|
N-acetylcysteine
''N''-acetylcysteine, also known as Acetylcysteine and NAC, is a mucolytics that is used to treat paracetamol (acetaminophen) overdose and to loosen thick mucus in individuals with chronic bronchopulmonary disorders, such as pneumonia and bronchitis. It has been used to treat lactobezoar in infants. It can be taken intravenously, orally (swallowed by mouth), or inhaled as a mist by use of a nebulizer. It is also sometimes used as a dietary supplement. Common side effects include nausea and vomiting when taken orally. The skin may occasionally become red and itchy with any route of administration. A non-immune type of anaphylaxis may also occur. It appears to be safe in pregnancy. For paracetamol overdose, it works by increasing the level of glutathione, an antioxidant that can neutralize the toxic breakdown products of paracetamol. When inhaled, it acts as a mucolytic by decreasing the thickness of mucus. Acetylcysteine was initially patented in 1960 and came into me ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Paracetamol Overdose
Paracetamol poisoning, also known as acetaminophen poisoning, is caused by excessive use of the medication paracetamol (acetaminophen). Most people have few or non-specific symptoms in the first 24 hours following overdose. These symptoms include feeling tired, abdominal pain, or nausea. This is typically followed by absence of symptoms for a couple of days, after which yellowish skin, blood clotting problems, and confusion occurs as a result of liver failure. Additional complications may include kidney failure, pancreatitis, low blood sugar, and lactic acidosis. If death does not occur, people tend to recover fully over a couple of weeks. Without treatment, death from toxicity occurs 4 to 18 days later. Paracetamol poisoning can occur accidentally or as an attempt to die by suicide. Risk factors for toxicity include alcoholism, malnutrition, and the taking of certain other hepatotoxic medications. Liver damage results not from paracetamol itself, but from one of its ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Acute Liver Failure
Acute liver failure is the appearance of severe complications rapidly after the first signs (such as jaundice) of liver disease, and indicates that the liver has sustained severe damage (loss of function of 80–90% of liver cells). The complications are hepatic encephalopathy and impaired protein synthesis (as measured by the levels of serum albumin and the prothrombin time in the blood). The 1993 classification defines ''hyperacute'' as within 1 week, ''acute'' as 8–28 days, and ''subacute'' as 4–12 weeks; both the speed with which the disease develops and the underlying cause strongly affect outcomes. Signs and symptoms The main features of acute liver failure are rapid-onset jaundice, weakness, and eventually, changes in mental status that can begin as mild confusion but progress to coma, known as hepatic encephalopathy. Encephalopathy and cerebral edema In ALF, hepatic encephalopathy leads to cerebral edema, coma, brain herniation, and eventually death. Detection of e ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Glutathione
Glutathione (GSH, ) is an organic compound with the chemical formula . It is an antioxidant in plants, animals, fungi, and some bacteria and archaea. Glutathione is capable of preventing damage to important cellular components caused by sources such as reactive oxygen species, free radicals, peroxides, lipid peroxides, and heavy metals. It is a tripeptide with a gamma peptide linkage between the carboxyl group of the glutamate side chain and cysteine. The carboxyl group of the cysteine residue is attached by normal peptide linkage to glycine. Biosynthesis and occurrence Glutathione biosynthesis involves two adenosine triphosphate-dependent steps: *First, γ-glutamylcysteine is synthesized from L-glutamate and L-cysteine. This conversion requires the enzyme glutamate–cysteine ligase (GCL, glutamate cysteine synthase). This reaction is the rate-limiting step in glutathione synthesis. *Second, glycine is added to the C-terminal of γ-glutamylcysteine. This condensation is ca ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
N-Acetylcarnosine
''N''-Acetylcarnosine (NAC) (not to be confused with N-Acetylcysteine, which is also abbreviated "NAC") is a naturally occurring Chemical compound, compound chemically related to the dipeptide carnosine. The NAC molecular structure is identical to carnosine with the exception that it carries an additional acetyl group. The acetylation makes NAC more resistant to degradation by carnosinase, an enzyme that breaks down carnosine to its constituent amino acids, beta-Alanine, beta-alanine and histidine. Actions Carnosine and metabolic derivatives of carnosine, including NAC, are found in a variety of tissues but particularly muscle tissue. These compounds have varying degrees of activity as free radical scavengers. It has been suggested that NAC is particularly active against lipid peroxidation in the different parts of the lens (anatomy), lens in the eye. It is an ingredient in eye drops that are marketed as a dietary supplement (not a drug) and have been promoted for the prevention a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Itch
An itch (also known as pruritus) is a sensation that causes a strong desire or reflex to scratch. Itches have resisted many attempts to be classified as any one type of sensory experience. Itches have many similarities to pain, and while both are unpleasant sensory experiences, their behavioral response patterns are different. Pain creates a withdrawal reflex, whereas itches leads to a scratch reflex. Unmyelinated nerve fibers for itches and pain both originate in the skin. Information for them is conveyed centrally in two distinct systems that both use the same nerve bundle and spinothalamic tract. Classification Most commonly, an itch is felt in one place. If it is felt all over the body, then it is called ''generalized itch'' or ''generalized pruritus''. Generalized itch is infrequently a symptom of a serious underlying condition, such as cholestatic liver disease. If the sensation of itching persists for six weeks or longer, then it is called ''chronic itch'' or ''c ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Bioavailability
In pharmacology, bioavailability is a subcategory of absorption and is the fraction (%) of an administered drug that reaches the systemic circulation. By definition, when a medication is administered intravenously, its bioavailability is 100%. However, when a medication is administered via routes other than intravenous, its bioavailability is lower due to intestinal epithelium absorption and first-pass metabolism. Thereby, mathematically, bioavailability equals the ratio of comparing the area under the plasma drug concentration curve versus time (AUC) for the extravascular formulation to the AUC for the intravascular formulation. AUC is used because AUC is proportional to the dose that has entered the systemic circulation. Bioavailability of a drug is an average value; to take population variability into account, deviation range is shown as ±. To ensure that the drug taker who has poor absorption is dosed appropriately, the bottom value of the deviation range is employed ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hepatocyte
A hepatocyte is a cell of the main parenchymal tissue of the liver. Hepatocytes make up 80% of the liver's mass. These cells are involved in: * Protein synthesis * Protein storage * Transformation of carbohydrates * Synthesis of cholesterol, bile salts and phospholipids * Detoxification, modification, and excretion of exogenous and endogenous substances * Initiation of formation and secretion of bile Structure The typical hepatocyte is cubical with sides of 20-30 μm, (in comparison, a human hair has a diameter of 17 to 180 μm).The diameter of human hair ranges from 17 to 181 μm. The typical volume of a hepatocyte is 3.4 x 10−9 cm3. Smooth endoplasmic reticulum is abundant in hepatocytes, in contrast to most other cell types. Microanatomy Hepatocytes display an eosinophilic cytoplasm, reflecting numerous mitochondria, and basophilic stippling due to large amounts of rough endoplasmic reticulum and free ribosomes. Brown lipofuscin granules are also observed (wit ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Xenobiotic Metabolism
Xenobiotic metabolism (from the Greek xenos (Greek), xenos "stranger" and biotic "related to living beings") is the set of metabolic pathways that modify the chemical structure of xenobiotics, which are compounds foreign to an organism's normal biochemistry, such as drugs and poisons. These pathways are a form of biotransformation present in all major groups of organisms, and are considered to be of ancient origin. These reactions often act to detoxification, detoxify poisonous compounds; however, in cases such as in the metabolism of Alcohol (drug), alcohol, the intermediates in xenobiotic metabolism can themselves be the cause of toxic effects. Xenobiotic metabolism is divided into three phases. In phase I, enzymes such as cytochrome P450 oxidases introduce reactive or polar groups into xenobiotics. These modified compounds are then conjugated to polar compounds in phase II reactions. These reactions are catalysed by transferase enzymes such as glutathione S-transferases. Fin ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
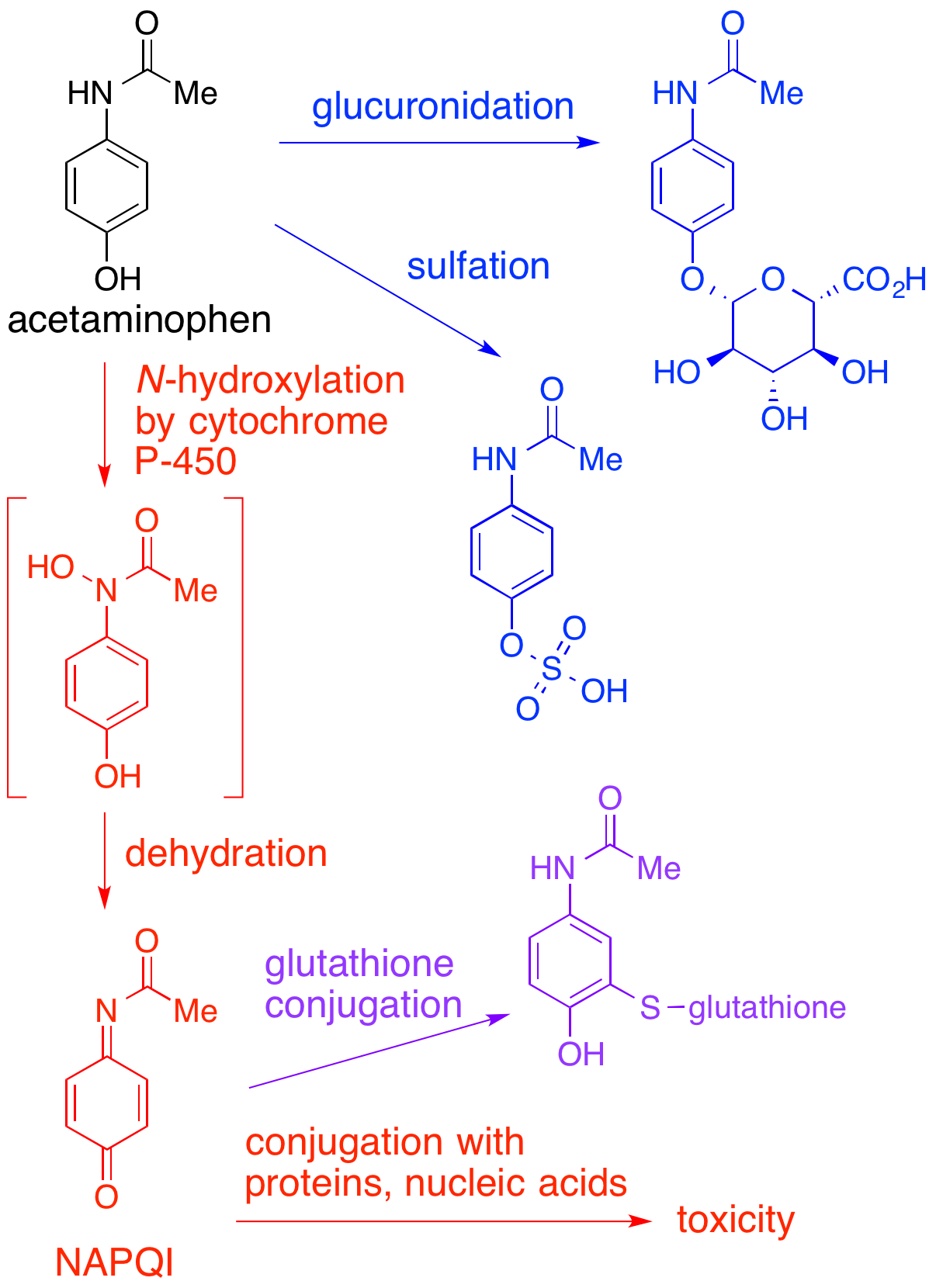
NAPQI
NAPQI, also known as NAPBQI or ''N''-acetyl-''p''-benzoquinone imine, is a toxic byproduct produced during the xenobiotic metabolism of the analgesic paracetamol (acetaminophen). It is normally produced only in small amounts, and then almost immediately detoxified in the liver. However, under some conditions in which NAPQI is not effectively detoxified (usually in the case of paracetamol overdose), it causes severe damage to the liver. This becomes apparent 3–4 days after ingestion and may result in death from fulminant liver failure several days after the overdose. Metabolism In adults, the primary metabolic pathway for paracetamol is glucuronidation. This yields a relatively non-toxic metabolite, which is excreted into bile and passed out of the body. A small amount of the drug is metabolized via the cytochrome P-450 pathway (to be specific, CYP3A4 and CYP2E1) into NAPQI, which is extremely toxic to liver tissue, as well as being a strong biochemical oxidizer. In an av ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Paracetamol
Paracetamol, or acetaminophen, is a non-opioid analgesic and antipyretic agent used to treat fever and mild to moderate pain. It is a widely available over-the-counter drug sold under various brand names, including Tylenol and Panadol. Paracetamol relieves pain in both acute mild migraine and episodic tension headache. At a standard dose, paracetamol slightly reduces fever, though it is inferior to ibuprofen in that respect and the benefits of its use for fever are unclear, particularly in the context of fever of viral origins. The aspirin/paracetamol/caffeine combination also helps with both conditions when the pain is mild and is recommended as a Therapy#Lines of therapy, first-line treatment for them. Paracetamol is effective for post-surgical pain, but is inferior to ibuprofen for this purpose. The paracetamol/ibuprofen combination increases the drugs' potency and is superior to either drug alone. The pain relief paracetamol provides in osteoarthritis is small and clinica ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Methionine
Methionine (symbol Met or M) () is an essential amino acid in humans. As the precursor of other non-essential amino acids such as cysteine and taurine, versatile compounds such as SAM-e, and the important antioxidant glutathione, methionine plays a critical role in the metabolism and health of many species, including humans. Methionine is also involved in angiogenesis and various processes related to DNA transcription, epigenetic expression, and gene regulation. Methionine was first isolated in 1921 by John Howard Mueller. It is Genetic code, encoded by the codon AUG. It was named by Satoru Odake in 1925, as an abbreviation of its structural description 2-amino-4-(methylthio)butanoic acid. Biochemical details Methionine (abbreviated as Met or M; encoded by the codon AUG) is an α-amino acid that is used in the biosynthesis of proteins. It contains a carboxyl group (which is in the deprotonated −COO− form under biological pH conditions), an amino group (which is in the proton ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cysteine
Cysteine (; symbol Cys or C) is a semiessential proteinogenic amino acid with the chemical formula, formula . The thiol side chain in cysteine enables the formation of Disulfide, disulfide bonds, and often participates in enzymatic reactions as a nucleophile. Cysteine is chiral, but both D and L-cysteine are found in nature. LCysteine is a protein monomer in all biota, and D-cysteine acts as a signaling molecule in mammalian nervous systems. Cysteine is named after its discovery in urine, which comes from the urinary bladder or cyst, from Ancient Greek, Greek κύστις ''kýstis'', "bladder". The thiol is susceptible to oxidation to give the disulfide bond, disulfide derivative cystine, which serves an important structural role in many proteins. In this case, the symbol Cyx is sometimes used. The deprotonated form can generally be described by the symbol Cym as well. When used as a food additive, cysteine has the E number E920. Cysteine is Genetic code, encoded by the codo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |