|
Soil Fumigation
Fumigation is a method of pest control or the removal of harmful microorganisms by completely filling an area with gaseous pesticides, or fumigants, to asphyxia, suffocate or poison the pest (organism), pests within. It is used to control pests in buildings (structural fumigation), soil, grain, and produce. Fumigation is also used during the processing of goods for import or export to prevent the transfer of introduced species, exotic organisms. Structural fumigation targets pests inside buildings (usually residences), including pests that inhabit the physical structure itself, such as woodborers and drywood termites. Commodity fumigation, on the other hand, is also to be conducted inside a physical structure, such as a storage unit, but it aims to eliminate pests from infesting physical goods, usually food products, by killing pests within the container which will house them. Each fumigation lasts for a certain duration. This is because after spraying the pesticides, or fumig ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ethylene Oxide
Ethylene oxide is an organic compound with the chemical formula, formula . It is a cyclic ether and the simplest epoxide: a three-membered ring (chemistry), ring consisting of one oxygen atom and two carbon atoms. Ethylene oxide is a colorless and flammable gas with a faintly sweet odor. Because it is a strained ring, ethylene oxide easily participates in a number of addition reactions that result in ring-opening. Ethylene oxide is isomeric with acetaldehyde and with vinyl alcohol. Ethylene oxide is industrially produced by oxidation of ethylene in the presence of a silver catalyst. The reactivity that is responsible for many of ethylene oxide's hazards also makes it useful. Although too dangerous for direct household use and generally unfamiliar to consumers, ethylene oxide is used for making many consumer products as well as non-consumer chemicals and intermediates. These products include detergents, thickeners, solvents, plastics, and various organic chemicals such as ethylen ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Iodoform
Iodoform (also known as triiodomethane) is the organoiodine compound with the chemical formula . It is a pale yellow, crystalline, volatile substance, with a penetrating and distinctive odor (in older chemistry texts, the smell is sometimes referred to as that of hospitals, where the compound is still commonly used) and, analogous to chloroform, sweetish taste. It is occasionally used as a disinfectant. Naming The name iodoform originates with the "formyle radical," an archaic term for the HC moiety, and is retained for historical consistency. A full, modern name is triiodomethane. Another possible name is "carbon hydride triiodide". The "hydride" in the latter is sometimes omitted, though the IUPAC recommends against doing so, as "carbon triiodide" could also mean (hexaiodoethane, a highly unstable compound). Structure The molecule adopts a tetrahedral molecular geometry, tetrahedral geometry with C3v symmetry group, symmetry. Synthesis and reactions The synthesis of iodofo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrogen Cyanide
Hydrogen cyanide (formerly known as prussic acid) is a chemical compound with the chemical formula, formula HCN and structural formula . It is a highly toxic and flammable liquid that boiling, boils slightly above room temperature, at . HCN is produced on an industrial scale and is a highly valued Precursor (chemistry), precursor to many chemical compounds ranging from polymers to pharmaceuticals. Large-scale applications are for the production of potassium cyanide and adiponitrile, used in mining and plastics, respectively. It is more toxic than solid cyanide compounds due to its Volatility (chemistry), volatile nature. A solution of hydrogen cyanide in water (molecule), water, represented as HCN(aqueous, aq), is called ''hydrocyanic acid''. The Salt (chemistry), salts of the cyanide anion are known as cyanides. Whether hydrogen cyanide is an organic compound or not is a topic of debate among chemists, and opinions vary from author to author. Traditionally, it is considered ino ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
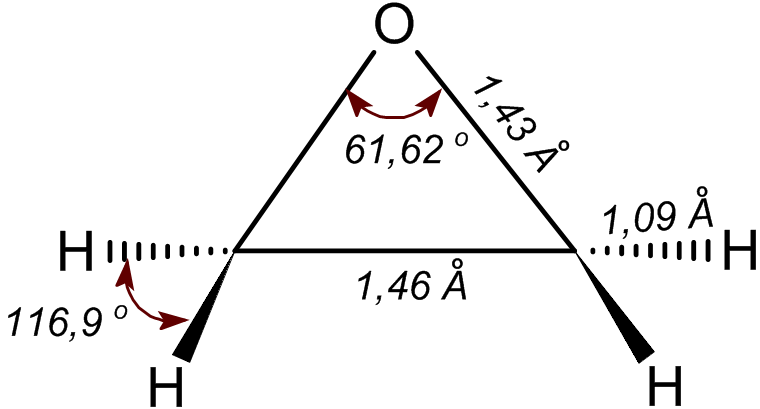
Formaldehyde
Formaldehyde ( , ) (systematic name methanal) is an organic compound with the chemical formula and structure , more precisely . The compound is a pungent, colourless gas that polymerises spontaneously into paraformaldehyde. It is stored as aqueous solutions (formalin), which consists mainly of the hydrate CH2(OH)2. It is the simplest of the aldehydes (). As a precursor to many other materials and chemical compounds, in 2006 the global production of formaldehyde was estimated at 12 million tons per year. It is mainly used in the production of industrial resins, e.g., for particle board and coatings. Formaldehyde also occurs naturally. It is derived from the degradation of serine, dimethylglycine, and lipids. Demethylases act by converting N-methyl groups to formaldehyde. Formaldehyde is classified as a group 1 carcinogen and can cause respiratory and skin irritation upon exposure. Forms Formaldehyde is more complicated than many simple carbon compounds in that i ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
DBCP
1,2-Dibromo-3-chloropropane (dibromochloropropane), better known as DBCP, is the organic compound with the formula BrCH(CHBr)(CHCl). It is a dense colorless liquid although commercial samples often appear amber or even brown. It is the active ingredient in the nematicide Nemagon, also known as Fumazone. It is a soil fumigant formerly used in American agriculture. In mammals, it causes male sterility at high levels of exposure. After discovery of its deleterious health effects on humans, the compound was banned from use in 1979 by the United States Environmental Protection Agency (EPA).Hazard Summary . - United States Environmental Protection Agency. - EPA.gov. The continuing presence of the chemical as a contaminant in ground water remains a problem for many communities for years after end of use. Stereoisomerism ...[...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Methyl Isothiocyanate
Methyl isothiocyanate is the organosulfur compound with the formula CH3N=C=S. This low melting colorless solid is a powerful lachrymator. As a precursor to a variety of valuable bioactive compounds, it is the most important organic isothiocyanate in industry. Synthesis It is prepared industrially by two routes. Annual production in 1993 was estimated to be 4,000 tonnes. The main method involves the thermal rearrangement of methyl thiocyanate: :CH3S−C≡N → CH3N=C=S It is also prepared via with the reaction of methylamine with carbon disulfide followed by oxidation of the resulting dithiocarbamate with hydrogen peroxide. A related method is useful to prepare this compound in the laboratory. MITC forms naturally upon the enzymatic degradation of glucocapparin, a glucoside found in capers. Reactions A characteristic reaction is with amines to give methyl thioureas: :CH3NCS + R2NH → R2NC(S)NHCH3 : Other nucleophiles add similarly. Applications Solutions of MITC ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dazomet
Dazomet is a common soil fumigant that acts as a herbicide, fungicide, slimicide, and nematicide. Applications Dazomet is a chemical used to kill pests that inhibit plant growth through gaseous degradation. Dazomet is used as a soil sterilant on a variety of sites such as golf courses, nurseries, turf sites, and potting soils. Dazomet is used for soil sterilization as an alternative to methyl bromide Bromomethane, commonly known as methyl bromide, is an organobromine compound with chemical formula, formula Carbon, CHydrogen, H3Bromine, Br. This colorless, odorless, nonflammable gas is Bromine cycle, produced both industrially and biologically .... Although less effective it is still used to kill pests because of its comparatively lower toxicity. Dazomet is applied to wet soil, which causes dazomet itself to decompose into a gaseous form, which is what actively controls pests. The decomposition of dazomet releases methyl isothiocyanate (MITC) a gas toxic to pests that would ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Methallyl Chloride
Methallyl chloride is the organic compound with the formula CH2=C(CH3)CH2Cl. It is a colorless liquid and a lacrymator. Its properties are similar to those of allyl chloride. It is a strong alkylating agent used to install isobutenyl groups. Reactivity It is also a precursor to meth allyl ligand. It is an isomer of crotyl chloride. Methylenecyclopropane can be synthesised via an intramolecular cyclisation reaction from methallyl chloride by treatment with a strong base such as sodium amide Sodium amide, commonly called sodamide (systematic name sodium azanide), is the inorganic compound with the formula . It is a salt composed of the sodium cation and the azanide anion. This solid, which is dangerously reactive toward water, is whit .... References {{Reflist Chloroalkenes IARC Group 3 carcinogens Allyl compounds ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ethylene Dibromide
1,2-Dibromoethane, also known as ethylene dibromide (EDB), is an organobromine compound with the chemical formula . Although trace amounts occur naturally in the ocean, where it is probably formed by algae and kelp, substantial amounts are produced industrially. It is a dense colorless liquid with a faint, sweet odor, detectable at 10 ppm. It is a widely used and sometimes-controversial fumigant. The combustion of 1,2-dibromoethane produces hydrogen bromide gas that is significantly corrosive. Preparation and use It is produced by the reaction of ethylene gas with bromine, in a classic halogen addition reaction: :CH=CH + Br → BrCH–CHBr Historically, 1,2-dibromoethane was used as a component in anti-knock additives in leaded fuels. It reacts with lead residues to generate volatile lead bromides, thereby preventing fouling of the engine with lead deposits. Pesticide It has been used as a pesticide in soil and on various crops. The applications were initiated after the forced re ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
1,3-dichloropropene
1,3-Dichloropropene, sold under diverse trade names, is an organochlorine compound with the formula . It is a colorless liquid with a sweet smell. It is feebly soluble in water and evaporates easily. It is used mainly in farming as a pesticide, specifically as a preplant fumigant and nematicide. It acts non-specifically and is in IRAC class 8A. It is widely used in the US and other countries, but is banned in 34 countries (including the European Union). Production, chemical properties, biodegradation It is a byproduct in the chlorination of propene to make allyl chloride. It is usually obtained as a mixture of the geometric isomers, called (''Z'')-1,3-dichloropropene, and (''E'')-1,3-dichloropropene. Although it was first applied in agriculture in the 1950s, at least two biodegradation pathways have evolved. One pathway degrades the chlorocarbon to acetaldehyde via chloroacrylic acid. Safety The TLV- TWA for 1,3-dichloropropene (DCP) is 1 ppm.Robert L. Metcalf "Insect Con ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phosphine
Phosphine (IUPAC name: phosphane) is a colorless, flammable, highly toxic compound with the chemical formula , classed as a pnictogen hydride. Pure phosphine is odorless, but technical grade samples have a highly unpleasant odor like rotting fish, due to the presence of substituted phosphine and diphosphane (). With traces of present, is spontaneously flammable in air ( pyrophoric), burning with a luminous flame. Phosphine is a highly toxic respiratory poison, and is immediately dangerous to life or health at 50 ppm. Phosphine has a trigonal pyramidal structure. Phosphines are compounds that include and the organophosphines, which are derived from by substituting one or more hydrogen atoms with organic groups. They have the general formula . Phosphanes are saturated phosphorus hydrides of the form , such as triphosphane. Phosphine () is the smallest of the phosphines and the smallest of the phosphanes. History Philippe Gengembre (1764–1838), a student of Lavoisi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |