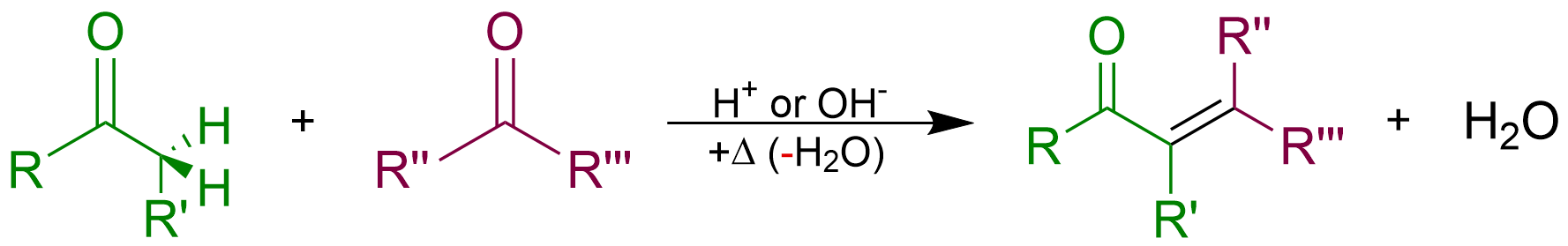|
Chichibabin Synthesis
The Chichibabin pyridine synthesis () is a method for synthesizing pyridine rings. The reaction involves the condensation reaction of aldehydes, ketones, α,β-unsaturated carbonyl compounds, or any combination of the above, with ammonia. It was reported by Aleksei Chichibabin in 1924. Methyl-substituted pyridines, which show widespread uses among multiple fields of applied chemistry, are prepared by this methodology. Representative syntheses : The syntheses are presently conduced commercially in the presence of oxide catalysts such as modified alumina (Al2O3) or silica (SiO2). The reactants are passed over the catalyst at 350–500 °C. 2-Methylpyridine- and 4-methylpyridine are produced as a mixture from acetaldehyde and ammonia. 3-Methylpyridine and pyridine are produced from acrolein and ammonia. Acrolein and propionaldehyde react with ammonia affords mainly 3-methylpyridine. 5-Ethyl-2-methylpyridine is produced from paraldehyde and ammonia. Mechanism and optimiz ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Aleksei Chichibabin
Alekséy Yevgényevich Chichibábin (; – 15 August 1945) was a Soviet Union, Soviet Russian organic chemist. His name is also written ''Alexei Yevgenievich Chichibabin'' and ''Alexei Euguenievich Tchitchibabine''. Life Chichibábin was born at Kuzemin on March 17, 1871. He studied at the University of Moscow from 1888 until 1892, and received his PhD from the University of Saint Petersburg. In 1896, Chichibáin married Vera Vladimirovna as his wife. He became a professor at the Imperial College of Technology in Moscow in 1909, and remained there until 1929. After losing his daughter Natacha, a chemist, to an industrial oleum accident (explosion) that he deemed preventable, Chichibábin moved to Paris where he remained despite threat of and eventual stripping of his Soviet citizenship and his position in the Academy of Sciences (1936, Academy standing restored posthumously, 1990).David Lewis, 2012, Early Russian Organic Chemists and Their Legacy (Vol. 4, SpringerBriefs in Mole ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Aldol Condensation
An aldol condensation is a condensation reaction in organic chemistry in which two carbonyl moieties (of aldehydes or ketones) react to form a β-hydroxyaldehyde or β-hydroxyketone (an aldol reaction), and this is then followed by dehydration to give a conjugated enone. The overall reaction equation is as follows (where the Rs can be H) Aldol condensations are important in organic synthesis and biochemistry as ways to form carbon–carbon bonds. In its usual form, it involves the nucleophilic addition of a ketone enolate to an aldehyde to form a β-hydroxy ketone, or aldol (aldehyde + alcohol), a structural unit found in many naturally occurring molecules and pharmaceuticals. The term ''aldol condensation'' is also commonly used, especially in biochemistry, to refer to just the first (addition) stage of the process—the aldol reaction itself—as catalyzed by aldolases. However, the first step is formally an addition reaction rather than a condensation reaction bec ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Heterocycle Forming Reactions
A heterocyclic compound or ring structure is a cyclic compound that has atoms of at least two different elements as members of its ring(s). Heterocyclic organic chemistry is the branch of organic chemistry dealing with the synthesis, properties, and applications of organic heterocycles. Examples of heterocyclic compounds include all of the nucleic acids, the majority of drugs, most biomass (cellulose and related materials), and many natural and synthetic dyes. More than half of known compounds are heterocycles. 59% of US FDA-approved drugs contain nitrogen heterocycles. Classification The study of organic heterocyclic chemistry focuses especially on organic unsaturated derivatives, and the preponderance of work and applications involves unstrained organic 5- and 6-membered rings. Included are pyridine, thiophene, pyrrole, and furan. Another large class of organic heterocycles refers to those fused to benzene rings. For example, the fused benzene derivatives of pyridin ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pyridine Forming Reactions
Pyridine is a basic heterocyclic organic compound with the chemical formula . It is structurally related to benzene, with one methine group replaced by a nitrogen atom . It is a highly flammable, weakly alkaline, water-miscible liquid with a distinctive, unpleasant fish-like smell. Pyridine is colorless, but older or impure samples can appear yellow, due to the formation of extended, unsaturated polymeric chains, which show significant electrical conductivity. The pyridine ring occurs in many important compounds, including agrochemicals, pharmaceuticals, and vitamins. Historically, pyridine was produced from coal tar. As of 2016, it is synthesized on the scale of about 20,000 tons per year worldwide. Properties Physical properties Pyridine is diamagnetic. Its critical parameters are: pressure 5.63 MPa, temperature 619 K and volume 248 cm3/mol. In the temperature range 340–426 °C its vapor pressure ''p'' can be described with the Antoine equation :\ ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ciamician–Dennstedt Rearrangement
Pyridine is a basic (chemistry), basic heterocyclic compound, heterocyclic organic compound with the chemical formula . It is structurally related to benzene, with one methine group replaced by a nitrogen atom . It is a highly flammable, weakly alkaline, water-miscible liquid with a distinctive, unpleasant fish-like smell. Pyridine is colorless, but older or impure samples can appear yellow, due to the formation of extended, unsaturated Polymer, polymeric chains, which show significant electrical conductivity. The pyridine ring occurs in many important compounds, including agrochemicals, pharmaceuticals, and vitamins. Historically, pyridine was produced from coal tar. As of 2016, it is synthesized on the scale of about 20,000 tons per year worldwide. Properties Physical properties Pyridine is diamagnetism, diamagnetic. Its critical point (thermodynamics), critical parameters are: pressure 5.63 MPa, temperature 619 K and volume 248 cm3/mol. In the temperatur ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hantzsch Pyridine Synthesis
The Hantzsch pyridine synthesis or Hantzsch dihydropyridine synthesis is a multi-component organic reaction between an aldehyde such as formaldehyde Formaldehyde ( , ) (systematic name methanal) is an organic compound with the chemical formula and structure , more precisely . The compound is a pungent, colourless gas that polymerises spontaneously into paraformaldehyde. It is stored as ..., 2 equivalents of a Keto ester, β-keto ester such as ethyl acetoacetate and a nitrogen donor such as ammonium acetate or ammonia. The initial reaction product is a dihydropyridine which can be organic oxidation, oxidized in a subsequent step to a pyridine. The driving force for this second reaction step is aromatization. This reaction was reported in 1881 by Arthur Rudolf Hantzsch. A 1,4-dihydropyridine dicarboxylate is also called a 1,4-DHP compound or a Hantzsch ester. These compounds are an important class of calcium channel blockers and as such commercialized in for instance nife ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chichibabin Reaction
The Chichibabin reaction (pronounced ' (chē')-chē-bā-bēn) is a method for producing 2-aminopyridine derivatives by the reaction of pyridine with sodium amide. It was reported by Aleksei Chichibabin in 1914. The following is the overall form of the general reaction: The direct amination of pyridine with sodium amide can take place in liquid ammonia or an aprotic solvent such as xylene is commonly used. Following the Addition-elimination reaction, addition elimination mechanism first a nucleophilic addition, nucleophilic NH2− is added while a hydride (H−) is leaving. The reaction formally is a nucleophilic substitution of hydrogen SNH. Ciganek describes an example of an intramolecular Chichibabin reaction in which a nitrile group on a fused ring is the source of nitrogen in amination. Mechanism It is widely accepted that the Chichibabin reaction mechanism is an addition-elimination reaction that proceeds through an σ-adduct (Meisenheimer complex, Meisenheimer adduct) ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Alkyne Trimerization
