|
Tetrahydroxozincate
In chemistry, tetrahydroxozincate or tetrahydroxidozincate is a valence (chemistry), divalent anion (negative ion) with formula , with a central zinc atom in the +2 or (II) valence state coordination compound, coordinated to four hydroxide groups. It has Sp3 hybridization. It is the most common of the zincate anions, and is often called just zincate. These names are also used for the salts containing that anion, such as sodium zincate Na2Zn(OH)4 and calcium zincate CaZn(OH)4·2H2O Zincate salts can be obtained by reaction of zinc oxide (ZnO) or zinc hydroxide () and a strong base (chemistry), base like sodium hydroxide. It is now generally accepted that the resulting solutions contain the tetrahydroxozincate ion. Earlier Raman studies had been interpreted as indicating the existence of linear ions. Related anions and salts The name "zincate" may also refer to a polymeric anion with formula approaching []''n'', which forms salts such as ·, or to mixed oxides of zinc and less ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Zincate
In chemistry the term zincate may refer to several substances containing the element zinc: * usually the anion Zn(OH)42−, more properly called tetrahydroxozincate or salt (chemistry), salts thereof, such as sodium zincate . * the polymeric anion [Zn(OH)3−] and its salts, for example NaZn(OH)3· H2O. * an oxide containing zinc and a less electronegativity, electronegative element e.g. Na2ZnO2. In the health supplement industry zincate may also mean a commercially available zinc supplement, typically formulated as zinc sulfate. Solution (chemistry), Solutions prepared from dissolving zinc hydroxide or zinc oxide in a strong alkali like sodium hydroxide, which contains various zincate anions, are used in the metal plating industry, in processes such as immersion zinc plating and electroplating (electrogalvanization). Any of these techniques may be called zincate process. Inorganic compound nomenclature In the IUPAC nomenclature of inorganic chemistry, naming of inorganic comp ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
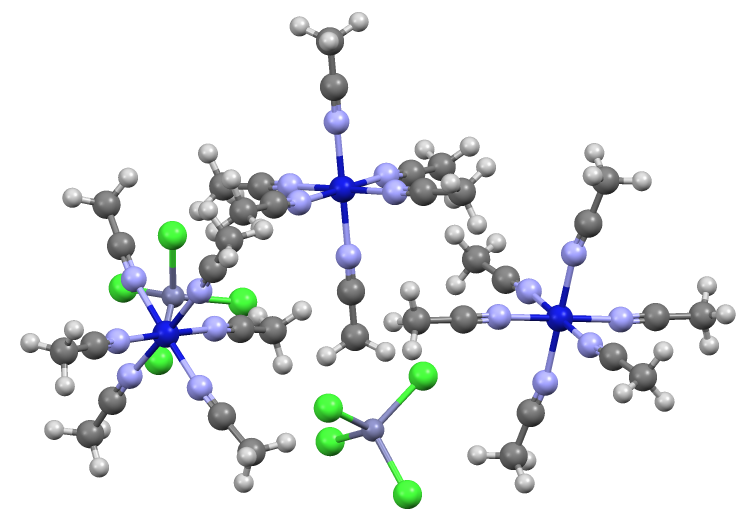
Sodium Zincate
Sodium zincate refers to anionic zinc oxides or hydroxides, depending on conditions. In the applications of these materials, the exact formula is not necessarily important and it is likely that aqueous zincate solutions consist of mixtures. Hydroxyzincates Solutions of sodium zincate may be prepared by dissolving zinc, zinc hydroxide, or zinc oxide in an aqueous solution of sodium hydroxide Sodium hydroxide, also known as lye and caustic soda, is an inorganic compound with the formula . It is a white solid ionic compound consisting of sodium cations and hydroxide anions . Sodium hydroxide is a highly corrosive base (chemistry), .... Simplified equations for these complex processes are: :ZnO + H2O + 2 NaOH → Na2Zn(OH)4 :Zn + 2 H2O + 2 NaOH → Na2Zn(OH)4 + H2 From such solutions, one can crystallize salts of containing the anions Zn(OH)42−, Zn2(OH)62−, and Zn(OH)64−. Na2Zn(OH)4 consists of tetrahedral zincate ion and octahedral sodium cations. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chemistry
Chemistry is the scientific study of the properties and behavior of matter. It is a physical science within the natural sciences that studies the chemical elements that make up matter and chemical compound, compounds made of atoms, molecules and ions: their composition, structure, properties, behavior and the changes they undergo during chemical reaction, reactions with other chemical substance, substances. Chemistry also addresses the nature of chemical bonds in chemical compounds. In the scope of its subject, chemistry occupies an intermediate position between physics and biology. It is sometimes called the central science because it provides a foundation for understanding both Basic research, basic and Applied science, applied scientific disciplines at a fundamental level. For example, chemistry explains aspects of plant growth (botany), the formation of igneous rocks (geology), how atmospheric ozone is formed and how environmental pollutants are degraded (ecology), the prop ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sodium Hydroxide
Sodium hydroxide, also known as lye and caustic soda, is an inorganic compound with the formula . It is a white solid ionic compound consisting of sodium cations and hydroxide anions . Sodium hydroxide is a highly corrosive base (chemistry), base and alkali that decomposes lipids and proteins at ambient temperatures and at high concentrations may cause severe chemical burns. It is highly soluble in water, and readily absorbs moisture and carbon dioxide from the air. It forms a series of hydrates . The monohydrate crystallizes from water solutions between 12.3 and 61.8 °C. The commercially available "sodium hydroxide" is often this monohydrate, and published data may refer to it instead of the anhydrous compound. As one of the simplest hydroxides, sodium hydroxide is frequently used alongside neutral water and acidic hydrochloric acid to demonstrate the pH scale to chemistry students. Sodium hydroxide is used in many industries: in the making of wood pulp and paper, tex ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
IUPAC Nomenclature Of Inorganic Chemistry 2005
Nomenclature of Inorganic Chemistry, IUPAC Recommendations 2005 is the 2005 version of ''Nomenclature of Inorganic Chemistry'' (which is informally called the Red Book). It is a collection of rules for naming inorganic compounds, as recommended by the International Union of Pure and Applied Chemistry (IUPAC). Summary The 2005 edition replaces their previous recommendations ''Nomenclature The Red Book of Inorganic Chemistry, IUPAC Recommendations 1990 (Red Book I)'', and "where appropriate" (sic) ''Nomenclature of Inorganic Chemistry II, IUPAC Recommendations 2000 (Red Book II)''. The recommendations take up over 300 pages''Nomenclature of Inorganic Chemistry IUPAC Recommendations'' 2005 ed. N. G. Connelly et al. RSC Publishing https://iupac.org/what-we-do/books/redbook/ and the full text can be downloaded from IUPAC. Corrections have been issued. Apart from a reorganisation of the content, there is a new section on organometallics and a formal element list to be used in place o ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Zeitschrift Für Anorganische Und Allgemeine Chemie
The ''Zeitschrift für anorganische und allgemeine Chemie'' (''Journal of Inorganic and General Chemistry'') is a semimonthly peer-reviewed scientific journal covering inorganic chemistry, published by Wiley-VCH. The editors-in-chief are Thomas F. Fässler, Christian Limberg, Guodong Qian, and David Scheschkewitz. Originally the journal was published in German, but nowadays it is completely in English. Abstracting and indexing The journal is abstracted and indexed in: According to the ''Journal Citation Reports'', the journal has a 2022 impact factor of 1.4. History In 1892, Gerhard Krüss (Ludwig Maximilian University of Munich), established the journal under the name ''Zeitschrift für Anorganische Chemie''. Krüss died three years later, and was succeeded by Richard Lorenz and Walther Nernst Walther Hermann Nernst (; 25 June 1864 – 18 November 1941) was a German physical chemist known for his work in thermodynamics, physical chemistry, electrochemistry, and solid-sta ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tetrachlorozincate
Tetrachlorozincate is an anion with the formula . It is a counterion that is often used in conjunction with strong electrophiles. Being dianionic, tetrachlorozincate is not classified as a weakly coordinating anion. On the other hand, being dianionic, tetrachlorozincate facilitates the crystallization of many salts. It has a tetrahedral molecular geometry. A simple example is (ammonium tetrachlorozincate). Zincates are anionic zinc complexes. Related to the preparation of Lucas' reagent, tetrachlorozincates are often generated by combining hydrochloric acid and zinc chloride. A related anion is , in which again Zn(II) adopts a tetrahedral geometry. References {{Reflist Tetrachlorozincates, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electronegativity
Electronegativity, symbolized as , is the tendency for an atom of a given chemical element to attract shared electrons (or electron density) when forming a chemical bond. An atom's electronegativity is affected by both its atomic number and the distance at which its valence electrons reside from the charged nucleus. The higher the associated electronegativity, the more an atom or a substituent group attracts electrons. Electronegativity serves as a simple way to quantitatively estimate the bond energy, and the sign and magnitude of a bond's chemical polarity, which characterizes a bond along the continuous scale from covalent to ionic bonding. The loosely defined term electropositivity is the opposite of electronegativity: it characterizes an element's tendency to donate valence electrons. On the most basic level, electronegativity is determined by factors like the nuclear charge (the more protons an atom has, the more "pull" it will have on electrons) and the number and lo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Raman Studies
Raman spectroscopy () (named after physicist C. V. Raman) is a spectroscopic technique typically used to determine vibrational modes of molecules, although rotational and other low-frequency modes of systems may also be observed. Raman spectroscopy is commonly used in chemistry to provide a structural fingerprint by which molecules can be identified. Raman spectroscopy relies upon inelastic scattering of photons, known as Raman scattering. A source of monochromatic light, usually from a laser in the visible, near infrared, or near ultraviolet range is used, although X-rays can also be used. The laser light interacts with molecular vibrations, phonons or other excitations in the system, resulting in the energy of the laser photons being shifted up or down. The shift in energy gives information about the vibrational modes in the system. Time-resolved spectroscopy and infrared spectroscopy typically yields similar yet complementary information. Typically, a sample is illuminated ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Zinc Hydroxide
Zinc hydroxide Zn( OH)2 is an inorganic chemical compound. It also occurs naturally as 3 rare minerals: wülfingite (orthorhombic), ashoverite and sweetite (both tetragonal). Like the hydroxides of other metals, such as lead, aluminium, beryllium, tin and chromium, Zinc hydroxide (and Zinc oxide), is amphoteric. Thus it will dissolve readily in a dilute solution of a strong acid, such as HCl, and also in a solution of an alkali such as sodium hydroxide. Preparation It can be prepared by first dissolving zinc oxide in concentrated aqueous solution of sodium hydroxide. The resulting solution is strongly diluted. :Zn2+ + 2 OH− → Zn(OH)2. The initial colorless solution contains the zincate ion: :Zn(OH)2 + 2 OH− → Zn(OH)42−. Zinc hydroxide will dissolve because the ion is normally surrounded by water ligands; when excess sodium hydroxide is added to the solution the hydroxide ions will reduce the complex to a −2 charge and make it soluble. When excess ammonia i ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Base (chemistry)
In chemistry, there are three definitions in common use of the word "base": '' Arrhenius bases'', '' Brønsted bases'', and '' Lewis bases''. All definitions agree that bases are substances that react with acid An acid is a molecule or ion capable of either donating a proton (i.e. Hydron, hydrogen cation, H+), known as a Brønsted–Lowry acid–base theory, Brønsted–Lowry acid, or forming a covalent bond with an electron pair, known as a Lewis ...s, as originally proposed by Guillaume-François Rouelle, G.-F. Rouelle in the mid-18th century. In 1884, Svante Arrhenius proposed that a base is a substance which dissociates in aqueous solution to form hydroxide ions OH−. These ions can react with Hydron (chemistry), hydrogen ions (H+ according to Arrhenius) from the dissociation of acids to form water in an acid–base reaction. A base was therefore a metal hydroxide such as NaOH or Calcium hydroxide, Ca(OH)2. Such aqueous hydroxide solutions were also described by ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |