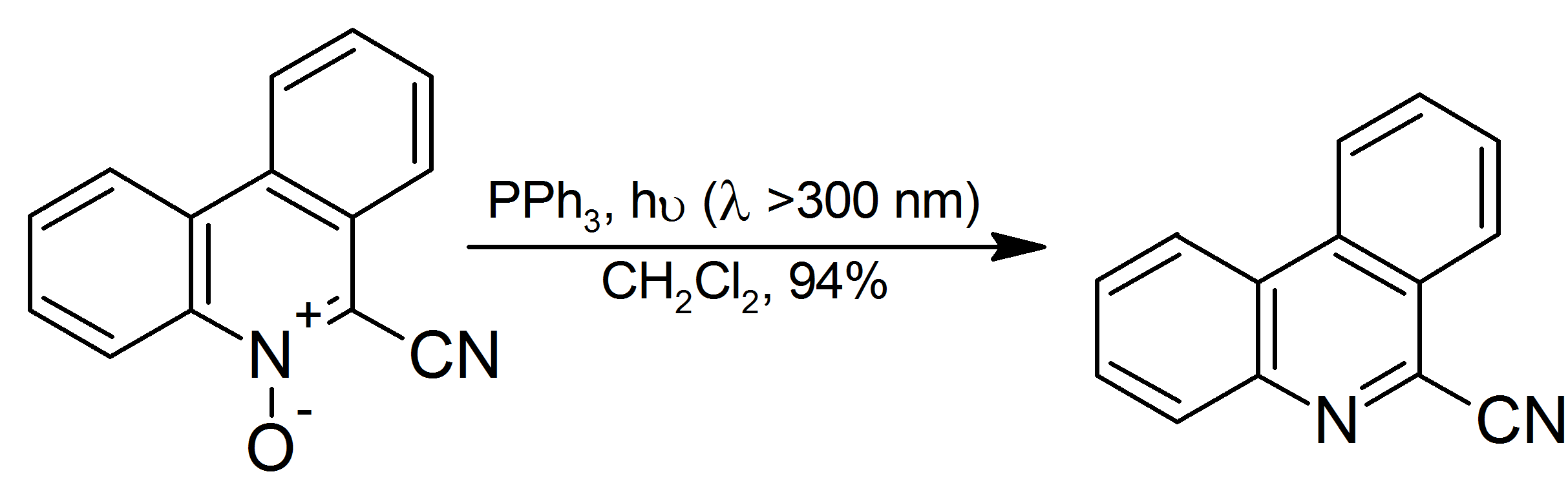
Triphenylphosphine
Triphenylphosphine (IUPAC name: triphenylphosphane) is a common organophosphorus compound with the formula P(C6H5)3 and often abbreviated to P Ph3 or Ph3P. It is versatile compound that is widely used as a reagent in organic synthesis and as a ligand for transition metal complexes, including ones that serve as catalysts in organometallic chemistry. PPh3 exists as relatively air stable, colorless crystals at room temperature. It dissolves in non-polar organic solvents such as benzene and diethyl ether. Preparation and structure Triphenylphosphine can be prepared in the laboratory by treatment of phosphorus trichloride with phenylmagnesium bromide or phenyllithium. The industrial synthesis involves the reaction between phosphorus trichloride, chlorobenzene, and sodium: :PCl3 + 3 PhCl + 6 Na → PPh3 + 6 NaCl Triphenylphosphine crystallizes in triclinic and monoclinic modification. In both cases, the molecule adopts a pyramidal structure with propeller-like arrangement of the thre ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Coordination Complex
A coordination complex is a chemical compound consisting of a central atom or ion, which is usually metallic and is called the ''coordination centre'', and a surrounding array of chemical bond, bound molecules or ions, that are in turn known as ''ligands'' or complexing agents. Many metal-containing chemical compound, compounds, especially those that include transition metals (elements like titanium that belong to the periodic table's d-block), are coordination complexes. Nomenclature and terminology Coordination complexes are so pervasive that their structures and reactions are described in many ways, sometimes confusingly. The atom within a ligand that is bonded to the central metal atom or ion is called the donor atom. In a typical complex, a metal ion is bonded to several donor atoms, which can be the same or different. A Ligand#Polydentate and polyhapto ligand motifs and nomenclature, polydentate (multiple bonded) ligand is a molecule or ion that bonds to the central atom ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dimethyl Sulfide
Dimethyl sulfide (DMS) or methylthiomethane is an organosulfur compound with the formula . It is the simplest thioether and has a characteristic disagreeable odor. It is a flammable liquid that boils at . It is a component of the smell produced from cooking of certain vegetables (notably maize, cabbage, and beetroot) and seafoods. It is also an indication of bacterial contamination in malt production and brewing. It is a breakdown product of dimethylsulfoniopropionate (DMSP), and is also produced by the bacterial metabolism of methanethiol. Occurrence and production DMS originates primarily from DMSP, a major secondary metabolite in some marine algae. DMS is the most abundant biological sulfur compound emitted to the atmosphere. Emission occurs over the oceans by phytoplankton. DMS is also produced naturally by bacterial transformation of dimethyl sulfoxide (DMSO) waste that is disposed of into sewers, where it can cause environmental odor problems. DMS is oxidized in ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |