Structural Biology on:
[Wikipedia]
[Google]
[Amazon]
Structural biology deals with structural analysis of living material (formed, composed of, and/or maintained and refined by living cells) at every level of organization.
Early structural biologists throughout the 19th and early 20th centuries were primarily only able to study structures to the limit of the naked eye's visual acuity and through magnifying glasses and light microscopes. In the 20th century, a variety of experimental techniques were developed to examine the 3D structures of biological molecules. The most prominent techniques are 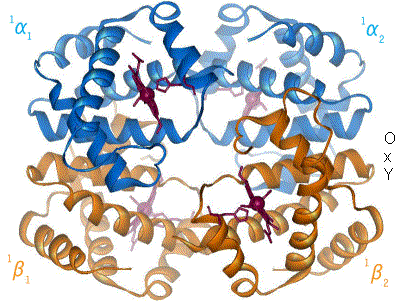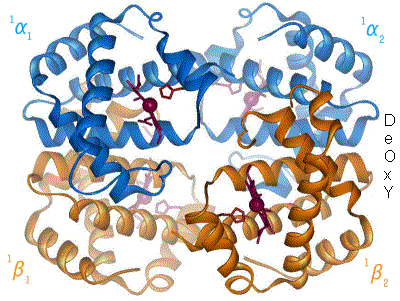
 Structural biologists have made significant contributions towards understanding the molecular components and mechanisms underlying human diseases. For example, cryo-EM and ssNMR have been used to study the aggregation of amyloid fibrils, which are associated with
Structural biologists have made significant contributions towards understanding the molecular components and mechanisms underlying human diseases. For example, cryo-EM and ssNMR have been used to study the aggregation of amyloid fibrils, which are associated with
''Nature: Structural & Molecular Biology'' magazine website
Journal of Structural Biology
Structural Biology - The Virtual Library of Biochemistry, Molecular Biology and Cell Biology
''Structural Biology in Europe''
{{Authority control Molecular biology Protein structure Biophysics
X-ray crystallography
X-ray crystallography is the experimental science of determining the atomic and molecular structure of a crystal, in which the crystalline structure causes a beam of incident X-rays to Diffraction, diffract in specific directions. By measuring th ...
, nuclear magnetic resonance
Nuclear magnetic resonance (NMR) is a physical phenomenon in which nuclei in a strong constant magnetic field are disturbed by a weak oscillating magnetic field (in the near field) and respond by producing an electromagnetic signal with a ...
, and electron microscopy
An electron microscope is a microscope that uses a beam of electrons as a source of illumination. It uses electron optics that are analogous to the glass lenses of an optical light microscope to control the electron beam, for instance focusing i ...
. Through the discovery of X-rays
An X-ray (also known in many languages as Röntgen radiation) is a form of high-energy electromagnetic radiation with a wavelength shorter than those of ultraviolet rays and longer than those of gamma rays. Roughly, X-rays have a wavelength ran ...
and its applications to protein crystals, structural biology was revolutionized, as now scientists could obtain the three-dimensional structures of biological molecules in atomic detail. Likewise, NMR spectroscopy allowed information about protein structure and dynamics to be obtained. Finally, in the 21st century, electron microscopy also saw a drastic revolution with the development of more coherent electron sources, aberration correction for electron microscopes, and reconstruction software that enabled the successful implementation of high resolution cryo-electron microscopy, thereby permitting the study of individual proteins and molecular complexes in three-dimensions at angstrom resolution.
With the development of these three techniques, the field of structural biology expanded and also became a branch of molecular biology
Molecular biology is a branch of biology that seeks to understand the molecule, molecular basis of biological activity in and between Cell (biology), cells, including biomolecule, biomolecular synthesis, modification, mechanisms, and interactio ...
, biochemistry
Biochemistry, or biological chemistry, is the study of chemical processes within and relating to living organisms. A sub-discipline of both chemistry and biology, biochemistry may be divided into three fields: structural biology, enzymology, a ...
, and biophysics
Biophysics is an interdisciplinary science that applies approaches and methods traditionally used in physics to study biological phenomena. Biophysics covers all scales of biological organization, from molecular to organismic and populations ...
concerned with the molecular structure of biological macromolecule
A macromolecule is a "molecule of high relative molecular mass, the structure of which essentially comprises the multiple repetition of units derived, actually or conceptually, from molecules of low relative molecular mass." Polymers are physi ...
s (especially protein
Proteins are large biomolecules and macromolecules that comprise one or more long chains of amino acid residue (biochemistry), residues. Proteins perform a vast array of functions within organisms, including Enzyme catalysis, catalysing metab ...
s, made up of amino acid
Amino acids are organic compounds that contain both amino and carboxylic acid functional groups. Although over 500 amino acids exist in nature, by far the most important are the 22 α-amino acids incorporated into proteins. Only these 22 a ...
s, RNA
Ribonucleic acid (RNA) is a polymeric molecule that is essential for most biological functions, either by performing the function itself (non-coding RNA) or by forming a template for the production of proteins (messenger RNA). RNA and deoxyrib ...
or DNA
Deoxyribonucleic acid (; DNA) is a polymer composed of two polynucleotide chains that coil around each other to form a double helix. The polymer carries genetic instructions for the development, functioning, growth and reproduction of al ...
, made up of nucleotide
Nucleotides are Organic compound, organic molecules composed of a nitrogenous base, a pentose sugar and a phosphate. They serve as monomeric units of the nucleic acid polymers – deoxyribonucleic acid (DNA) and ribonucleic acid (RNA), both o ...
s, and membranes, made up of lipid
Lipids are a broad group of organic compounds which include fats, waxes, sterols, fat-soluble vitamins (such as vitamins A, D, E and K), monoglycerides, diglycerides, phospholipids, and others. The functions of lipids include storing ...
s), how they acquire the structures they have, and how alterations in their structures affect their function. This subject is of great interest to biologists because macromolecules carry out most of the functions of cells, and it is only by coiling into specific three-dimensional shapes that they are able to perform these functions. This architecture, the "tertiary structure
Protein tertiary structure is the three-dimensional shape of a protein. The tertiary structure will have a single polypeptide chain "backbone" with one or more protein secondary structures, the protein domains. Amino acid side chains and the ...
" of molecules, depends in a complicated way on each molecule's basic composition, or "primary structure
Protein primary structure is the linear sequence of amino acids in a peptide or protein. By convention, the primary structure of a protein is reported starting from the amino-terminal (N) end to the carboxyl-terminal (C) end. Protein biosynthe ...
." At lower resolutions, tools such as FIB-SEM tomography have allowed for greater understanding of cells and their organelles in 3-dimensions, and how each hierarchical level of various extracellular matrices contributes to function (for example in bone). In the past few years it has also become possible to predict highly accurate physical molecular model
A molecular model is a physical model of an atomistic system that represents molecules and their processes. They play an important role in understanding chemistry and generating and testing hypotheses. The creation of mathematical models of mole ...
s to complement the experimental study of biological structures. Computational techniques such as molecular dynamics
Molecular dynamics (MD) is a computer simulation method for analyzing the Motion (physics), physical movements of atoms and molecules. The atoms and molecules are allowed to interact for a fixed period of time, giving a view of the dynamics ( ...
simulations can be used in conjunction with empirical structure determination strategies to extend and study protein structure, conformation and function.

History
In 1912Max Von Laue
Max Theodor Felix von Laue (; 9 October 1879 – 24 April 1960) was a German physicist who received the Nobel Prize in Physics in 1914 "for his discovery of the X-ray diffraction, diffraction of X-rays by crystals".
In addition to his scientifi ...
directed X-rays at crystallized copper sulfate Copper sulfate may refer to:
* Copper(II) sulfate, CuSO4, a common, greenish blue compound used as a fungicide and herbicide
* Copper(I) sulfate, Cu2SO4, an unstable white solid which is uncommonly used
{{chemistry index
Copper compounds ...
generating a diffraction pattern. These experiments led to the development of X-ray crystallography
X-ray crystallography is the experimental science of determining the atomic and molecular structure of a crystal, in which the crystalline structure causes a beam of incident X-rays to Diffraction, diffract in specific directions. By measuring th ...
, and its usage in exploring biological structures. In 1951, Rosalind Franklin
Rosalind Elsie Franklin (25 July 192016 April 1958) was a British chemist and X-ray crystallographer. Her work was central to the understanding of the molecular structures of DNA (deoxyribonucleic acid), RNA (ribonucleic acid), viruses, coal ...
and Maurice Wilkins
Maurice Hugh Frederick Wilkins (15 December 1916 – 5 October 2004) was a New Zealand-born British biophysicist and Nobel laureate whose research spanned multiple areas of physics and biophysics, contributing to the scientific understanding ...
used X-ray diffraction patterns to capture the first image of deoxyribonucleic acid (DNA). Francis Crick
Francis Harry Compton Crick (8 June 1916 – 28 July 2004) was an English molecular biologist, biophysicist, and neuroscientist. He, James Watson, Rosalind Franklin, and Maurice Wilkins played crucial roles in deciphering the Nucleic acid doub ...
and James Watson
James Dewey Watson (born April 6, 1928) is an American molecular biology, molecular biologist, geneticist, and zoologist. In 1953, he co-authored with Francis Crick the academic paper in ''Nature (journal), Nature'' proposing the Nucleic acid ...
modeled the double helical structure of DNA using this same technique in 1953 and received the Nobel Prize in Medicine along with Wilkins in 1962.
Pepsin
Pepsin is an endopeptidase that breaks down proteins into smaller peptides and amino acids. It is one of the main digestive enzymes in the digestive systems of humans and many other animals, where it helps digest the proteins in food. Pe ...
crystals were the first proteins to be crystallized for use in X-ray diffraction, by Theodore Svedberg who received the 1962 Nobel Prize in Chemistry. The first tertiary protein structure, that of myoglobin
Myoglobin (symbol Mb or MB) is an iron- and oxygen-binding protein found in the cardiac and skeletal muscle, skeletal Muscle, muscle tissue of vertebrates in general and in almost all mammals. Myoglobin is distantly related to hemoglobin. Compar ...
, was published in 1958 by John Kendrew. During this time, modeling of protein structures was done using balsa wood or wire
file:Sample cross-section of high tension power (pylon) line.jpg, Overhead power cabling. The conductor consists of seven strands of steel (centre, high tensile strength), surrounded by four outer layers of aluminium (high conductivity). Sample d ...
models. With the invention of modeling software such as CCP4 in the late 1970s, modeling is now done with computer assistance. Recent developments in the field have included the generation of X-ray free electron lasers, allowing analysis of the dynamics and motion of biological molecules, and the use of structural biology in assisting synthetic biology
Synthetic biology (SynBio) is a multidisciplinary field of science that focuses on living systems and organisms. It applies engineering principles to develop new biological parts, devices, and systems or to redesign existing systems found in nat ...
.
In the late 1930s and early 1940s, the combination of work done by Isidor Rabi, Felix Bloch
Felix Bloch (; ; 23 October 1905 – 10 September 1983) was a Swiss-American physicist who shared the 1952 Nobel Prize in Physics with Edward Mills Purcell "for their development of new methods for nuclear magnetic precision measurements and di ...
, and Edward Mills Purcell
Edward Mills Purcell (August 30, 1912 – March 7, 1997) was an American physicist who shared the 1952 Nobel Prize for Physics for his independent discovery (published 1946) of nuclear magnetic resonance in liquids and in solids. Nuclear magn ...
led to the development of nuclear magnetic resonance (NMR). Currently, solid-state NMR
Solid-state nuclear magnetic resonance (ssNMR) is a spectroscopy technique used to characterize atomic-level structure and dynamics in solid materials. ssNMR spectra are broader due to nuclear spin interactions which can be categorized as dipolar ...
is widely used in the field of structural biology to determine the structure and dynamic nature of proteins (protein NMR
Nuclear magnetic resonance spectroscopy of proteins (usually abbreviated protein NMR) is a field of structural biology in which NMR spectroscopy is used to obtain information about the structure and dynamics of proteins, and also nucleic acids, and ...
).
In 1990, Richard Henderson produced the first three-dimensional, high resolution image of bacteriorhodopsin using cryogenic electron microscopy
Cryogenic electron microscopy (cryo-EM) is a transmission electron microscopy technique applied to samples cooled to cryogenic temperatures. For biological specimens, the structure is preserved by embedding in an environment of vitreous ice. An ...
(cryo-EM). Since then, cryo-EM has emerged as an increasingly popular technique to determine three-dimensional, high resolution structures of biological images.
More recently, computational methods have been developed to model and study biological structures. For example, molecular dynamics
Molecular dynamics (MD) is a computer simulation method for analyzing the Motion (physics), physical movements of atoms and molecules. The atoms and molecules are allowed to interact for a fixed period of time, giving a view of the dynamics ( ...
(MD) is commonly used to analyze the dynamic movements of biological molecules. In 1975, the first simulation of a biological folding process using MD was published in Nature. Recently, protein structure prediction
Protein structure prediction is the inference of the three-dimensional structure of a protein from its amino acid sequence—that is, the prediction of its Protein secondary structure, secondary and Protein tertiary structure, tertiary structure ...
was significantly improved by a new machine learning method called AlphaFold. Some claim that computational approaches are starting to lead the field of structural biology research.
Techniques
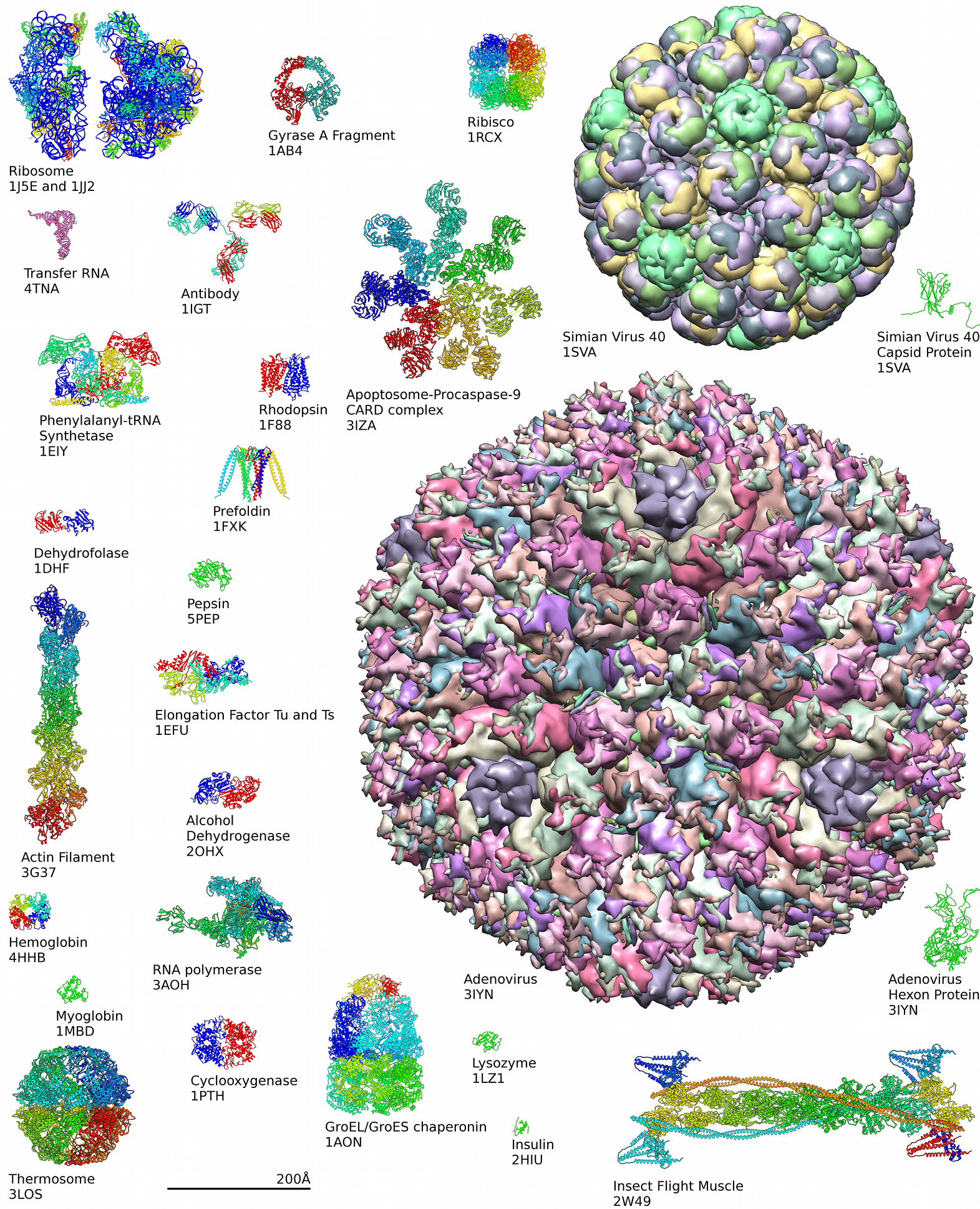
Biomolecule
A biomolecule or biological molecule is loosely defined as a molecule produced by a living organism and essential to one or more typically biological processes. Biomolecules include large macromolecules such as proteins, carbohydrates, lipids ...
s are too small to see in detail even with the most advanced light microscope
A microscope () is a laboratory equipment, laboratory instrument used to examine objects that are too small to be seen by the naked eye. Microscopy is the science of investigating small objects and structures using a microscope. Microscopic ...
s. The methods that structural biologists use to determine their structures generally involve measurements on vast numbers of identical molecules at the same time. These methods include:
* Mass spectrometry
Mass spectrometry (MS) is an analytical technique that is used to measure the mass-to-charge ratio of ions. The results are presented as a ''mass spectrum'', a plot of intensity as a function of the mass-to-charge ratio. Mass spectrometry is used ...
* Macromolecular crystallography
* Neutron diffraction
Neutron diffraction or elastic neutron scattering is the application of neutron scattering to the determination of the atomic and/or magnetic structure of a material. A sample to be examined is placed in a beam of Neutron temperature, thermal or ...
* Proteolysis
Proteolysis is the breakdown of proteins into smaller polypeptides or amino acids. Protein degradation is a major regulatory mechanism of gene expression and contributes substantially to shaping mammalian proteomes. Uncatalysed, the hydrolysis o ...
* Nuclear magnetic resonance spectroscopy of proteins (NMR)
* Electron paramagnetic resonance
Electron paramagnetic resonance (EPR) or electron spin resonance (ESR) spectroscopy is a method for studying materials that have unpaired electrons. The basic concepts of EPR are analogous to those of nuclear magnetic resonance (NMR), but the spin ...
(EPR)
* Cryogenic electron microscopy
Cryogenic electron microscopy (cryo-EM) is a transmission electron microscopy technique applied to samples cooled to cryogenic temperatures. For biological specimens, the structure is preserved by embedding in an environment of vitreous ice. An ...
(cryoEM)
* Electron crystallography and microcrystal electron diffraction
* Multiangle light scattering
* Small angle scattering
* Ultrafast laser spectroscopy
* Anisotropic terahertz microspectroscopy
* Two-dimensional infrared spectroscopy
* Dual-polarization interferometry and circular dichroism
Circular dichroism (CD) is dichroism involving circular polarization, circularly polarized light, i.e., the differential Absorption (electromagnetic radiation), absorption of left- and right-handed light. Left-hand circular (LHC) and right-hand ci ...
Most often researchers use them to study the "native state
In biochemistry, the native state of a protein or nucleic acid is its properly Protein folding, folded and/or assembled form, which is operative and functional. The native state of a biomolecule may possess all four levels of biomolecular structu ...
s" of macromolecules. But variations on these methods are also used to watch nascent or denatured molecules assume or reassume their native states. See protein folding
Protein folding is the physical process by which a protein, after Protein biosynthesis, synthesis by a ribosome as a linear chain of Amino acid, amino acids, changes from an unstable random coil into a more ordered protein tertiary structure, t ...
.
A third approach that structural biologists take to understanding structure is bioinformatics
Bioinformatics () is an interdisciplinary field of science that develops methods and Bioinformatics software, software tools for understanding biological data, especially when the data sets are large and complex. Bioinformatics uses biology, ...
to look for patterns among the diverse sequence
In mathematics, a sequence is an enumerated collection of objects in which repetitions are allowed and order matters. Like a set, it contains members (also called ''elements'', or ''terms''). The number of elements (possibly infinite) is cal ...
s that give rise to particular shapes. Researchers often can deduce aspects of the structure of integral membrane protein
An integral, or intrinsic, membrane protein (IMP) is a type of membrane protein that is permanently attached to the biological membrane. All transmembrane proteins can be classified as IMPs, but not all IMPs are transmembrane proteins. IMPs comp ...
s based on the membrane topology predicted by hydrophobicity analysis. See protein structure prediction
Protein structure prediction is the inference of the three-dimensional structure of a protein from its amino acid sequence—that is, the prediction of its Protein secondary structure, secondary and Protein tertiary structure, tertiary structure ...
.
Applications
 Structural biologists have made significant contributions towards understanding the molecular components and mechanisms underlying human diseases. For example, cryo-EM and ssNMR have been used to study the aggregation of amyloid fibrils, which are associated with
Structural biologists have made significant contributions towards understanding the molecular components and mechanisms underlying human diseases. For example, cryo-EM and ssNMR have been used to study the aggregation of amyloid fibrils, which are associated with Alzheimer's disease
Alzheimer's disease (AD) is a neurodegenerative disease and the cause of 60–70% of cases of dementia. The most common early symptom is difficulty in remembering recent events. As the disease advances, symptoms can include problems wit ...
, Parkinson's disease
Parkinson's disease (PD), or simply Parkinson's, is a neurodegenerative disease primarily of the central nervous system, affecting both motor system, motor and non-motor systems. Symptoms typically develop gradually and non-motor issues become ...
, and type II diabetes
Type 2 diabetes (T2D), formerly known as adult-onset diabetes, is a form of diabetes mellitus that is characterized by high blood sugar, insulin resistance, and relative lack of insulin. Common symptoms include increased thirst, frequent ...
. In addition to amyloid proteins, scientists have used cryo-EM to produce high resolution models of tau filaments in the brain of Alzheimer's patients which may help develop better treatments in the future. Structural biology tools can also be used to explain interactions between pathogens and hosts. For example, structural biology tools have enabled virologists to understand how the HIV envelope allows the virus to evade human immune responses.
Structural biology is also an important component of drug discovery
In the fields of medicine, biotechnology, and pharmacology, drug discovery is the process by which new candidate medications are discovered.
Historically, drugs were discovered by identifying the active ingredient from traditional remedies or ...
. Scientists can identify targets using genomics, study those targets using structural biology, and develop drugs that are suited for those targets. Specifically, ligand-NMR
Nuclear magnetic resonance (NMR) is a physical phenomenon in which atomic nucleus, nuclei in a strong constant magnetic field are disturbed by a weak oscillating magnetic field (in the near and far field, near field) and respond by producing ...
, mass spectrometry
Mass spectrometry (MS) is an analytical technique that is used to measure the mass-to-charge ratio of ions. The results are presented as a ''mass spectrum'', a plot of intensity as a function of the mass-to-charge ratio. Mass spectrometry is used ...
, and X-ray crystallography
X-ray crystallography is the experimental science of determining the atomic and molecular structure of a crystal, in which the crystalline structure causes a beam of incident X-rays to Diffraction, diffract in specific directions. By measuring th ...
are commonly used techniques in the drug discovery process. For example, researchers have used structural biology to better understand Met, a protein encoded by a protooncogene that is an important drug target in cancer
Cancer is a group of diseases involving Cell growth#Disorders, abnormal cell growth with the potential to Invasion (cancer), invade or Metastasis, spread to other parts of the body. These contrast with benign tumors, which do not spread. Po ...
. Similar research has been conducted for HIV targets to treat people with AIDS
The HIV, human immunodeficiency virus (HIV) is a retrovirus that attacks the immune system. Without treatment, it can lead to a spectrum of conditions including acquired immunodeficiency syndrome (AIDS). It is a Preventive healthcare, pr ...
. Researchers are also developing new antimicrobials for mycobacterial infections using structure-driven drug discovery.
See also
*Primary structure
Protein primary structure is the linear sequence of amino acids in a peptide or protein. By convention, the primary structure of a protein is reported starting from the amino-terminal (N) end to the carboxyl-terminal (C) end. Protein biosynthe ...
* Secondary structure
Protein secondary structure is the local spatial conformation of the polypeptide backbone excluding the side chains. The two most common Protein structure#Secondary structure, secondary structural elements are alpha helix, alpha helices and beta ...
* Tertiary structure
Protein tertiary structure is the three-dimensional shape of a protein. The tertiary structure will have a single polypeptide chain "backbone" with one or more protein secondary structures, the protein domains. Amino acid side chains and the ...
* Quaternary structure
* Structural domain
In molecular biology, a protein domain is a region of a protein's Peptide, polypeptide chain that is self-stabilizing and that Protein folding, folds independently from the rest. Each domain forms a compact folded Protein tertiary structure, thre ...
* Structural motif
In a chain-like biological molecule, such as a protein or nucleic acid, a structural motif is a common three-dimensional structure that appears in a variety of different, evolutionarily unrelated molecules. A structural motif does not have t ...
* Protein subunit
In structural biology, a protein subunit is a polypeptide chain or single protein molecule that assembles (or "''coassembles''") with others to form a protein complex.
Large assemblies of proteins such as viruses often use a small number of t ...
* Molecular model
A molecular model is a physical model of an atomistic system that represents molecules and their processes. They play an important role in understanding chemistry and generating and testing hypotheses. The creation of mathematical models of mole ...
* Cooperativity
* Chaperonin
* Structural genomics
* Stereochemistry
Stereochemistry, a subdiscipline of chemistry, studies the spatial arrangement of atoms that form the structure of molecules and their manipulation. The study of stereochemistry focuses on the relationships between stereoisomers, which are defined ...
* Resolution (electron density)
Resolution in the context of structural biology is the ability to distinguish the presence or absence of Atom, atoms or groups of atoms in a Biomolecule, biomolecular structure. Usually, the structure originates from methods such as X-ray crystallo ...
* Proteopedia The collaborative, 3D encyclopedia of proteins and other molecules
A molecule is a group of two or more atoms that are held together by attractive forces known as chemical bonds; depending on context, the term may or may not include ions that satisfy this criterion. In quantum physics, organic chemistry ...
.
* Protein structure prediction
Protein structure prediction is the inference of the three-dimensional structure of a protein from its amino acid sequence—that is, the prediction of its Protein secondary structure, secondary and Protein tertiary structure, tertiary structure ...
* SBGrid Consortium
References
Further reading
* *External links
*''Nature: Structural & Molecular Biology'' magazine website
Journal of Structural Biology
Structural Biology - The Virtual Library of Biochemistry, Molecular Biology and Cell Biology
''Structural Biology in Europe''
{{Authority control Molecular biology Protein structure Biophysics