|
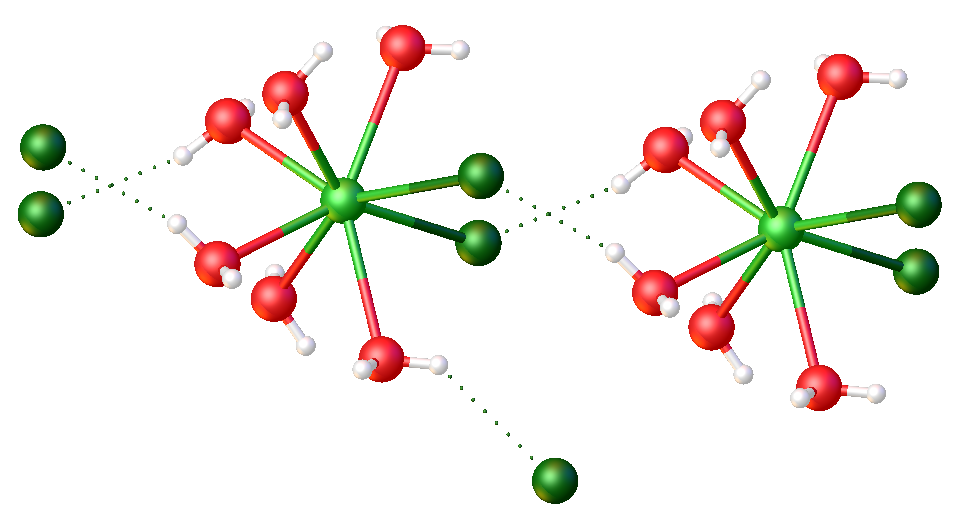
Ammonium Chloride Route
Lanthanide trichlorides are a family of inorganic compound with the formula Ln Cl3, where Ln stands for a lanthanide metal. The trichlorides are standard reagents in applied and academic chemistry of the lanthanides. They exist as anhydrous solids and as hydrates. Properties The anhydrous solids have melting points range from ca. 582 (Tb) - 925 °C (Lu). They are generally pale colored, often white. As coordination polymers, they only dissolve in donor solvents, including water. Preparation The lanthanide oxides and carbonates dissolve in hydrochloric acid to give chloride salt of the hydrated cations: :M2O3 + 6HCl + n H2O → 2 n(H2O)nl3 Industrial routes Anhydrous trichlorides are produced commercially by carbothermic reaction of the oxide: :M2O3 + 3Cl2 + 3C → 2MCl3 + 3CO Ammonium chloride route The ammonium chloride route refers to a general procedure to produce anhydrous lanthanide chlorides. The method has the advantages of being general for the 14 la ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Inorganic Compound
An inorganic compound is typically a chemical compound that lacks carbon–hydrogen bondsthat is, a compound that is not an organic compound. The study of inorganic compounds is a subfield of chemistry known as ''inorganic chemistry''. Inorganic compounds comprise most of the Earth's crust, although the compositions of the deep Mantle (geology), mantle remain active areas of investigation. All allotropes (structurally different pure forms of an element) and some simple carbon compounds are often considered inorganic. Examples include the allotropes of carbon (graphite, diamond, buckminsterfullerene, graphene, etc.), carbon monoxide , carbon dioxide , carbides, and salt (chemistry), salts of inorganic anions such as carbonates, cyanides, cyanates, thiocyanates, isothiocyanates, etc. Many of these are normal parts of mostly organic systems, including organisms; describing a chemical as inorganic does not necessarily mean that it cannot occur within life, living things. History ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Thulium Trichloride
Thulium(III) chloride or thulium trichloride is as an inorganic salt composed of thulium and chlorine with the formula TmCl3. It forms yellow crystals. Thulium(III) chloride has the YCl3 ( AlCl3) layer structure with octahedral thulium ions. It has been used as a starting material for some exotic nanostructures prepared for NIR photocatalysis. Preparation Thulium(III) chloride can be obtained by reacting thulium(III) oxide or thulium(III) carbonate and ammonium chloride: : The hexahydrate of thulium(III) chloride can be obtained by adding thulium(III) oxide to concentrated hydrochloric acid. : Thulium(III) chloride can also be obtained by directly reacting thulium and chlorine:WebelementsThulium/ref> : Properties Thulium(III) chloride is a light yellow powder. Its hexahydrate is a light green hygroscopic solid. Both are soluble in water. Thulium(III) chloride has a monoclinic crystal structure with the space group In mathematics, physics and chemistry, a space group ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ytterbium Oxide
Ytterbium(III) oxide is the chemical compound with the formula Yb2O3. It is one of the more commonly encountered compounds of ytterbium. It occurs naturally in trace amounts in the mineral gadolinite. It was first isolated from this in 1878 by Jean Charles Galissard de Marignac. Preparation Ytterbium(III) oxide can be obtained by directly reacting ytterbium with oxygen: : It can also be obtained by the thermal decomposition of ytterbium carbonate or ytterbium oxalate at temperatures around 700 °C: : : Properties Chemical Ytterbium(III) oxide is a white powder. It reacts with carbon tetrachloride or hot hydrochloric acid to form ytterbium(III) chloride: : : Physical Like the other trivalent oxides of the heavier lanthanides, ytterbium(III) oxide has the "rare-earth C-type sesquioxide" structure which is related to the fluorite structure with one quarter of the anions removed, leading to ytterbium atoms in two different six coordinate (non-octahedral) env ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Lutetium Oxide
Lutetium(III) oxide, a white solid, is a cubic compound of lutetium sometimes used in the preparation of specialty glasses. It is also called lutecia. It is a lanthanide oxide, also known as a rare earth.Lutetium Oxide. 1997-2007. Metall Rare Earth Limited. http://www.metall.com.cn/luo.htm History In 1879, Swiss chemist Jean Charles Galissard de Marignac (1817–1894) claimed to have discovered ytterbium, but he had found a mixture of elements. In 1907, French chemist Georges Urbain (1872–1938) reported that ytterbium was a mixture of two new elements and was not a single element. Two other chemists, Carl Auer von Welsbach (1858–1929) and Charles James (1880–1926) also extracted lutetium(III) oxide around the same time. All three scientists successfully separated Marignac's ytterbia into oxides of two elements which were eventually named ytterbium and lutetium Lutetium is a chemical element; it has symbol Lu and atomic number 71. It is a silvery white metal, which resists c ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Thulium(III) Oxide
Thulium(III) oxide is a pale green crystalline compound, with the formula Tm2 O3. It was first isolated in 1879, from an impure sample of erbia, by Swedish chemist A chemist (from Greek ''chēm(ía)'' alchemy; replacing ''chymist'' from Medieval Latin ''alchemist'') is a graduated scientist trained in the study of chemistry, or an officially enrolled student in the field. Chemists study the composition of ... Per Teodor Cleve, who named it ''thulia''. Synthesis Thulium(III) oxide has been made in the laboratory using various methods. One method involves burning thulium metal or its various salts in air. Thulium(III) oxide can be made using a hydrothermal method where thulium(III) acetate is mixed with an ammonia solution, which causes thulium(III) oxide to precipitate as a white solid. Properties Thulium(III) oxide (Tm₂O₃) is a pale green, thermally stable powder with a high melting point of 2,341 °C and a density of 8.6 g/cm³, typically forming a cubic crysta ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Erbium(III) Oxide
Erbium(III) oxide is the inorganic compound with the formula . It is a pink paramagnetic solid. It finds uses in various optical materials. Structure Erbium(III) oxide has a cubic structure resembling the bixbyite motif. The Er3+ centers are octahedral. Reactions Erbium oxide is produced by burning erbium metal. Erbium oxide is insoluble in water but soluble in mineral acids. Er2O3 does not readily absorb moisture and carbon dioxide from the atmosphere. It can react with acids to form the corresponding erbium(III) salts. For example, with hydrochloric acid, the oxide follows the following idealized reaction leading to erbium chloride: : In practice, such simple acid-base reactions are accompanied by hydration: : Properties One interesting property of erbium oxides is their ability to up convert photons. Photon upconversion takes place when infrared or visible radiation, low energy light, is converted to ultraviolet or violet radiation higher energy light via multiple tran ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Holmium Oxide
Holmium(III) oxide, or holmium oxide is a chemical compound of the rare-earth element holmium and oxygen with the formula Ho2O3. Together with dysprosium(III) oxide (Dy2O3), holmium oxide is one of the most powerfully paramagnetic substances known. The oxide, also called holmia, occurs as a component of the related erbium oxide mineral called erbia. Typically, the oxides of the trivalent lanthanides coexist in nature, and separation of these components requires specialized methods. Holmium oxide is used in making specialty colored glasses. Glass containing holmium oxide and holmium oxide solutions have a series of sharp optical absorption peaks in the visible spectral range. They are therefore traditionally used as a convenient calibration standard for optical spectrophotometers. Properties Appearance Holmium oxide has some fairly dramatic color changes depending on the lighting conditions. In daylight, it is a tannish yellow color. Under trichromatic light, it is a fiery orange ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dysprosium Oxide
Dysprosium oxide (Dy2O3) is a sesquioxide compound of the rare earth metal dysprosium. It is a pastel yellowish-greenish, slightly hygroscopic powder having specialized uses in ceramics, glass, phosphors, lasers, dysprosium metal halide lamps, and as a Faraday rotator A Faraday rotator is a polarization rotator based on the Faraday effect, a magneto-optic effect involving transmission of light through a material when a longitudinal static magnetic field is present. The state of polarization (such as the axis o .... It can react with acids to produce the corresponding dysprosium(III) salts: :Dy2O3 + 6 HCl → 2 DyCl3 + 3 H2O References Dysprosium compounds Sesquioxides {{Inorganic-compound-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Yttrium
Yttrium is a chemical element; it has Symbol (chemistry), symbol Y and atomic number 39. It is a silvery-metallic transition metal chemically similar to the lanthanides and has often been classified as a "rare-earth element". Yttrium is almost always found in combination with lanthanide elements in rare-earth minerals and is never found in nature as a free element. 89Y is the only stable isotope and the only isotope found in the Crust (geology), Earth's crust. The most important present-day use of yttrium is as a component of phosphors, especially those used in LEDs. Historically, it was once widely used in the red phosphors in television set cathode ray tube displays. Yttrium is also used in the production of electrodes, electrolytes, electronic filters, lasers, superconductors, various medical applications, and Trace element, tracing various materials to enhance their properties. Yttrium has no known Biology, biological role. Exposure to yttrium compounds can cause Respiratory ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Scandium
Scandium is a chemical element; it has Symbol (chemistry), symbol Sc and atomic number 21. It is a silvery-white metallic d-block, d-block element. Historically, it has been classified as a rare-earth element, together with yttrium and the lanthanides. It was discovered in 1879 by spectral analysis of the minerals euxenite and gadolinite from Scandinavia. Scandium is present in most of the deposits of rare-earth and uranium compounds, but it is extracted from these ores in only a few mines worldwide. Because of the low availability and difficulties in the preparation of metallic scandium, which was first done in 1937, applications for scandium were not developed until the 1970s, when the positive effects of scandium on aluminium alloys were discovered. Its use in such alloys remains its only major application. The global trade of scandium oxide is 15–20 tonnes per year. The properties of scandium compounds are intermediate between those of aluminium and yttrium. A diagonal rela ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Anhydrous
A substance is anhydrous if it contains no water. Many processes in chemistry can be impeded by the presence of water; therefore, it is important that water-free reagents and techniques are used. In practice, however, it is very difficult to achieve perfect dryness; anhydrous compounds gradually absorb water from the atmosphere so they must be stored carefully. Solids Many salts and solids can be dried using heat, or under vacuum. Desiccators can also be used to store reagents in dry conditions. Common desiccants include phosphorus pentoxide and silica gel. Chemists may also require dry glassware for sensitive reactions. This can be achieved by drying glassware in an oven, by flame, or under vacuum. Dry solids can be produced by freeze-drying, which is also known as lyophilization. Liquids or solvents In many cases, the presence of water can prevent a reaction from happening, or cause undesirable products to form. To prevent this, anhydrous solvents must be used when perform ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Carbothermic Reaction
Carbothermic reactions involve the reduction of substances, often metal oxides (O2-), using carbon (C) as the reducing agent. The reduction is usually conducted in the electric arc furnace or reverberatory furnace, depending on the metal ore. These chemical reactions are usually conducted at temperatures of several hundred degrees Celsius. Such processes are applied for production of the elemental forms of many elements. The ability of metals to participate in carbothermic reactions can be predicted from Ellingham diagrams. Carbothermal reactions produce carbon monoxide (CO) and sometimes carbon dioxide (CO2). The facility of these conversions is attributable to the entropy of reaction: two solids, the metal oxide (and flux) and carbon, are converted to a new solid (metal) and a gas (CO), the latter having high entropy. Applications A prominent example is that of iron ore smelting. Many reactions are involved, but the simplified equation is usually shown as: : 2 + 3 C � ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |