|
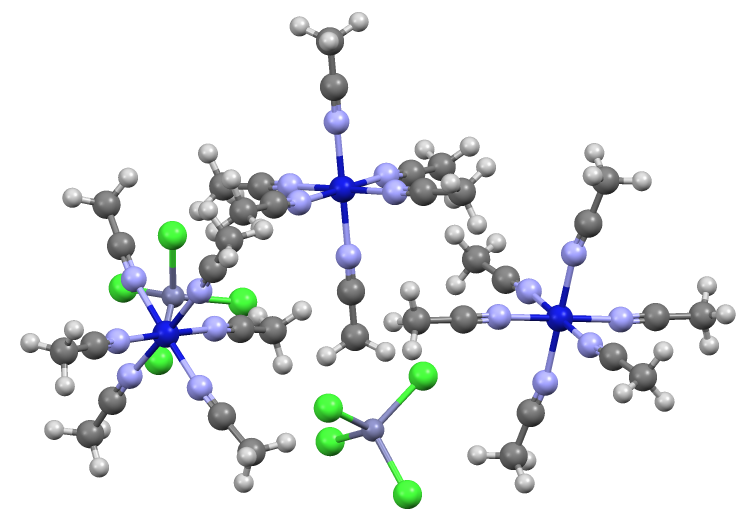
Ammonium Tetrachlorozincate
Zinc ammonium chloride is the inorganic compound with the formula (NH4)2ZnCl4. It is the ammonium salt of tetrachlorozincate. It used as a flux in the process of hot-dip galvanizing. Uses Steel to be galvanized passes through an acidic cleaning process to remove iron oxide "mill scale". After this process, the surface of the steel is very active and oxide layers begin forming immediately upon exposure to the atmosphere. Zinc ammonium chloride flux in aqueous solution is applied to the steel to reduce any oxides that are formed and/or inhibit them from forming altogether. This allows the molten zinc in the proceeding galvanizing step to maximally adhere to and alloy with the surface of the steel. Precautions Zinc ammonium chloride is a Class 9 hazardous material (Miscellaneous) according to the U.S. DOT. References {{Ammonium salts ammonium chloride Tetrachlorozincates zinc Zinc is a chemical element; it has symbol Zn and atomic number 30. It is a slightly brittle m ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Inorganic Compound
An inorganic compound is typically a chemical compound that lacks carbon–hydrogen bondsthat is, a compound that is not an organic compound. The study of inorganic compounds is a subfield of chemistry known as ''inorganic chemistry''. Inorganic compounds comprise most of the Earth's crust, although the compositions of the deep Mantle (geology), mantle remain active areas of investigation. All allotropes (structurally different pure forms of an element) and some simple carbon compounds are often considered inorganic. Examples include the allotropes of carbon (graphite, diamond, buckminsterfullerene, graphene, etc.), carbon monoxide , carbon dioxide , carbides, and salt (chemistry), salts of inorganic anions such as carbonates, cyanides, cyanates, thiocyanates, isothiocyanates, etc. Many of these are normal parts of mostly organic systems, including organisms; describing a chemical as inorganic does not necessarily mean that it cannot occur within life, living things. History ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ammonium
Ammonium is a modified form of ammonia that has an extra hydrogen atom. It is a positively charged (cationic) polyatomic ion, molecular ion with the chemical formula or . It is formed by the protonation, addition of a proton (a hydrogen nucleus) to ammonia (). Ammonium is also a general name for positively charged (protonated) substituted amines and quaternary ammonium cations (), where one or more hydrogen atoms are replaced by Organic compound, organic or other groups (indicated by R). Not only is ammonium a source of nitrogen and a key metabolite for many living organisms, but it is an integral part of the global nitrogen cycle. As such, human impact in recent years could have an effect on the biological communities that depend on it. Acid–base properties The ammonium ion is generated when ammonia, a weak base, reacts with Brønsted–Lowry acid–base theory, Brønsted acids (proton donors): : The ammonium ion is mildly acidic, reacting with Brønsted bases to return ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tetrachlorozincate
Tetrachlorozincate is an anion with the formula . It is a counterion that is often used in conjunction with strong electrophiles. Being dianionic, tetrachlorozincate is not classified as a weakly coordinating anion. On the other hand, being dianionic, tetrachlorozincate facilitates the crystallization of many salts. It has a tetrahedral molecular geometry. A simple example is (ammonium tetrachlorozincate). Zincates are anionic zinc complexes. Related to the preparation of Lucas' reagent, tetrachlorozincates are often generated by combining hydrochloric acid and zinc chloride. A related anion is , in which again Zn(II) adopts a tetrahedral geometry. References {{Reflist Tetrachlorozincates, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Flux (metallurgy)
In metallurgy, a flux is a chemical reducing agent, flowing agent, or purifying agent. Fluxes may have more than one function at a time. They are used in both extractive metallurgy and metal joining. Some of the earliest known fluxes were sodium carbonate, potash, charcoal, coke, borax, lime, lead sulfide and certain minerals containing phosphorus. Iron ore was also used as a flux in the smelting of copper. These agents served various functions, the simplest being a reducing agent, which prevented oxides from forming on the surface of the molten metal, while others absorbed impurities into slag, which could be scraped off molten metal. Fluxes are also used in foundries for removing impurities from molten nonferrous metals such as aluminium, or for adding desirable trace elements such as titanium. As reducing agents, fluxes facilitate soldering, brazing, and welding by removing oxidation from the metals to be joined. In some applications molten flux also serves as ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hot-dip Galvanizing
Hot-dip galvanization is a form of galvanization. It is the process of coating iron and steel with zinc, which alloys with the surface of the base metal when immersing the metal in a bath of molten zinc at a temperature of around . When exposed to the atmosphere, the pure zinc (Zn) reacts with oxygen (Oxygen, O2) to form zinc oxide (ZnO), which further reacts with carbon dioxide (CO2, CO2) to form zinc carbonate (Smithsonite, ZnCO3), a usually dull grey, fairly strong material that protects the steel underneath from further corrosion in many circumstances. Galvanized steel is widely used in applications where corrosion resistance is needed without the cost of stainless steel, and is considered superior in terms of cost and life-cycle. It can be identified by the crystallization patterning on the surface (often called a "spangle"). Galvanized steel can be welded; however, one must exercise caution around the resulting toxic zinc fumes. Galvanized fumes are released when the ga ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Steel
