|
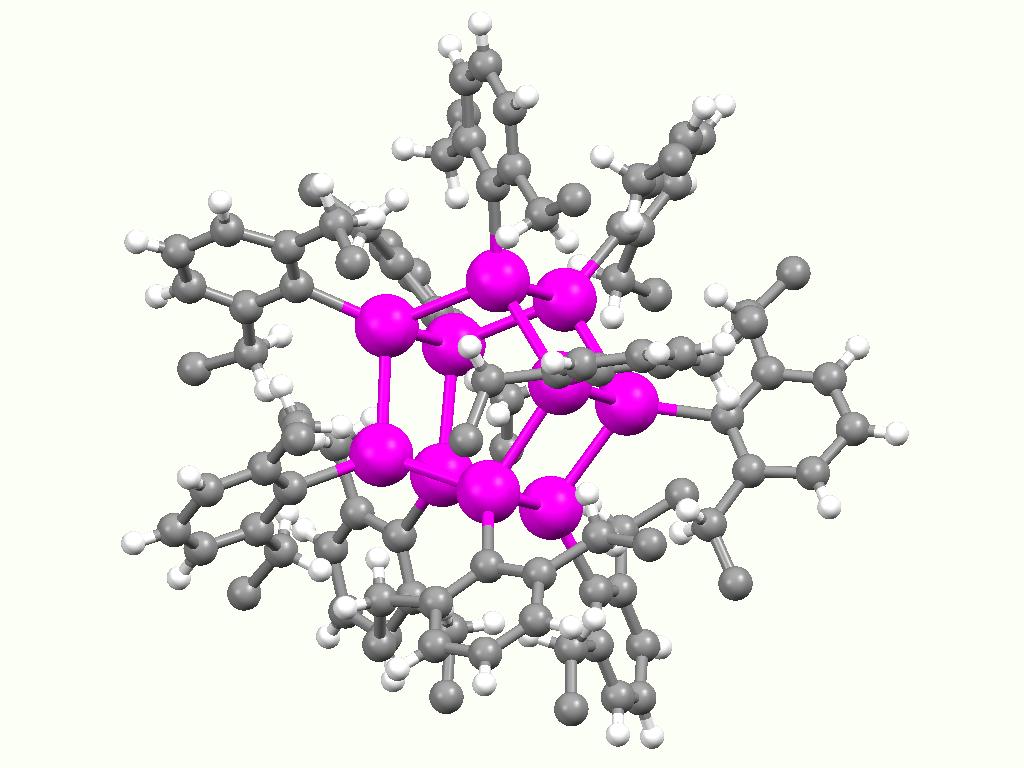
Stannylenes
Stannylenes (R2Sn:) are a class of organotin chemistry, organotin(II) compounds that are analogues of carbene. Unlike carbene, which usually has a triplet ground state, stannylenes have a singlet ground state since valence orbitals of tin (Sn) have less tendency to form hybrid orbitals and thus the electrons in 5s orbital are still paired up. Free stannylenes are stabilized by steric protection. Adducts with Lewis bases are also known. History The first persistent stannylene, [(Me3Si)2CH]2Sn, was reported by Michael F. Lappert in 1973. Lappert used the same synthetic approach to synthesize the first diamidostannylene [(Me3Si)2N]2Sn in 1974. The short-lived, transient stannylene Me2Sn has been generated by thermal decomposition, thermolysis of ''cyclo''-(Me2Sn)6. Synthesis and characterization Persistent stannylene Most alkyl stannylenes have been synthesized by alkylation of tin(II) dihalides with organolithium reagents. For example, the first stannylene, [(Me3Si)2CH]2Sn, was sy ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Stannylene Synthesis
Stannylenes (R2Sn:) are a class of organotin(II) compounds that are analogues of carbene. Unlike carbene, which usually has a triplet ground state, stannylenes have a singlet ground state since valence orbitals of tin (Sn) have less tendency to form hybrid orbitals and thus the electrons in 5s orbital are still paired up. Free stannylenes are stabilized by steric protection. Adducts with Lewis bases are also known. History The first persistent stannylene, Me3Si)2CHsub>2Sn, was reported by Michael F. Lappert in 1973. Lappert used the same synthetic approach to synthesize the first diamidostannylene Me3Si)2Nsub>2Sn in 1974. The short-lived, transient stannylene Me2Sn has been generated by thermolysis of ''cyclo''-(Me2Sn)6. Synthesis and characterization Persistent stannylene Most alkyl stannylenes have been synthesized by alkylation of tin(II) dihalides with organolithium reagents. For example, the first stannylene, Me3Si)2CHsub>2Sn, was synthesized using (Me3Si)2CHLi and SnCl ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tin(II) Compounds
Tin is a chemical element with the symbol Sn (from la, stannum) and atomic number 50. Tin is a silvery-coloured metal. Tin is soft enough to be cut with little force and a bar of tin can be bent by hand with little effort. When bent, the so-called "tin cry" can be heard as a result of twinning in tin crystals; this trait is shared by indium, cadmium, zinc, and mercury in the solid state. Pure tin after solidifying presents a mirror-like appearance similar to most metals. In most tin alloys (such as pewter) the metal solidifies with a dull gray color. Tin is a post-transition metal in group 14 of the periodic table of elements. It is obtained chiefly from the mineral cassiterite, which contains stannic oxide, . Tin shows a chemical similarity to both of its neighbors in group 14, germanium and lead, and has two main oxidation states, +2 and the slightly more stable +4. Tin is the 49th most abundant element on Earth and has, with 10 stable isotopes, the largest n ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Carbene Analogs
Carbene analogs in chemistry are carbenes with the carbon atom replaced by another chemical element. Just as regular carbenes they appear in chemical reactions as reactive intermediates and with special precautions they can be stabilized and isolated as chemical compounds. Carbenes have some practical utility in organic synthesis but carbene analogs are mostly laboratory curiosities only investigated in academia. Carbene analogs are known for elements of group 13 elements, group 13, group 14 elements, group 14, group 15 elements, group 15 and group 16 elements, group 16. Group 13 carbene analogs In group 13 elements the boron carbene analog is called a borylene or boranylidene. Group 14 carbene analogs The heavier group 14 element, group 14 carbenes are silylenes, R2Si:, germylenes R2Ge: (example diphosphagermylene), stannylenes R2Sn: and plumbylenes R2Pb:, collectively known as metallylenes and regarded as monomers for polymetallanes. The oxidation state for these compounds is +2 a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Michael F
Michael may refer to: People * Michael (given name), a given name * Michael (surname), including a list of people with the surname Michael Given name "Michael" * Michael (archangel), ''first'' of God's archangels in the Jewish, Christian and Islamic religions * Michael (bishop elect), English 13th-century Bishop of Hereford elect * Michael (Khoroshy) (1885–1977), cleric of the Ukrainian Orthodox Church of Canada * Michael Donnellan (1915–1985), Irish-born London fashion designer, often referred to simply as "Michael" * Michael (footballer, born 1982), Brazilian footballer * Michael (footballer, born 1983), Brazilian footballer * Michael (footballer, born 1993), Brazilian footballer * Michael (footballer, born February 1996), Brazilian footballer * Michael (footballer, born March 1996), Brazilian footballer * Michael (footballer, born 1999), Brazilian footballer Rulers =Byzantine emperors= * Michael I Rangabe (d. 844), married the daughter of Emperor Nikephor ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Oxidative Addition
Oxidative addition and reductive elimination are two important and related classes of reactions in organometallic chemistry. Oxidative addition is a process that increases both the oxidation state and coordination number of a metal centre. Oxidative addition is often a step in catalytic cycles, in conjunction with its reverse reaction, reductive elimination. Role in transition metal chemistry For transition metals, oxidative reaction results in the decrease in the d''n'' to a configuration with fewer electrons, often 2e fewer. Oxidative addition is favored for metals that are (i) basic and/or (ii) easily oxidized. Metals with a relatively low oxidation state often satisfy one of these requirements, but even high oxidation state metals undergo oxidative addition, as illustrated by the oxidation of Pt(II) with chlorine: : tCl4sup>2− + Cl2 → tCl6sup>2− In classical organometallic chemistry, the formal oxidation state of the metal and the electron count of the complex bot ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Organotin Compounds
Organotin compounds or stannanes are chemical compounds based on tin with hydrocarbon substituents. Organotin chemistry is part of the wider field of organometallic chemistry. The first organotin compound was diethyltin diiodide (), discovered by Edward Frankland in 1849. The area grew rapidly in the 1900s, especially after the discovery of the Grignard reagents, which are useful for producing Sn–C bonds. The area remains rich with many applications in industry and continuing activity in the research laboratory. Structure Organotin compounds are generally classified according to their oxidation states. Tin(IV) compounds are much more common and more useful. Organic derivatives of tin(IV) The tetraorgano derivatives are invariably tetrahedral. Compounds of the type SnRR'R''R have been resolved into individual enantiomers. Organotin halides Organotin chlorides have the formula for values of ''n'' up to 3. Bromides, iodides, and fluorides are also known but less important. T ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Silylene
Silylene is a chemical compound with the formula SiH2. It is the silicon analog of methylene, the simplest carbene. Silylene is a stable molecule as a gas but rapidly reacts in a bimolecular manner when condensed. Unlike carbenes, which can exist in the singlet or triplet state, silylene (and all of its derivatives) are singlets. Silylenes are formal derivatives of silylene with its hydrogens replaced by other substituents. Most examples feature amido (NR2) or alkyl/aryl groups. Silylenes have been proposed as reactive intermediates. They are carbene analogs. Synthesis and properties Silylenes are generally synthesized by thermolysis or photolysis of polysilanes, by silicon atom reactions ( insertion, addition or abstraction), by pyrolysis of silanes, or by reduction of 1,1-dihalosilane. It has long been assumed that the conversion of metallic Si to tetravalent silicon compounds proceeds via silylene intermediates: :Si + Cl2 → SiCl2 :SiCl2 + Cl2 → SiCl4 Similar considera ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |