|
Shpolskii Matrix
Shpolskii systems are low-temperature host–guest systems – they are typically rapidly frozen solutions of polycyclic aromatic hydrocarbons in suitable low molecular weight normal alkanes. The emission and absorption spectra of lowest energy electronic transitions in the Shpolskii systems exhibit narrow lines instead of the inhomogeneously broadened features normally associated with spectra of chromophores in liquids and amorphous solids. The effect was first described by Eduard Shpolskii in the 1950s and 1960's in the journals ''Transactions of the U.S.S.R. Academy of Sciences'' and ''Soviet Physics Uspekhi''. Subsequent detailed studies of concentration and speed of cooling behavior of Shpolskii systems by L. A. Nakhimovsky and coauthors led to a hypothesis that these systems are ''metastable segregational'' solid solutions formed when one or ''more'' chromophores replace two or more molecules in the host crystalline lattice. The solid state quasi-equilibrium solubility i ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Host–guest Chemistry
In supramolecular chemistry, host–guest chemistry describes inclusion compound, complexes that are composed of two or more molecules or ions that are held together in unique structural relationships by forces other than those of full covalent bonds. Host–guest chemistry encompasses the idea of molecular recognition and interactions through non-covalent bonding. Non-covalent bonding is critical in maintaining the 3D structure of large molecules, such as proteins and is involved in many biological processes in which large molecules bind specifically but transiently to one another. Although non-covalent interactions could be roughly divided into those with more electrostatic or dispersive contributions, there are few commonly mentioned types of non-covalent interactions: ionic bonding, hydrogen bond, hydrogen bonding, van der Waals forces and hydrophobic effect, hydrophobic interactions. Overview Host–guest chemistry is a branch of supramolecular chemistry in which a host mo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Polycyclic Aromatic Hydrocarbon
A polycyclic aromatic hydrocarbon (PAH) is a class of organic compounds that is composed of multiple aromatic rings. The simplest representative is naphthalene, having two aromatic rings and the three-ring compounds anthracene and phenanthrene. PAHs are uncharged, non-polar and planar. Many are colorless. Many of them are found in coal and in oil deposits, and are also produced by the combustion of organic matter—for example, in engines and incinerators or when biomass burns in forest fires. Polycyclic aromatic hydrocarbons are discussed as possible starting materials for abiotic syntheses of materials required by the earliest forms of life. Nomenclature and structure The terms polyaromatic hydrocarbon or polynuclear aromatic hydrocarbon are also used for this concept. By definition, polycyclic aromatic hydrocarbons have multiple rings, precluding benzene from being considered a PAH. Some sources, such as the US EPA and CDC, consider naphthalene to be the simplest PA ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
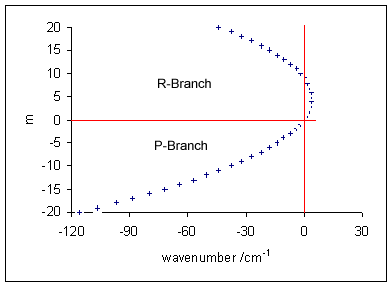
Molecular Electronic Transition
Molecular electronic transitions take place when electrons in a molecule are excited from one energy level to a higher energy level. The energy change associated with this transition provides information on the structure of a molecule and determines many molecular properties such as colour. The relationship between the energy involved in the electronic transition and the frequency of radiation is given by Planck's relation. Organic molecules and other molecules The electronic transitions in organic compounds and some other compounds can be determined by ultraviolet–visible spectroscopy, provided that transitions in the ultraviolet (UV) or visible range of the electromagnetic spectrum exist for this compound. Electrons occupying a HOMO of a sigma bond can get excited to the LUMO of that bond. This process is denoted as a σ → σ* transition. Likewise promotion of an electron from a π-bonding orbital to an antibonding π orbital* is denoted as a π → π* transition. A ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chromophore
A chromophore is the part of a molecule responsible for its color. The color that is seen by our eyes is the one not absorbed by the reflecting object within a certain wavelength spectrum of visible light. The chromophore is a region in the molecule where the energy difference between two separate molecular orbitals falls within the range of the visible spectrum. Visible light that hits the chromophore can thus be absorbed by exciting an electron from its ground state into an excited state. In biological molecules that serve to capture or detect light energy, the chromophore is the moiety that causes a conformational change in the molecule when hit by light. Conjugated pi-bond system chromophores Just like how two adjacent p-orbitals in a molecule will form a pi-bond, three or more adjacent p-orbitals in a molecule can form a conjugated pi-system. In a conjugated pi-system, electrons are able to capture certain photons as the electrons resonate along a certain distanc ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Eduard Shpolsky
Eduard Vladimirovich Shpolsky, also Shpolsk'ii, Shpolskii (russian: Эдуард Владимирович Шпольский, born September 23, 1892 in Voronezh – died August 21, 1975 in Moscow) was a Russian and Soviet physicist and educator, co-founder and lifelong editor of '' Uspekhi Fizicheskikh Nauk'' journal (''Soviet Physics Uspekhi'' and ''Physics-Uspekhi'' in English translation). Shpolsky primary scientific contribution belongs to the field of molecular spectroscopy, particularly luminescence and absorption spectra of polycyclic aromatic hydrocarbons. In 1952 Shpolsky and his junior researchers A. A. Ilyina and L. A. Klimov discovered Shpolsky effect (Shpolskii matrixes, an optical analogy to Mössbauer effect) in organic compounds, a property that allows highly selective spectroscopic identification of substances that normally do not possess clearly defined spectral lines or bands. The discovery evolved into a discipline of its own, ''Shpolsky spectroscopy' ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Vibronic Spectroscopy
Vibronic spectroscopy is a branch of molecular spectroscopy concerned with vibronic transitions: the simultaneous changes in electronic and vibrational energy levels of a molecule due to the absorption or emission of a photon of the appropriate energy. In the gas phase, vibronic transitions are accompanied by changes in rotational energy also. Vibronic spectra of diatomic molecules have been analysed in detail; emission spectra are more complicated than absorption spectra. The intensity of allowed vibronic transitions is governed by the Franck–Condon principle. Vibronic spectroscopy may provide information, such as bond length, on electronic excited states of stable molecules. It has also been applied to the study of unstable molecules such as dicarbon, C2, in discharges, flames and astronomical objects.Hollas, p. 211. Principles Electronic transitions are typically observed in the visible and ultraviolet regions, in the wavelength range approximately 200–700& ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phonon
In physics, a phonon is a collective excitation in a periodic, elastic arrangement of atoms or molecules in condensed matter, specifically in solids and some liquids. A type of quasiparticle, a phonon is an excited state in the quantum mechanical quantization of the modes of vibrations for elastic structures of interacting particles. Phonons can be thought of as quantized sound waves, similar to photons as quantized light waves. The study of phonons is an important part of condensed matter physics. They play a major role in many of the physical properties of condensed matter systems, such as thermal conductivity and electrical conductivity, as well as in models of neutron scattering and related effects. The concept of phonons was introduced in 1932 by Soviet physicist Igor Tamm. The name ''phonon'' comes from the Greek word (), which translates to ''sound'' or ''voice'', because long-wavelength phonons give rise to sound. The name is analogous to the word ''photon''. Definiti ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Zero-phonon Line And Phonon Sideband
The zero-phonon line and the phonon sideband jointly constitute the line shape of individual light absorbing and emitting molecules (chromophores) embedded into a transparent solid matrix. When the host matrix contains many chromophores, each will contribute a zero-phonon line and a phonon sideband to the absorption and emission spectra. The spectra originating from a collection of identical chromophores in a matrix is said to be inhomogeneously broadened because each chromophore is surrounded by a somewhat different matrix environment which modifies the energy required for an electronic transition. In an inhomogeneous distribution of chromophores, individual zero-phonon line and phonon sideband positions are therefore shifted and overlapping. Figure 1 shows the typical line shape for electronic transitions of individual chromophores in a solid matrix. The zero-phonon line is located at a frequency ω’ determined by the intrinsic difference in energy levels between ground and ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Liquid Nitrogen
Liquid nitrogen—LN2—is nitrogen in a liquid state at low temperature. Liquid nitrogen has a boiling point of about . It is produced industrially by fractional distillation of liquid air. It is a colorless, low viscosity liquid that is widely used as a coolant. Physical properties The diatomic character of the N2 molecule is retained after liquefaction. The weak van der Waals interaction between the N2 molecules results in little interatomic interaction, manifested in its very low boiling point. The temperature of liquid nitrogen can readily be reduced to its freezing point by placing it in a vacuum chamber pumped by a vacuum pump. Liquid nitrogen's efficiency as a coolant is limited by the fact that it boils immediately on contact with a warmer object, enveloping the object in an insulating layer of nitrogen gas bubbles. This effect, known as the Leidenfrost effect, occurs when any liquid comes in contact with a surface which is significantly hotter than its b ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Kelvins
The kelvin, symbol K, is the primary unit of temperature in the International System of Units (SI), used alongside its prefixed forms and the degree Celsius. It is named after the Belfast-born and University of Glasgow-based engineer and physicist William Thomson, 1st Baron Kelvin (1824–1907). The Kelvin scale is an absolute thermodynamic temperature scale, meaning it uses absolute zero as its null (zero) point. Historically, the Kelvin scale was developed by shifting the starting point of the much-older Celsius scale down from the melting point of water to absolute zero, and its increments still closely approximate the historic definition of a degree Celsius, but since 2019 the scale has been defined by fixing the Boltzmann constant to be exactly . Hence, one kelvin is equal to a change in the thermodynamic temperature that results in a change of thermal energy by . The temperature in degree Celsius is now defined as the temperature in kelvins minus 273.15, meaning t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |

.jpeg/1200px-Fall_Leaves_(199582361).jpeg)