Steel is an alloy of iron and carbon that demonstrates improved mechanical properties compared to the pure form of iron. Due to steel's high Young's modulus, elastic modulus, Yield (engineering), yield strength, Fracture, fracture strength and low raw material cost, steel is one of the most commonly manufactured materials in the world. Steel is used in structures (as concrete Rebar, reinforcing rods), in Bridge, bridges, infrastructure, Tool, tools, Ship, ships, Train, trains, Car, cars, Bicycle, bicycles, Machine, machines, Home appliance, electrical appliances, furniture, and Weapon, weapons. Iron is always the main element in steel, but other elements are used to produce various grades of steel demonstrating altered material, mechanical, and microstructural properties. Stainless steels, for example, typically contain 18% chromium and exhibit improved corrosion and Redox, oxidation resistance versus its carbon steel counterpart. Under atmospheric pressures, steels generally ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pickling (metal)
Pickling is a metal surface treatment used to remove impurities, such as stains, inorganic contaminants, and rust or scale from ferrous metals, copper, precious metals and aluminium alloys. A solution called ''pickle liquor'', which usually contains acid, is used to remove the surface impurities. It is commonly used to descale or clean steel in various steelmaking processes. Process Metal surfaces can contain impurities that may affect usage of the product or further processing like plating with metal or painting. Various chemical solutions are usually used to clean these impurities. Strong acids, such as hydrochloric acid and sulfuric acid are common, but different applications use various other acids. Also alkaline solutions can be used for cleaning metal surfaces. Solutions usually also contain additives such as wetting agents and corrosion inhibitors. Pickling is sometimes called acid cleaning if descaling is not needed. Many hot working processes and other processes that o ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Aqueous Solution
An aqueous solution is a solution in which the solvent is water. It is mostly shown in chemical equations by appending (aq) to the relevant chemical formula. For example, a solution of table salt, also known as sodium chloride (NaCl), in water would be represented as . The word ''aqueous'' (which comes from ''aqua'') means pertaining to, related to, similar to, or dissolved in, water. As water is an excellent solvent and is also naturally abundant, it is a ubiquitous solvent in chemistry. Since water is frequently used as the solvent in experiments, the word solution refers to an aqueous solution, unless the solvent is specified. A ''non-aqueous solution'' is a solution in which the solvent is a liquid, but is not water. Characteristics Substances that are ''hydrophobic'' ('water-fearing') do not dissolve well in water, whereas those that are '' hydrophilic'' ('water-friendly') do. An example of a hydrophilic substance is sodium chloride. In an aqueous solution the hydrogen ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
HAZMAT Class 9 Miscellaneous
The miscellaneous hazardous materials category encompasses all hazardous materials that do not fit one of the definitions listed in Class 1 through Class 8. Divisions The miscellaneous hazardous material is a material that presents a hazard during transportation but which does not meet the definition of any other hazard class. This class includes: #Any material which has an anesthetic, noxious or other similar property which could cause extreme annoyance or discomfort to a flight crew member so as to prevent the correct performance of assigned duties; or #Any material that meets the definition in 49 CFR 171.8 for an elevated temperature material, a hazardous substance, a hazardous waste, or a marine pollutant. A new sub-class, class 9A, has been in effect since January 1, 2017. This is limited to the labeling of the transport of lithium batteries Lithium battery may refer to: * Lithium metal battery, a non-rechargeable battery with lithium as an anode ** Lithium–air batter ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
United States Department Of Transportation
The United States Department of Transportation (USDOT or DOT) is one of the executive departments of the U.S. federal government. It is headed by the secretary of transportation, who reports directly to the president of the United States and is a member of the president's Cabinet. The department's fiscal year 2022–2026 strategic plan states that its mission is "to deliver the world's leading transportation system, serving the American people and economy through the safe, efficient, sustainable, and equitable movement of people and goods." History In 1965, Najeeb Halaby, was granted authority over aviation and railroads through the commerce clause of the Constitution, the Federal Highway Administration and Federal Transit Administration primarily provided funding for state and local projects, without significant influence over road construction and operation. Halaby emphasized the need for improved coordination and expressed frustration at the lack of an overall plan. " ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Zinc Compounds
Zinc compounds are chemical compounds containing the element zinc which is a member of the group 12 element, group 12 of the periodic table. The oxidation state of zinc in most compounds is the group oxidation state of +2. Zinc may be classified as a post-transition main group element with zinc(II). Zinc compounds are noteworthy for their nondescript appearance and behavior: they are generally colorless (unlike compounds of other elements with oxidation number +2, which are colored), do not readily engage in redox reactions, and generally adopt symmetrical structures. General characteristics In its compounds, Zn2+ ions have an electronic configuration [Ar] 3d10. As such, Zn2+ tends to have a symmetrical coordination geometry in both its complexes and compounds. In both ZnO and ZnS, (zincblende) zinc is bound tetrahedrally bound to four ligands (oxide and sulfide, respectively). Many coordination complex, complexes, such as ZnCl42−, are tetrahedral. Tetrahedrally coordinated zin ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |