|
Radiolysis
Radiolysis is the dissociation of molecules by ionizing radiation. It is the cleavage of one or several chemical bonds resulting from exposure to high-energy flux. The radiation in this context is associated with ionizing radiation; radiolysis is therefore distinguished from, for example, photolysis of the Cl2 molecule into two Cl- radicals, where (ultraviolet or visible spectrum) light is used. The chemistry of concentrated solutions under ionizing radiation is extremely complex. Radiolysis can locally modify redox conditions, and therefore the speciation and the solubility of the compounds. Water decomposition Of all the radiation-based chemical reactions that have been studied, the most important is the decomposition of water. When exposed to radiation, water undergoes a breakdown sequence into hydrogen peroxide, hydrogen radicals, and assorted oxygen compounds, such as ozone, which when converted back into oxygen releases great amounts of energy. Some of these are explosive ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
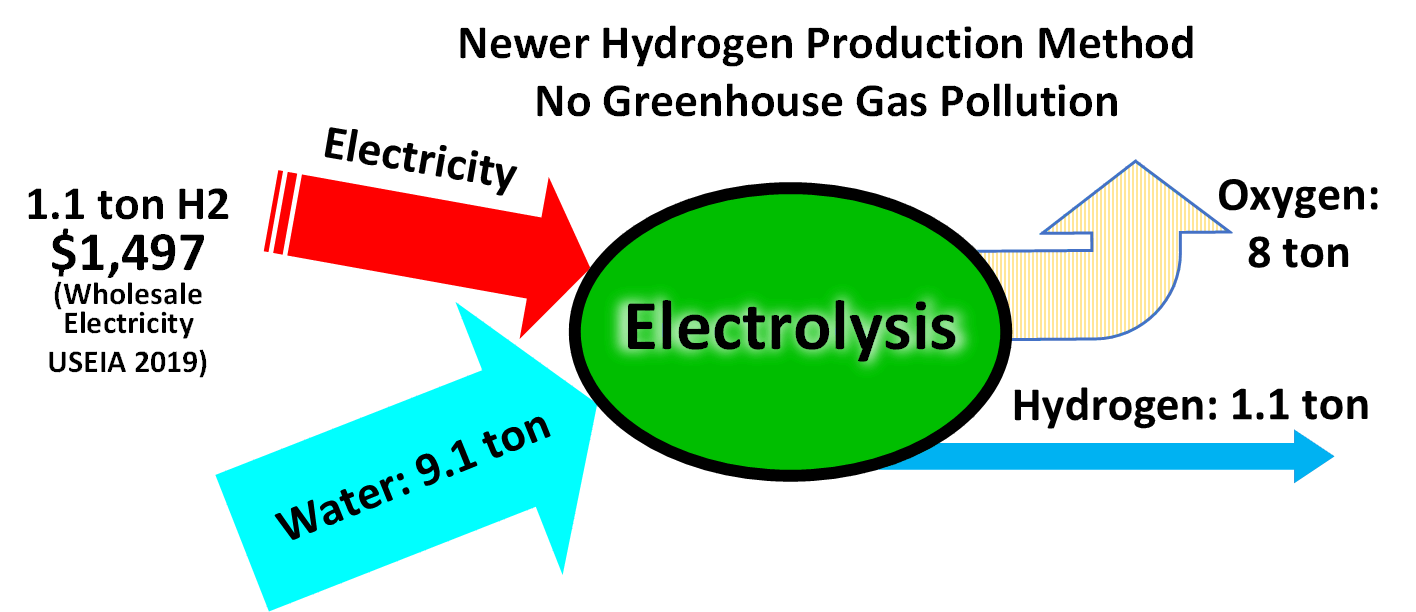
Hydrogen Production
Hydrogen gas is produced by several industrial methods. Nearly all of the world's current supply of hydrogen is created from fossil fuels. Article in press. Most hydrogen is ''gray hydrogen'' made through steam methane reforming. In this process, hydrogen is produced from a chemical reaction between steam and methane, the main component of natural gas. Producing one tonne of hydrogen through this process emits 6.6–9.3 tonnes of carbon dioxide. When carbon capture and storage is used to remove a large fraction of these emissions, the product is known as ''blue hydrogen''. ''Green hydrogen'' is usually understood to be produced from Renewable energy, renewable electricity via electrolysis of water. Less frequently, definitions of ''green hydrogen'' include hydrogen produced from other low-emission sources such as Biomass (energy), biomass. Producing green hydrogen is currently more expensive than producing gray hydrogen, and the efficiency of energy conversion is inherently low. O ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dissociation (chemistry)
Dissociation in chemistry is a general process in which molecules (or ionic compounds such as salt (chemistry), salts, or coordination complex, complexes) separate or split into other things such as atoms, ions, or radical (chemistry), radicals, usually in a reversible manner. For instance, when an acid dissolves in water, a covalent bond between an electronegativity, electronegative atom and a hydrogen atom is broken by heterolytic fission, which gives a proton (H+) and a negative ion. Dissociation is the opposite of association or recombination. Dissociation constant For reversible dissociations in a chemical equilibrium :AB A + B the dissociation constant ''K''d is the ratio of dissociated to undissociated compound :K_d = \mathrm where the brackets denote the equilibrium concentrations of the species. Dissociation degree The dissociation degree \alpha is the fraction of original solute molecules that have dissociated. It is usually indicated by the Greek symbol α. More acc ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrogen Peroxide
Hydrogen peroxide is a chemical compound with the formula . In its pure form, it is a very pale blue liquid that is slightly more viscosity, viscous than Properties of water, water. It is used as an oxidizer, bleaching agent, and antiseptic, usually as a dilute solution (3%–6% by weight) in water for consumer use and in higher concentrations for industrial use. Concentrated hydrogen peroxide, or "high-test peroxide", decomposes explosively when heated and has been used as both a monopropellant and an oxidizer in rocketry. Hydrogen peroxide is a reactive oxygen species and the simplest peroxide, a compound having an oxygen–oxygen single bond. It decomposes slowly into water and elemental oxygen when exposed to light, and rapidly in the presence of organic or reactive compounds. It is typically stored with a Stabilizer (chemistry), stabilizer in a weakly acidic solution in an opaque bottle. Hydrogen peroxide is found in biological systems including the human body. Enzymes that u ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sodium Borohydride
Sodium borohydride, also known as sodium tetrahydridoborate and sodium tetrahydroborate, is an inorganic compound with the formula (sometimes written as ). It is a white crystalline solid, usually encountered as an aqueous basic solution. Sodium borohydride is a reducing agent that finds application in papermaking and dye industries. It is also used as a reagent in organic synthesis. The compound was discovered in the 1940s by H. I. Schlesinger, who led a team seeking volatile uranium compounds.Hermann I Schlesinger and Herbert C Brown (1945)Preparation of alkali metal compounds. US Patent 2461661. Granted on 1949-02-15; expired on 1966-02-15. Results of this wartime research were declassified and published in 1953. Properties The compound is soluble in alcohols, certain ethers, and water, although it slowly hydrolyzes. Sodium borohydride is an odorless white to gray-white microcrystalline powder that often forms lumps. It can be purified by recrystallization from warm ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sodium Borate
Sodium borate is a generic name for any salt (chemistry), salt of sodium with an anion consisting of boron and oxygen, and possibly hydrogen, or any hydrate thereof. It can be seen as a hydrated sodium salt of the appropriate boroxy acid, although the latter may not be a stable compound. Many sodium borates have important industrial and household applications; the best known being borax, = . The ternary phase diagram of the –– phase diagram in the 0–100 °C temperature range contains 13 unique hydrated crystalline sodium borates, including five important industrial products. Sodium borates, as well as boroxy acids, are often described as mixtures = , with ''x'', ''y'', and ''z'' chosen to fit the elemental formula, or a multiple thereof. Thus, for example, borax would be , and boric acid would be = . The elemental formula was often interpreted as a ''z''-hydrate of an "anhydrous" salt without any hydrogen, namely . However, later research uncovered that many bora ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Radioactive Waste
Radioactive waste is a type of hazardous waste that contains radioactive material. It is a result of many activities, including nuclear medicine, nuclear research, nuclear power generation, nuclear decommissioning, rare-earth mining, and nuclear weapons reprocessing. The storage and disposal of radioactive waste is regulated by government agencies in order to protect human health and the environment. Radioactive waste is broadly classified into 3 categories: low-level waste (LLW), such as paper, rags, tools, clothing, which contain small amounts of mostly short-lived radioactivity; intermediate-level waste (ILW), which contains higher amounts of radioactivity and requires some shielding; and high-level waste (HLW), which is highly radioactive and hot due to decay heat, thus requiring cooling and shielding. Spent nuclear fuel can be processed in nuclear reprocessing plants. One third of the total amount have already been reprocessed. With nuclear reprocessing 96% of the spent fue ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electronvolt
In physics, an electronvolt (symbol eV), also written electron-volt and electron volt, is the measure of an amount of kinetic energy gained by a single electron accelerating through an Voltage, electric potential difference of one volt in vacuum. When used as a Units of energy, unit of energy, the numerical value of 1 eV in joules (symbol J) is equal to the numerical value of the Electric charge, charge of an electron in coulombs (symbol C). Under the 2019 revision of the SI, this sets 1 eV equal to the exact value Historically, the electronvolt was devised as a standard unit of measure through its usefulness in Particle accelerator#Electrostatic particle accelerators, electrostatic particle accelerator sciences, because a particle with electric charge ''q'' gains an energy after passing through a voltage of ''V''. Definition and use An electronvolt is the amount of energy gained or lost by a single electron when it moves through an Voltage, electric potential differenc ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nuclear Reprocessing
Nuclear reprocessing is the chemical separation of fission products and actinides from spent nuclear fuel. Originally, reprocessing was used solely to extract plutonium for producing nuclear weapons. With commercialization of nuclear power, the reprocessed plutonium was recycled back into MOX nuclear fuel for thermal reactors. The reprocessed uranium, also known as the spent fuel material, can in principle also be re-used as fuel, but that is only economical when uranium supply is low and prices are high. Nuclear reprocessing may extend beyond fuel and include the reprocessing of other nuclear reactor material, such as Zircaloy cladding. The high radioactivity of spent nuclear material means that reprocessing must be highly controlled and carefully executed in advanced facilities by specialized personnel. Numerous processes exist, with the chemical based PUREX process dominating. Alternatives include heating to drive off volatile elements, burning via oxidation, and fluoride vola ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Spent Nuclear Fuel
Spent nuclear fuel, occasionally called used nuclear fuel, is nuclear fuel that has been irradiated in a nuclear reactor (usually at a nuclear power plant). It is no longer useful in sustaining a nuclear reaction in an ordinary thermal reactor and, depending on its point along the nuclear fuel cycle, it will have different isotopic constituents than when it started. Nuclear fuel rods become progressively more radioactive (and less thermally useful) due to neutron activation as they are fissioned, or "burnt", in the reactor. A fresh rod of low enriched uranium pellets (which can be safely handled with gloved hands) will become a highly lethal gamma emitter after 1–2 years of core irradiation, unsafe to approach unless under many feet of water shielding. This makes their invariable accumulation and safe temporary storage in spent fuel pools a prime source of high level radioactive waste and a major ongoing issue for future permanent disposal. Nature of spent fuel Nanomaterial pro ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nuclear Reactor
A nuclear reactor is a device used to initiate and control a Nuclear fission, fission nuclear chain reaction. They are used for Nuclear power, commercial electricity, nuclear marine propulsion, marine propulsion, Weapons-grade plutonium, weapons production and Research reactor, research. Fissile material, Fissile nuclei (primarily uranium-235 or plutonium-239) absorb single neutron, neutrons and split, releasing energy and multiple neutrons, which can induce further fission. Reactors stabilize this, regulating Neutron absorber, neutron absorbers and neutron moderator, moderators in the core. Fuel efficiency is exceptionally high; Enriched uranium#Low-enriched uranium (LEU), low-enriched uranium is 120,000 times more energy dense than coal. Heat from nuclear fission is passed to a working fluid Nuclear reactor#By coolant, coolant. In commercial reactors, this drives Turbine, turbines and electrical generator shafts. Some reactors are used for district heating, and isotopes, isoto ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Corrosion
Corrosion is a natural process that converts a refined metal into a more chemically stable oxide. It is the gradual deterioration of materials (usually a metal) by chemical or electrochemical reaction with their environment. Corrosion engineering is the field dedicated to controlling and preventing corrosion. In the most common use of the word, this means electrochemical oxidation of metal in reaction with an oxidant such as oxygen, hydrogen, or hydroxide. Rusting, the formation of red-orange iron oxides, is a well-known example of electrochemical corrosion. This type of corrosion typically produces oxides or salts of the original metal and results in a distinctive coloration. Corrosion can also occur in materials other than metals, such as ceramics or polymers, although in this context, the term "degradation" is more common. Corrosion degrades the useful properties of materials and structures including mechanical strength, appearance, and permeability to liquids and ga ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Light-water Reactor
The light-water reactor (LWR) is a type of thermal-neutron reactor that uses normal water, as opposed to heavy water, as both its coolant and neutron moderator; furthermore a solid form of fissile elements is used as fuel. Thermal-neutron reactors are the most common type of nuclear reactor, and light-water reactors are the most common type of thermal-neutron reactor. There are three varieties of light-water reactors: the pressurized water reactor (PWR), the boiling water reactor (BWR), and (most designs of) the supercritical water reactor (SCWR). History Early concepts and experiments After the discoveries of fission, moderation and of the theoretical possibility of a nuclear chain reaction, early experimental results rapidly showed that natural uranium could only undergo a sustained chain reaction using graphite or heavy water as a moderator. While the world's first reactors ( CP-1, X10 etc.) were successfully reaching criticality, uranium enrichment began to develop ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |