Tetrachlorozincate on:
[Wikipedia]
[Google]
[Amazon]
Tetrachlorozincate is an 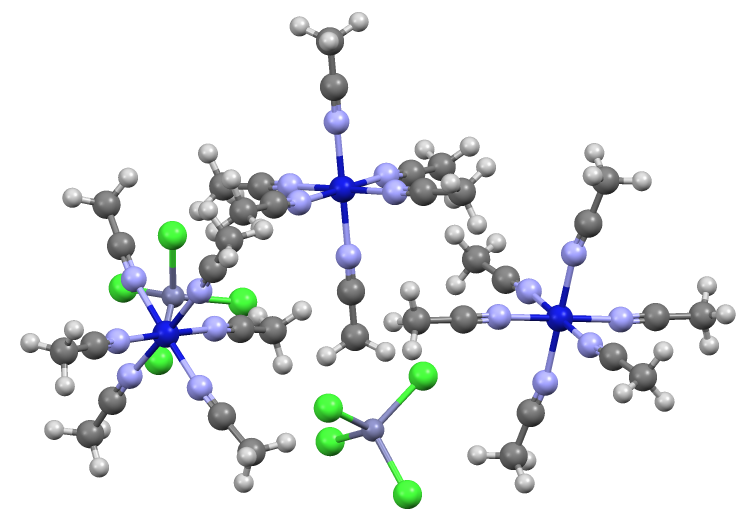

anion
An ion () is an atom or molecule with a net electrical charge. The charge of an electron is considered to be negative by convention and this charge is equal and opposite to the charge of a proton, which is considered to be positive by conven ...
with the formula . It is a counterion
160px, cation-exchange_resin.html" ;"title="Polystyrene sulfonate, a cation-exchange resin">Polystyrene sulfonate, a cation-exchange resin, is typically supplied with as the counterion.
In chemistry, a counterion (sometimes written as "counter ...
that is often used in conjunction with strong electrophiles. Being dianionic, tetrachlorozincate is not classified as a weakly coordinating anion
Anions that interact weakly with cations are termed non-coordinating anions, although a more accurate term is weakly coordinating anion. Non-coordinating anions are useful in studying the reactivity of electrophilic cations. They are commonly found ...
. On the other hand, being dianionic, tetrachlorozincate facilitates the crystallization of many salts. It has a tetrahedral molecular geometry
In a tetrahedral molecular geometry, a central atom is located at the center with four substituents that are located at the corners of a tetrahedron. The bond angles are arccos(−) = 109.4712206...° ≈ 109.5° when all four substituents are ...
. A simple example is (ammonium tetrachlorozincate
Zinc ammonium chloride is the inorganic compound with the formula (NH4)2ZnCl4. It is the ammonium salt of tetrachlorozincate. It used as a flux in the process of hot-dip galvanizing.
Uses
Steel to be galvanized passes through an acidic cleani ...
). Zincates are anionic zinc complexes.
Related to the preparation of Lucas' reagent
"Lucas' reagent" is a solution of anhydrous zinc chloride in concentrated hydrochloric acid. This solution is used to classify alcohols of low molecular weight. The reaction is a substitution in which the chloride replaces a hydroxyl group. A ...
, tetrachlorozincates are often generated by combining hydrochloric acid
Hydrochloric acid, also known as muriatic acid or spirits of salt, is an aqueous solution of hydrogen chloride (HCl). It is a colorless solution with a distinctive pungency, pungent smell. It is classified as a acid strength, strong acid. It is ...
and zinc chloride
Zinc chloride is an Inorganic chemistry, inorganic chemical compound with the chemical formula, formula ZnCl2·''n''H2O, with ''n'' ranging from 0 to 4.5, forming water of hydration, hydrates. Zinc chloride, anhydrous and its hydrates, are colo ...
.
A related anion is , in which again Zn(II) adopts a tetrahedral geometry.


References
{{Reflist