An alkyne trimerisation is a +2+2nbsp; cycloaddition reaction in which three alkyne units () react to form a benzene ring. The reaction requires a metal catalyst. The process is of historic interest as well as being applicable to organic synthesis. Being a cycloaddition reaction, it has high atom economy. Many variations have been developed, including cyclisation of mixtures of alkynes and alkenes as well as alkynes and nitriles. Mechanism and stereochemistry Trimerisation of acetylene to benzene is highly exergonic, proceeding with a free energy change of 142 kcal/mol at room temperature. Kinetic barriers however prevent the reaction from proceeding smoothly. The breakthrough came in 1948, when Walter Reppe and W. J. Schweckendiek reported their wartime results showing that nickel compounds are effective catalysts: : Since this discovery, many other cyclotrimerisations have been reported. Mechanism In terms of mechanism, the reactions begin with the formation of metal- ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Organocobalt Compound
Organocobalt chemistry is the chemistry of organometallic compounds containing a carbon to cobalt chemical bond. Organocobalt compounds are involved in several organic reactions and the important biomolecule vitamin B12, vitamin B12 has a cobalt-carbon bond. Many organocobalt compounds exhibit useful catalytic properties, the preeminent example being dicobalt octacarbonyl. Alkyl complexes Most fundamental are the cobalt complexes with only alkyl ligands. Examples include Co(4-norbornyl)4 and its cation. Alkylcobalt is represented by vitamin B12, vitamin B12 and related enzymes. In methylcobalamin the ligand is a methyl group, which is electrophilic. in vitamin B12, the alkyl ligand is an adenosyl group. Related to vitamin B12 are cobalt porphyrins, dimethylglyoxime, dimethylglyoximates, and related complexes of Schiff base ligands. These synthetic compounds also form alkyl derivatives that undergo diverse reactions reminiscent of the biological processes. The weak cobalt(III) ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Acrylonitrile
Acrylonitrile is an organic compound with the formula and the structure . It is a colorless, volatile liquid. It has a pungent odor of garlic or onions. Its molecular structure consists of a vinyl group () linked to a nitrile (). It is an important monomer for the manufacture of useful plastics such as polyacrylonitrile. It is reactive and toxic at low doses. Acrylonitrile is one of the components of ABS plastic (acrylonitrile butadiene styrene). Structure and basic properties Acrylonitrile is an organic compound with the formula and the structure . It is a colorless, volatile liquid although commercial samples can be yellow due to impurities. It has a pungent odor of garlic or onions. Its molecular structure consists of a vinyl group () linked to a nitrile (). It is an important monomer for the manufacture of useful plastics such as polyacrylonitrile. It is reactive and toxic at low doses. Production Acrylonitrile was first synthesized by the French chemist Charle ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Zeolite
Zeolites are a group of several microporous, crystalline aluminosilicate minerals commonly used as commercial adsorbents and catalysts. They mainly consist of silicon, aluminium, oxygen, and have the general formula ・y where is either a metal ion or H+. The term was originally coined in 1756 by Swedish mineralogist Axel Fredrik Cronstedt, who observed that rapidly heating a material, believed to have been stilbite, produced large amounts of steam from water that had been adsorbed by the material. Based on this, he called the material ''zeolite'', from the Greek , meaning "to boil" and , meaning "stone". Zeolites occur naturally, but are also produced industrially on a large scale. , 253 unique zeolite frameworks have been identified, and over 40 naturally occurring zeolite frameworks are known. Every new zeolite structure that is obtained is examined by the International Zeolite Association Structure Commission (IZA-SC) and receives a three-letter designation. Character ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |