|
Equivalent Weight
In chemistry, equivalent weight (also known as gram equivalent) is the mass of one equivalent, that is the mass of a given substance which will combine with or displace a fixed quantity of another substance. The equivalent weight of an element is the mass which combines with or displaces 1.008 gram of hydrogen or 8.0 grams of oxygen or 35.5 grams of chlorine. These values correspond to the atomic weight divided by the usual valence; for oxygen as example that is 16.0 g / 2 = 8.0 g. For acid–base reactions, the equivalent weight of an acid or base is the mass which supplies or reacts with one mole of hydrogen cations (). For redox reactions, the equivalent weight of each reactant supplies or reacts with one mole of electrons (e−) in a redox reaction. Equivalent weight has the units of mass, unlike atomic weight, which is now used as a synonym for relative atomic mass and is dimensionless. Equivalent weights were originally determined by experiment, but (insofar as ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chemistry
Chemistry is the scientific study of the properties and behavior of matter. It is a natural science that covers the elements that make up matter to the compounds made of atoms, molecules and ions: their composition, structure, properties, behavior and the changes they undergo during a reaction with other substances. Chemistry also addresses the nature of chemical bonds in chemical compounds. In the scope of its subject, chemistry occupies an intermediate position between physics and biology. It is sometimes called the central science because it provides a foundation for understanding both basic and applied scientific disciplines at a fundamental level. For example, chemistry explains aspects of plant growth ( botany), the formation of igneous rocks ( geology), how atmospheric ozone is formed and how environmental pollutants are degraded ( ecology), the properties of the soil on the moon ( cosmochemistry), how medications work ( pharmacology), and how to collec ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Acid
In computer science, ACID ( atomicity, consistency, isolation, durability) is a set of properties of database transactions intended to guarantee data validity despite errors, power failures, and other mishaps. In the context of databases, a sequence of database operations that satisfies the ACID properties (which can be perceived as a single logical operation on the data) is called a ''transaction''. For example, a transfer of funds from one bank account to another, even involving multiple changes such as debiting one account and crediting another, is a single transaction. In 1983, Andreas Reuter and Theo Härder coined the acronym ''ACID'', building on earlier work by Jim Gray who named atomicity, consistency, and durability, but not isolation, when characterizing the transaction concept. These four properties are the major guarantees of the transaction paradigm, which has influenced many aspects of development in database systems. According to Gray and Reuter, the IBM Inf ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Karlsruhe Congress
The Karlsruhe Congress was an international meeting of chemists held in Karlsruhe, Germany from 3 to 5 September 1860. It was the first international conference of chemistry worldwide. The meeting The Karlsruhe Congress was called so that European chemists could discuss matters of chemical nomenclature, notation, and atomic weights. The organization, invitation, and sponsorship of the conference were handled by August Kekulé, Adolphe Wurtz, and Karl Weltzien. As an example of the problems facing the delegates, Kekulé's ''Lehrbuch der Organischen Chemie'' gave nineteen different formulas used by chemists for acetic acid, as shown in the figure on this page. An understanding was reached on the time and place of the meeting, and printing of a circular addressed to European chemists listed below, which explained the objectives and goals of an international congress was agreed upon. The circular concluded: "...with the aim of avoiding any unfortunate omissions, the undersigned ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Stanislao Cannizzaro
Stanislao Cannizzaro ( , also , ; 13 July 1826 – 10 May 1910) was an Italian chemist. He is famous for the Cannizzaro reaction and for his influential role in the atomic-weight deliberations of the Karlsruhe Congress in 1860. Biography Cannizzaro was born in Palermo in 1826. He entered the university there with the intention of making medicine his profession, but he soon turned to the study of chemistry. In 1845 and 1846, he acted as assistant to Raffaele Piria (1815–1865), known for his work on salicin, and who was then professor of chemistry at Pisa and subsequently occupied the same position at Turin. During the Sicilian revolution of independence of 1848, Cannizzaro served as an artillery officer at Messina and was also chosen deputy for Francavilla in the Sicilian parliament; and, after the fall of Messina in September 1848, he was stationed at Taormina. On the collapse of the insurgents, Cannizzaro escaped to Marseille in May 1849, and, after visiting vario ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Henri Victor Regnault
Henri Victor Regnault (21 July 1810 – 19 January 1878) was a French chemist and physicist best known for his careful measurements of the thermal properties of gases. He was an early thermodynamicist and was mentor to William Thomson in the late 1840s. He never used his first given name, and was known throughout his lifetime as Victor Regnault. Biography Born in Aix-la-Chapelle in 1810 (modern Aachen, Germany and at that time under French rule), he moved to Paris at the age of eight, following the death of his parents. There, he worked for an upholstery firm until he was eighteen. In 1830, he was admitted to the École Polytechnique, and in 1832 he graduated from the École des mines. Working under Justus von Liebig at Gießen, Regnault distinguished himself in the nascent field of organic chemistry by synthesizing several chlorinated hydrocarbons (e.g. vinyl chloride, polyvinylidene chloride, dichloromethane), and he was appointed professor of chemistry at the Univers ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Charles Frédéric Gerhardt
Charles Frédéric Gerhardt (21 August 1816 – 19 August 1856) was a French chemist, born in Alsace and active in Paris, Montpellier, and his native Strasbourg. Biography He was born in Strasbourg, which is where he attended the gymnasium (an advanced academic secondary school). He then studied at the Karlsruhe Institute of Technology, where Friedrich Walchner's lectures first stimulated his interest in chemistry. Next he attended the school of commerce in Leipzig, where he studied chemistry under Otto Linné Erdmann, who further developed his interest into a passion for questions of speculative chemistry. Returning home in 1834, he entered his father's white lead factory, but soon found that business was not to his liking, and after a sharp disagreement with his father in his 20th year he enlisted in a cavalry regiment. In a few months military life became equally distasteful, and he purchased his discharge with the assistance of the German chemist Justus von Liebig. After a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Gay-Lussac's Law
Gay-Lussac's law usually refers to Joseph-Louis Gay-Lussac's law of combining volumes of gases, discovered in 1808 and published in 1809. It sometimes refers to the proportionality of the volume of a gas to its absolute temperature at constant pressure. This law was published by Gay-Lussac in 1802, and in the article in which he described his work he cited earlier unpublished work from the 1780s by Jacques Charles. Consequently, the volume-temperature proportionality is usually known as Charles's Law. Law of combining volumes The law of combining volumes states that, when gases react together they do so in volume which bears simple whole number ratio provided that the temperature and pressure of the reacting gases and their products remain constant The ratio between the volumes of the reactant gases and the gaseous products can be expressed in simple whole numbers. For example, Gay-Lussac found that two volumes of hydrogen and one volume of oxygen would react to form two vo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
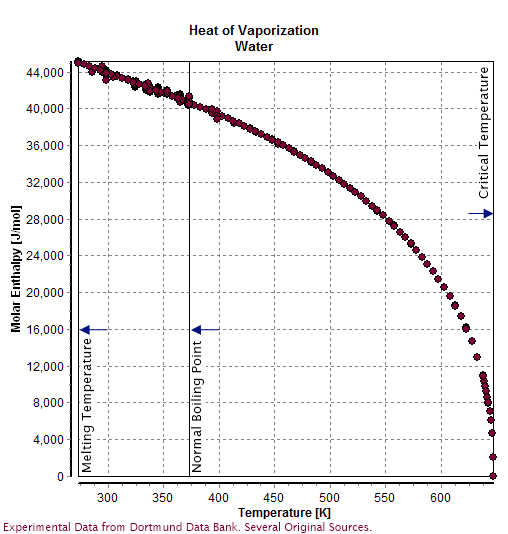
Water (molecule)
Water () is a polar inorganic compound that is at room temperature a tasteless and odorless liquid, which is nearly colorless apart from an inherent hint of blue. It is by far the most studied chemical compound and is described as the "universal solvent" and the "solvent of life". It is the most abundant substance on the surface of Earth and the only common substance to exist as a solid, liquid, and gas on Earth's surface. It is also the third most abundant molecule in the universe (behind molecular hydrogen and carbon monoxide). Water molecules form hydrogen bonds with each other and are strongly polar. This polarity allows it to dissociate ions in salts and bond to other polar substances such as alcohols and acids, thus dissolving them. Its hydrogen bonding causes its many unique properties, such as having a solid form less dense than its liquid form, a relatively high boiling point of 100 °C for its molar mass, and a high heat capacity. Water is amphoteric, mean ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Oxygen
Oxygen is the chemical element with the symbol O and atomic number 8. It is a member of the chalcogen group in the periodic table, a highly reactive nonmetal, and an oxidizing agent that readily forms oxides with most elements as well as with other compounds. Oxygen is Earth's most abundant element, and after hydrogen and helium, it is the third-most abundant element in the universe. At standard temperature and pressure, two atoms of the element bind to form dioxygen, a colorless and odorless diatomic gas with the formula . Diatomic oxygen gas currently constitutes 20.95% of the Earth's atmosphere, though this has changed considerably over long periods of time. Oxygen makes up almost half of the Earth's crust in the form of oxides.Atkins, P.; Jones, L.; Laverman, L. (2016).''Chemical Principles'', 7th edition. Freeman. Many major classes of organic molecules in living organisms contain oxygen atoms, such as proteins, nucleic acids, carbohydrates, and ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dalton's Atomic Theory
John Dalton (; 5 or 6 September 1766 – 27 July 1844) was an English chemist, physicist and meteorologist. He is best known for introducing the atomic theory into chemistry, and for his research into colour blindness, which he had. Colour blindness is known as ''Daltonism'' in several languages, being named after him. Early life John Dalton was born into a Quaker family in Eaglesfield, near Cockermouth, in Cumberland, England. His father was a weaver. He received his early education from his father and from Quaker John Fletcher, who ran a private school in the nearby village of Pardshaw Hall. Dalton's family was too poor to support him for long and he began to earn his living, from the age of ten, in the service of wealthy local Quaker Elihu Robinson. Early career When he was 15, Dalton joined his older brother Jonathan in running a Quaker school in Kendal, Westmorland, about from his home. Around the age of 23, Dalton may have considered studying law or medic ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrogen
Hydrogen is the chemical element with the symbol H and atomic number 1. Hydrogen is the lightest element. At standard conditions hydrogen is a gas of diatomic molecules having the formula . It is colorless, odorless, tasteless, non-toxic, and highly combustible. Hydrogen is the most abundant chemical substance in the universe, constituting roughly 75% of all normal matter.However, most of the universe's mass is not in the form of baryons or chemical elements. See dark matter and dark energy. Stars such as the Sun are mainly composed of hydrogen in the plasma state. Most of the hydrogen on Earth exists in molecular forms such as water and organic compounds. For the most common isotope of hydrogen (symbol 1H) each atom has one proton, one electron, and no neutrons. In the early universe, the formation of protons, the nuclei of hydrogen, occurred during the first second after the Big Bang. The emergence of neutral hydrogen atoms throughout the universe occur ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
John Dalton
John Dalton (; 5 or 6 September 1766 – 27 July 1844) was an English chemist, physicist and meteorologist. He is best known for introducing the atomic theory into chemistry, and for his research into Color blindness, colour blindness, which he had. Colour blindness is known as ''Daltonism'' in several languages, being named after him. Early life John Dalton was born into a Quaker family in Eaglesfield, Cumbria, Eaglesfield, near Cockermouth, in Cumberland, England. His father was a weaver. He received his early education from his father and from Quaker John Fletcher, who ran a private school in the nearby village of Pardshaw Young Friends' Centre, Pardshaw Hall. Dalton's family was too poor to support him for long and he began to earn his living, from the age of ten, in the service of wealthy local Quaker Elihu Robinson. Early career When he was 15, Dalton joined his older brother Jonathan in running a Quaker school in Kendal, Westmorland, about from his home. Aroun ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |