|
Tellurates
In chemistry, tellurate is a compound containing an oxyanion of tellurium where tellurium has an oxidation number of +6. In the naming of inorganic compounds it is a suffix that indicates a polyatomic ion, polyatomic anion with a central tellurium atom.Nomenclature of Inorganic Chemistry IUPAC Recommendations 2005 – Full text (PDF) Tellurium oxyanions Historically the name tellurate was only applied to oxyanions of tellurium with oxidation number +6, formally derived from telluric acid , and the name Tellurite (ion), tellurite referred to oxyanions of tellurium with oxidation number +4, formally derived from tellurous acid and these names are in common use. However tellurate and Tellurite (ion), tellurite are often referred to as tellurate(VI) ...[...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Telluric Acid
Telluric acid, or more accurately orthotelluric acid, is a chemical compound with the formula , often written as . It is a white crystalline solid made up of octahedral molecules which persist in aqueous solution. In the solid state, there are two forms, rhombohedral and monoclinic, and both contain octahedral molecules, containing one hexavalent tellurium (Te) atom in the +6 oxidation state, attached to six hydroxyl (–OH) groups, thus, it can be called tellurium(VI) hydroxide. Telluric acid is a weak acid which is dibasic, forming tellurate salts with strong bases and hydrogen tellurate salts with weaker bases or upon hydrolysis of tellurates in water.Holleman, A. F.; Wiberg, E. "Inorganic Chemistry" Academic Press: San Diego, 2001. . It is used as tellurium-source in the synthesis of oxidation catalysts. Preparation Telluric acid is formed by the oxidation of tellurium or tellurium dioxide with a powerful oxidising agent such as hydrogen peroxide, chromium trioxide or ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chalcogen Oxyanions
The chalcogens (ore forming) ( ) are the chemical elements in group 16 of the periodic table. This group is also known as the oxygen family. Group 16 consists of the elements oxygen (O), sulfur (S), selenium (Se), tellurium (Te), and the radioactive elements polonium (Po) and livermorium (Lv). Often, oxygen is treated separately from the other chalcogens, sometimes even excluded from the scope of the term "chalcogen" altogether, due to its very different chemical behavior from sulfur, selenium, tellurium, and polonium. The word "chalcogen" is derived from a combination of the Greek word () principally meaning copper (the term was also used for bronze, brass, any metal in the poetic sense, ore and coin), and the Latinized Greek word , meaning ''born'' or ''produced''. Sulfur has been known since antiquity, and oxygen was recognized as an element in the 18th century. Selenium, tellurium and polonium were discovered in the 19th century, and livermorium in 2000. All of the chalcog ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Polyatomic Ion
A polyatomic ion (also known as a molecular ion) is a covalent bonded set of two or more atoms, or of a metal complex, that can be considered to behave as a single unit and that usually has a net charge that is not zero, or in special case of zwitterion wear spatially separated charges where the net charge may be variable depending on acidity conditions. The term molecule may or may not be used to refer to a polyatomic ion, depending on the definition used. The prefix ''poly-'' carries the meaning "many" in Greek, but even ions of two atoms are commonly described as polyatomic. In older literature, a polyatomic ion may instead be referred to as a '' radical'' (or less commonly, as a ''radical group''). In contemporary usage, the term ''radical'' refers to various free radicals, which are species that have an unpaired electron and need not be charged. A simple example of a polyatomic ion is the hydroxide ion, which consists of one oxygen atom and one hydrogen ato ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Oxidation Number
In chemistry, the oxidation state, or oxidation number, is the hypothetical charge of an atom if all of its bonds to other atoms are fully ionic. It describes the degree of oxidation (loss of electrons) of an atom in a chemical compound. Conceptually, the oxidation state may be positive, negative or zero. Beside nearly-pure ionic bonding, many covalent bonds exhibit a strong ionicity, making oxidation state a useful predictor of charge. The oxidation state of an atom does not represent the "real" charge on that atom, or any other actual atomic property. This is particularly true of high oxidation states, where the ionization energy required to produce a multiply positive ion is far greater than the energies available in chemical reactions. Additionally, the oxidation states of atoms in a given compound may vary depending on the choice of electronegativity scale used in their calculation. Thus, the oxidation state of an atom in a compound is purely a formalism. It is nevertheles ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
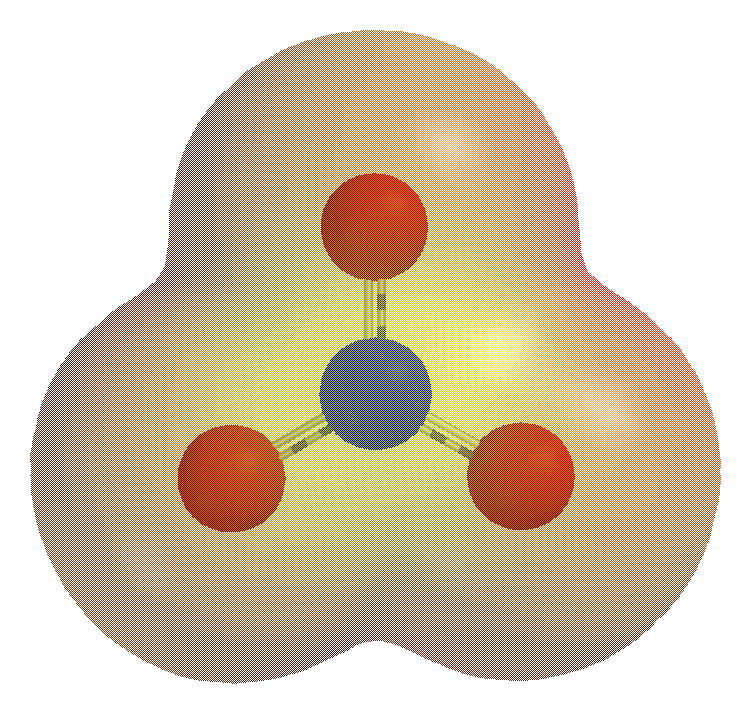
Tellurite (ion)
Tellurite is a oxyanion of tellurium with the formula . It is the ion of tellurous acid, and is chemically related to tellurium dioxide (), whose mineral appearance also bears the name tellurite. Tellurites are typically colorless or white salts, which in some ways are comparable to sulfite. Structure and reactions Tellurite dianion is pyramidal, like selenite and sulfite. The anion has C3v symmetry. Tellurites can be reduced to elemental tellurium by electrolysis or a strong reducing agent. When fused with nitrate salts, tellurite salts oxidize to tellurates (). Upon acidification of aqueous solutions of tellurite salts, solid hydrated tellurium dioxide (TeO2) precipitates. This reaction allows the separation of tellurium from selenium since selenous acid remains soluble at low pH. The intermediate in the protonation occurs at oxygen to give eO2(OH)sup>−. Compounds * Sodium tellurite * Potassium tellurite (K2TeO3) is used together with agar as part of a selecti ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tellurous Acid
Tellurous acid is an inorganic compound with the chemical formula, formula H2TeO3. It is the oxoacid of tellurium(IV). This compound is not well characterized. An alternative way of writing its formula is (HO)2TeO. In principle, tellurous acid would form by treatment of tellurium dioxide with water, that is by hydrolysis. The related conjugate base is well known in the form of several salts such as potassium hydrogen tellurite, KHTeO3. Properties In contrast to the analogous compound selenous acid, tellurous acid is only metastable. Most tellurite salts contain the ion. Oxidation of its aqueous solution with hydrogen peroxide gives the tellurate ion. It is usually prepared as an aqueous solution where it acts as a weak acid. :H2TeO3 + H2O H3O+ + ''K''a1 = : + H2O H3O+ + ''K''a2 = References Tellurites Chalcogen oxoacids {{inorganic-compound-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sulfate
The sulfate or sulphate ion is a polyatomic anion with the empirical formula . Salts, acid derivatives, and peroxides of sulfate are widely used in industry. Sulfates occur widely in everyday life. Sulfates are salts of sulfuric acid and many are prepared from that acid. Spelling "Sulfate" is the spelling recommended by IUPAC, but "sulphate" was traditionally used in British English. Structure The sulfate anion consists of a central sulfur atom surrounded by four equivalent oxygen atoms in a tetrahedral arrangement. The symmetry of the isolated anion is the same as that of methane. The sulfur atom is in the +6 oxidation state while the four oxygen atoms are each in the −2 state. The sulfate ion carries an overall charge of −2 and it is the conjugate base of the bisulfate (or hydrogensulfate) ion, , which is in turn the conjugate base of , sulfuric acid. Organic sulfate esters, such as dimethyl sulfate, are covalent compounds and esters of sulfuric acid. The tetrahedral ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Oxyanion
An oxyanion, or oxoanion, is an ion with the generic formula (where A represents a chemical element and O represents an oxygen atom). Oxyanions are formed by a large majority of the chemical elements. The formulae of simple oxyanions are determined by the octet rule. The corresponding oxyacid of an oxyanion is the compound . The structures of condensed oxyanions can be rationalized in terms of AO''n'' polyhedral units with sharing of corners or edges between polyhedra. The oxyanions (specifically, phosphate and polyphosphate esters) adenosine monophosphate ( AMP), adenosine diphosphate ( ADP) and adenosine triphosphate (ATP) are important in biology. Monomeric oxyanions The formula of monomeric oxyanions, , is dictated by the oxidation state of the element A and its position in the periodic table. Elements of the first row are limited to a maximum coordination number of 4. However, none of the first row elements has a monomeric oxyanion with that coordination number. Instead, c ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ethyl Group
In organic chemistry, an ethyl group (abbr. Et) is an alkyl substituent with the formula , derived from ethane (). ''Ethyl'' is used in the International Union of Pure and Applied Chemistry The International Union of Pure and Applied Chemistry (IUPAC ) is an international federation of National Adhering Organizations working for the advancement of the chemical sciences, especially by developing nomenclature and terminology. It is ...'s nomenclature of organic chemistry for a saturated two-carbon moiety in a molecule, while the prefix "''eth-''" is used to indicate the presence of two carbon atoms in the molecule. Ethylation Ethylation is the formation of a compound by introduction of the ethyl group. The most widely practiced example of this reaction is the ethylation of benzene with ethylene to yield ethylbenzene, a precursor to styrene, which is a precursor to polystyrene. Approximately 24.7 million tons of ethylbenzene were produced in 1999. :: Many ethyl ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Standard Reduction Potential
Redox potential (also known as oxidation / reduction potential, ''ORP'', ''pe'', ''E_'', or E_) is a measure of the tendency of a chemical species to acquire electrons from or lose electrons to an electrode and thereby be reduced or oxidised respectively. Redox potential is expressed in volts (V). Each species has its own intrinsic redox potential; for example, the more positive the reduction potential (reduction potential is more often used due to general formalism in electrochemistry), the greater the species' affinity for electrons and tendency to be reduced. Measurement and interpretation In aqueous solutions, redox potential is a measure of the tendency of the solution to either gain or lose electrons in a reaction. A solution with a higher (more positive) reduction potential than some other molecule will have a tendency to gain electrons from this molecule (i.e. to be reduced by oxidizing this other molecule) and a solution with a lower (more negative) reduction potential w ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |