|
RS-102,221
RS-102221 is a drug developed by Hoffmann–La Roche, which was one of the first compounds discovered that acts as a potent and selective Antagonist (pharmacology), antagonist at the serotonin 5-HT2C receptor, 5-HT2C Receptor (biochemistry), receptor, with around 100× selectivity over the closely related 5-HT2A receptor, 5-HT2A and 5-HT2B receptor, 5-HT2B receptors. It has anxiolytic effects in animal studies, increases the effectiveness of SSRI antidepressants, and shows a complex interaction with cocaine, increasing some effects but decreasing others, reflecting a role for the 5-HT2C receptor in regulation of the dopamine signalling system in the brain. See also * 6-Chloro-5-ethoxy-N-(pyridin-2-yl)indoline-1-carboxamide, CEPC * SB-242084 *Hydantoin References {{Serotonergics 5-HT2C antagonists Imides Ureas Lactams Nitrogen heterocycles Aromatic ketones Phenol ethers Trifluoromethyl compounds Spiro compounds Sulfonamides Hydantoins Heterocyclic compounds with 2 rings ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
5-HT2C Receptor
The 5-HT2C receptor is a subtype of the 5-HT2 receptor that binds the endogenous neurotransmitter serotonin (5-hydroxytryptamine, 5-HT). Like all 5-HT2 receptors, it is a G protein-coupled receptor (GPCR) that is coupled to Gq/G11 and mediates excitatory neurotransmission. ''HTR2C'' denotes the human gene encoding for the receptor, that in humans is located on the X chromosome. As males have one copy of the gene and females have one of the two copies of the gene repressed, polymorphisms at this receptor can affect the two sexes to differing extent. Structure At the cell surface the receptor exists as a homodimer. The crystal structure has been known since 2018. Distribution 5-HT2C receptors are located mainly in the choroid plexus, and in rats is also found in many other brain regions in high concentrations, including parts of the hippocampus, anterior olfactory nucleus, substantia nigra, several brainstem nuclei, amygdala, subthalamic nucleus and lateral habenula. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
6-Chloro-5-ethoxy-N-(pyridin-2-yl)indoline-1-carboxamide
6-Chloro-5-ethoxy-''N''-(pyridin-2-yl)indoline-1-carboxamide (CEPC) is a drug which acts as a potent and selective antagonist for the serotonin 5-HT2C receptor. In animal studies it was found to potentiate the conditioned place preference induced by low-dose amphetamine, demonstrating that 5-HT2C-mediated disinhibition of dopamine release can cause interactions with dopaminergic drugs. See also * RS-102,221 RS-102221 is a drug developed by Hoffmann–La Roche, which was one of the first compounds discovered that acts as a potent and selective Antagonist (pharmacology), antagonist at the serotonin 5-HT2C receptor, 5-HT2C Receptor (biochemistry), rece ... * SB-242,084 * SCQ1 References {{DEFAULTSORT:Chloro-5-ethoxy-''N''-(pyridin-2-yl)indoline-1-carboxamide, 6- 5-HT2C antagonists 2-Pyridyl compounds Chloroarenes Ureas Ethoxy compounds Indolines ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hoffmann–La Roche
F. Hoffmann-La Roche AG, commonly known as Roche (), is a Swiss multinational holding healthcare company that operates worldwide under two divisions: Pharmaceuticals and Diagnostics. Its holding company, Roche Holding AG, has shares listed on the SIX Swiss Exchange. The company headquarters are located in Basel. Roche is the fifth-largest pharmaceutical company in the world by revenue and the leading provider of cancer treatments globally. In 2023, the company’s seat in Forbes Global 2000 was 76. The company owns the American biotechnology company Genentech, which is a wholly owned independent subsidiary, and the Japanese biotechnology company Chugai Pharmaceuticals, as well as the United States–based companies Ventana and Foundation Medicine. Roche's revenues during fiscal year 2020, were 58.32 billion Swiss francs. Descendants of the founding Hoffmann and Oeri families own slightly over half of the bearer shares with voting rights (a pool of family shareholders 45%, a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
5-HT2C Antagonists
Serotonin (), also known as 5-hydroxytryptamine (5-HT), is a monoamine neurotransmitter with a wide range of functions in both the central nervous system (CNS) and also peripheral tissues. It is involved in mood, cognition, reward, learning, memory, and physiological processes such as vomiting and vasoconstriction. In the CNS, serotonin regulates mood, appetite, and sleep. Most of the body's serotonin—about 90%—is synthesized in the gastrointestinal tract by enterochromaffin cells, where it regulates intestinal movements. It is also produced in smaller amounts in the brainstem's raphe nuclei, the skin's Merkel cells, pulmonary neuroendocrine cells, and taste receptor cells of the tongue. Once secreted, serotonin is taken up by platelets in the blood, which release it during clotting to promote vasoconstriction and platelet aggregation. Around 8% of the body's serotonin is stored in platelets, and 1–2% is found in the CNS. Serotonin acts as both a vasoconstrictor and vas ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
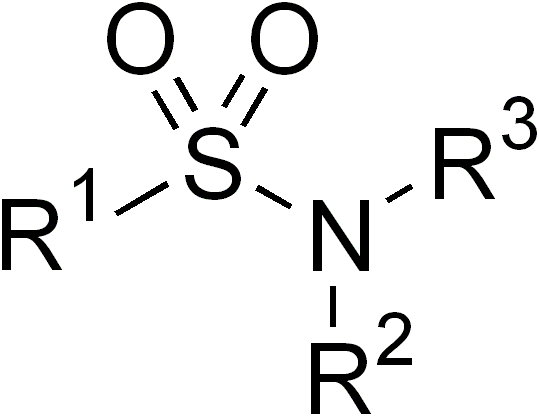
Sulfonamides
In organic chemistry, the sulfonamide functional group (also spelled sulphonamide) is an organosulfur group with the Chemical structure, structure . It consists of a sulfonyl group () connected to an amine group (). Relatively speaking this group is unreactive. Because of the rigidity of the functional group, sulfonamides are typically crystalline; for this reason, the formation of a sulfonamide is a classic method to convert an amine into a crystalline derivative which can be identified by its melting point. Many important drugs contain the sulfonamide group. A sulfonamide (compound) is a chemical compound that contains this group. The general formula is or , where each R is some organic group; for example, "methanesulfonamide" (where R = methane, R' = R" = hydrogen) is . Any sulfonamide can be considered as derived from a sulfonic acid by replacing a hydroxyl group () with an amine group. In medicine, the term "sulfonamide" is sometimes used as a synonym for Sulfonamide (m ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Spiro Compounds
In organic chemistry, spiro compounds are compounds that have at least two molecular rings sharing one common atom. Simple spiro compounds are bicyclic (having just two rings). The presence of only one common atom connecting the two rings distinguishes spiro compounds from other bicyclics.For all four categories, see The specific chapters can be found aan respectively, same access date. For the description featuring adjacent atoms for all but the isolated category, see Clayden, op. cit. Spiro compounds may be fully carbocyclic (all carbon) or heterocyclic (having one or more non-carbon atom). One common type of spiro compound encountered in educational settings is a heterocyclic one— the acetal formed by reaction of a diol with a cyclic ketone. The common atom that connects the two (or sometimes three) rings is called the ''spiro atom''. In carbocyclic spiro compounds like spiro .5ndecane, the spiro-atom is a quaternary carbon, and as the ''-ane'' ending implies, these ar ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Trifluoromethyl Compounds
The trifluoromethyl group is a functional group that has the formula In science, a formula is a concise way of expressing information symbolically, as in a mathematical formula or a ''chemical formula''. The informal use of the term ''formula'' in science refers to the general construct of a relationship betwe ... . The naming of is group is derived from the methyl group (which has the formula ), by replacing each hydrogen atom by a fluorine atom. Some common examples are trifluoromethane , 1,1,1-trifluoroethane , and hexafluoroacetone . Compounds with this group are a subclass of the organofluorines. Properties The trifluoromethyl group has a significant electronegativity that is often described as being intermediate between the electronegativities of fluorine and chlorine. For this reason, trifluoromethyl-substituted compounds are often strong acids, such as trifluoromethanesulfonic acid and trifluoroacetic acid. Conversely, the trifluoromethyl group lowers the basicity of ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phenol Ethers
Phenol (also known as carbolic acid, phenolic acid, or benzenol) is an aromatic organic compound with the molecular formula . It is a white crystalline solid that is volatile and can catch fire. The molecule consists of a phenyl group () bonded to a hydroxy group (). Mildly acidic, it requires careful handling because it can cause chemical burns. It is acutely toxic and is considered a health hazard. Phenol was first extracted from coal tar, but today is produced on a large scale (about 7 million tonnes a year) from petroleum-derived feedstocks. It is an important industrial commodity as a precursor to many materials and useful compounds, and is a liquid when manufactured. It is primarily used to synthesize plastics and related materials. Phenol and its chemical derivatives are essential for production of polycarbonates, epoxies, explosives such as picric acid, Bakelite, nylon, detergents, herbicides such as phenoxy herbicides, and numerous pharmaceutical drugs. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
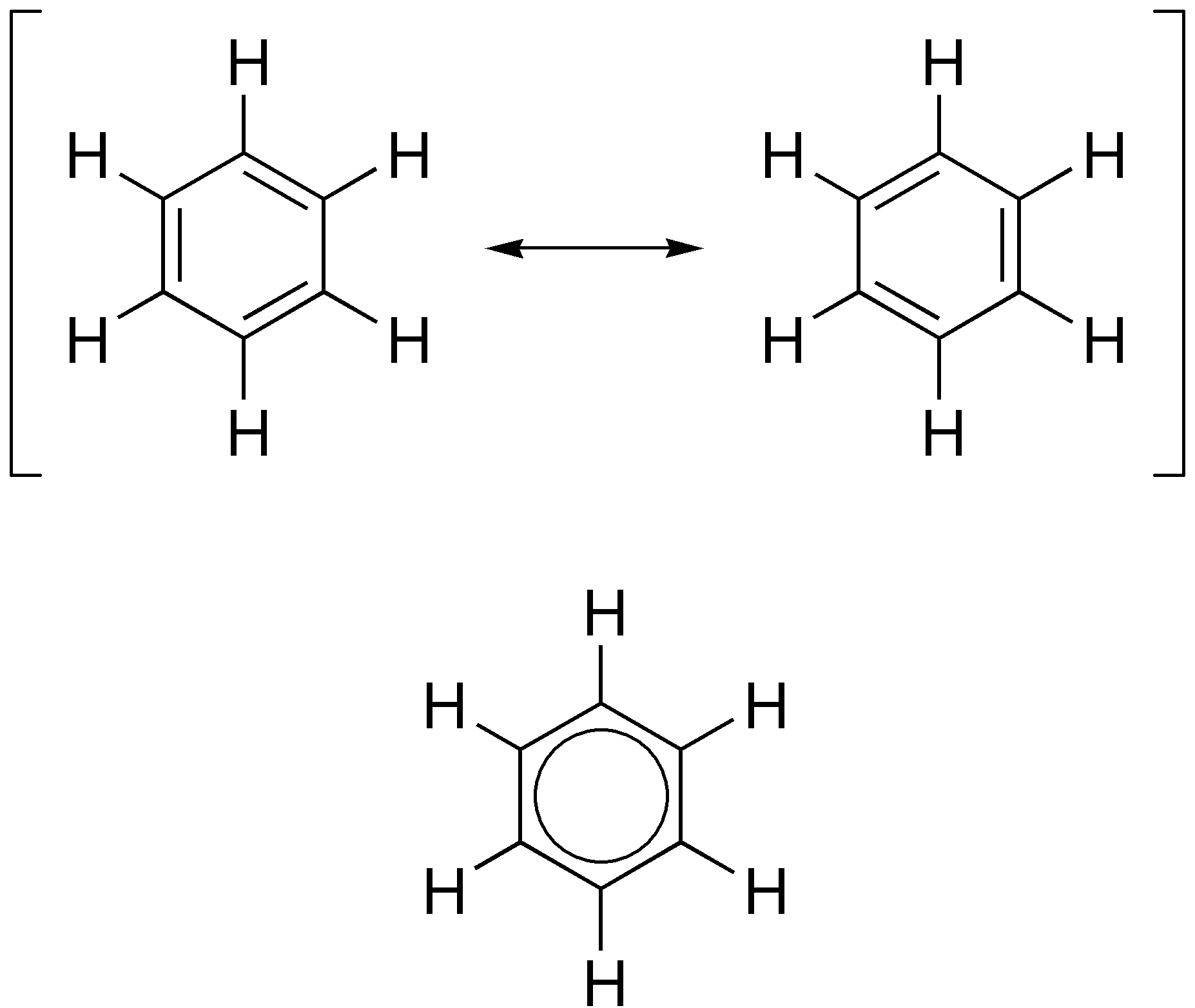
Aromatic Ketones
In organic chemistry, aromaticity is a chemical property describing the way in which a conjugated system, conjugated ring of unsaturated bonds, lone pairs, or empty orbitals exhibits a stabilization stronger than would be expected from conjugation alone. The earliest use of the term was in an article by August Wilhelm Hofmann in 1855. There is no general relationship between aromaticity as a chemical property and the olfaction, olfactory properties of such compounds. Aromaticity can also be considered a manifestation of cyclic delocalization and of Resonance (chemistry), resonance. This is usually considered to be because electrons are free to cycle around circular arrangements of atoms that are alternately single- and double-covalent bond, bonded to one another. This commonly seen model of aromatic rings, namely the idea that benzene was formed from a six-membered carbon ring with alternating single and double bonds (cyclohexatriene), was developed by Friedrich August Kekulé ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nitrogen Heterocycles
Nitrogen is a chemical element; it has symbol N and atomic number 7. Nitrogen is a nonmetal and the lightest member of group 15 of the periodic table, often called the pnictogens. It is a common element in the universe, estimated at seventh in total abundance in the Milky Way and the Solar System. At standard temperature and pressure, two atoms of the element bond to form N2, a colourless and odourless diatomic gas. N2 forms about 78% of Earth's atmosphere, making it the most abundant chemical species in air. Because of the volatility of nitrogen compounds, nitrogen is relatively rare in the solid parts of the Earth. It was first discovered and isolated by Scottish physician Daniel Rutherford in 1772 and independently by Carl Wilhelm Scheele and Henry Cavendish at about the same time. The name was suggested by French chemist Jean-Antoine-Claude Chaptal in 1790 when it was found that nitrogen was present in nitric acid and nitrates. Antoine Lavoisier suggested instead the nam ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Lactams
A lactam is a cyclic amide, formally derived from an amino alkanoic acid through cyclization reactions. The term is a portmanteau of the words '' lactone'' + ''amide''. Nomenclature Greek prefixes in alphabetical order indicate ring size. This ring-size nomenclature stems from the fact that hydrolysis of an α-lactam gives an α-amino acid and that of a β-Lactam gives a β-amino acid, and so on. Synthesis General synthetic methods are used for the organic synthesis of lactams. Beckmann rearrangement Lactams form by the acid-catalyzed rearrangement of oximes in the Beckmann rearrangement. Schmidt reaction Lactams form from cyclic ketones and hydrazoic acid in the Schmidt reaction. Cyclohexanone with hydrazoic acid, forms ε - Caprolactum, which upon treatment with excess acid forms Cardiazole, a heart stimulant. Cyclization of amino acids Lactams can be formed from cyclisation of amino acids via the coupling between an amine and a carboxylic acid within the s ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ureas
image:Biotin_structure.svg, 220 px, Biotin, a water-soluble B vitamins, B vitamin, is a bicyclic urea. In chemistry, ureas are a class of organic compounds with the formula (R2N)2CO where R = H, alkyl, aryl, etc. Thus, in addition to describing the specific chemical compound urea ((H2N)2CO), urea is the name of a functional group that is found in many compounds and materials of both practical and theoretical interest. Generally ureas are colorless crystalline solids, which, owing to the presence of fewer hydrogen bonds, exhibit melting points lower than that of urea itself. Synthesis Ureas can be prepared many methods, but rarely by direct carbonation, which is the route to urea itself. Instead, methods can be classified according those that assemble the urea functionality and those that start with preformed urea. Assembly of N-substituted urea functionality Phosgenation entails the reaction of amines with phosgene, proceeding via the isocyanate (or carbamoyl chloride) as an i ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |