|
Polysulphide
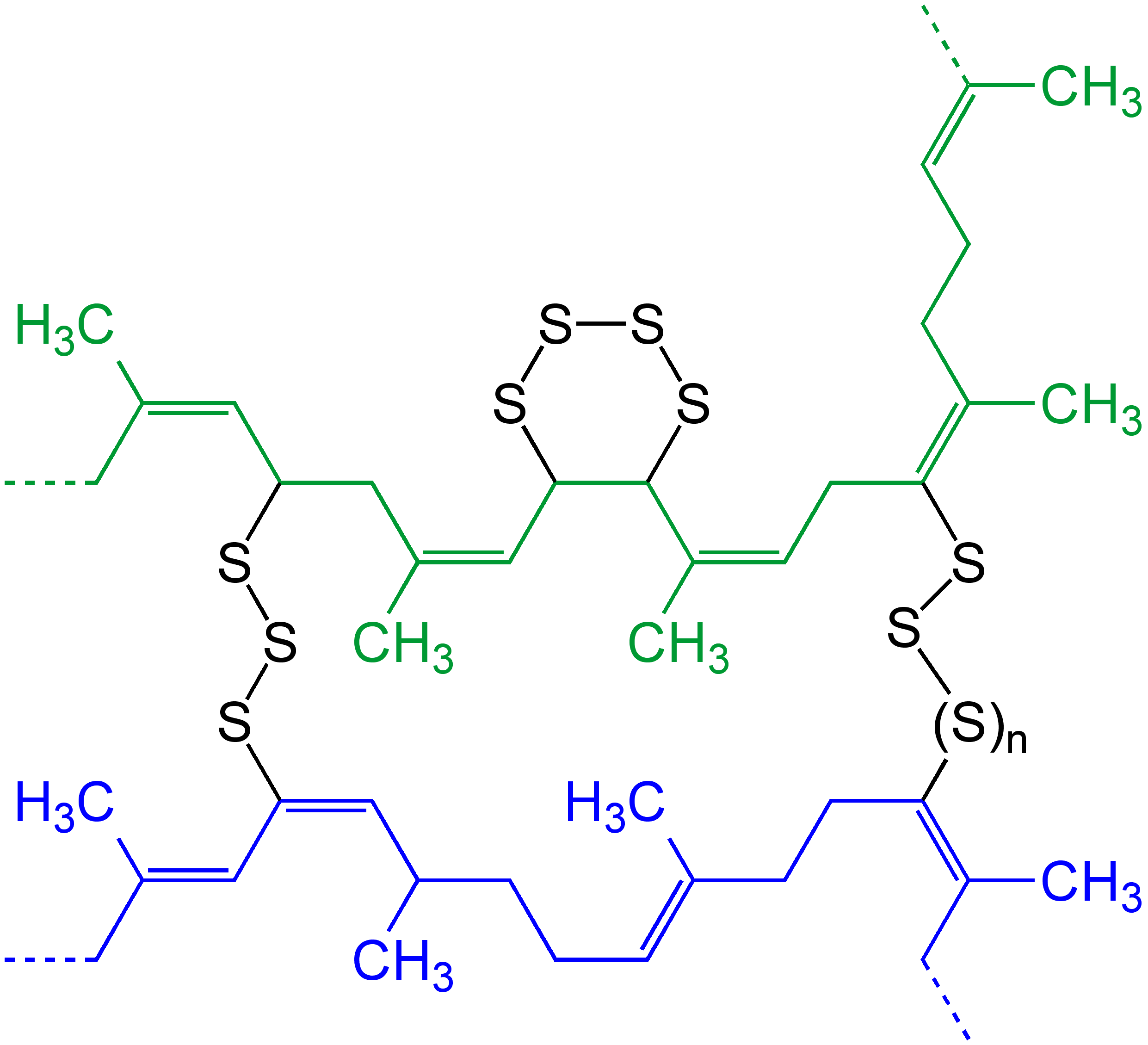
Polysulfides are a class of chemical compounds derived from anionic chains of sulfur atoms. There are two main classes of polysulfides: inorganic and organic. The inorganic polysulfides have the general formula . These anions are the conjugate bases of polysulfanes . Organic polysulfides generally have the formulae , where R is an alkyl or aryl group. Polysulfide salts and complexes The alkali metal polysulfides arise by treatment of a solution of the sulfide with elemental sulfur, e.g. sodium sulfide to sodium polysulfide: In some cases, these anions have been obtained as organic salts, which are soluble in organic solvents. The energy released in the reaction of sodium and elemental sulfur is the basis of battery technology. The sodium–sulfur battery and the lithium–sulfur battery require high temperatures to maintain liquid polysulfide and -conductive membranes that are unreactive toward sodium, sulfur, and sodium sulfide. Polysulfides are ligands in coordination chemi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Vulcanization
Vulcanization (British English: vulcanisation) is a range of processes for hardening rubbers. The term originally referred exclusively to the treatment of natural rubber with sulfur, which remains the most common practice. It has also grown to include the hardening of other (synthetic) rubbers via various means. Examples include silicone rubber via room temperature vulcanizing and chloroprene rubber (neoprene) using metal oxides. Vulcanization can be defined as the curing of elastomers, with the terms 'vulcanization' and 'curing' sometimes used interchangeably in this context. It works by forming cross-links between sections of the polymer chain which results in increased rigidity and durability, as well as other changes in the mechanical and electrical properties of the material. Vulcanization, in common with the curing of other thermosetting polymers, is generally irreversible. The word was suggested by William Brockedon (a friend of Thomas Hancock who attained the B ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Rubber
Rubber, also called India rubber, latex, Amazonian rubber, ''caucho'', or ''caoutchouc'', as initially produced, consists of polymers of the organic compound isoprene, with minor impurities of other organic compounds. Types of polyisoprene that are used as natural rubbers are classified as elastomers. Currently, rubber is harvested mainly in the form of the latex from the Hevea brasiliensis, Pará rubber tree (''Hevea brasiliensis'') or others. The latex is a sticky, milky and white colloid drawn off by making incisions in the bark and collecting the fluid in vessels in a process called "tapping". Manufacturers refine this latex into the rubber that is ready for commercial processing. Natural rubber is used extensively in many applications and products, either alone or in combination with other materials. In most of its useful forms, it has a large stretch ratio and high resilience and also is buoyant and water-proof. Industrial demand for rubber-like materials began to out ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Curing (chemistry)
Curing is a chemical process employed in polymer chemistry and process engineering that produces the toughening or hardening of a polymer material by cross-linking of polymer chains. Even if it is strongly associated with the production of thermosetting polymers, the term "curing" can be used for all the processes where a solid product is obtained from a liquid solution, such as with PVC plastisols. Curing process During the curing process, single monomers and oligomers, mixed with or without a curing agent, react to form a tridimensional polymeric network. In the very first part of the reaction Branching (polymer chemistry), branches of molecules with various architectures are formed, and their molecular weight increases in time with the extent of the reaction until the network size is equal to the size of the system. The system has lost its solubility and its viscosity tends to infinite. The remaining molecules start to coexist with the macroscopic network until they react wit ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cross-link
In chemistry and biology, a cross-link is a bond or a short sequence of bonds that links one polymer chain to another. These links may take the form of covalent bonds or ionic bonds and the polymers can be either synthetic polymers or natural polymers (such as proteins). In polymer chemistry "cross-linking" usually refers to the use of cross-links to promote a change in the polymers' physical properties. When "crosslinking" is used in the biological field, it refers to the use of a probe to link proteins together to check for protein–protein interactions, as well as other creative cross-linking methodologies. Although the term is used to refer to the "linking of polymer chains" for both sciences, the extent of crosslinking and specificities of the crosslinking agents vary greatly. Synthetic polymers : 260px, left, Chemical reactions associated with crosslinking of drying oils, the process that produces curing'' refers to the crosslinking of thermosetting">linoleum. Cros ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Elastomer
An elastomer is a polymer with viscoelasticity (i.e. both viscosity and elasticity) and with weak intermolecular forces, generally low Young's modulus (E) and high failure strain compared with other materials. The term, a portmanteau of ''elastic polymer'', is often used interchangeably with ''rubber'', although the latter is preferred when referring to vulcanisates. Each of the monomers which link to form the polymer is usually a compound of several elements among carbon, hydrogen, oxygen and silicon. Elastomers are amorphous polymers maintained above their glass transition temperature, so that considerable molecular reconformation is feasible without breaking of covalent bonds. Rubber-like solids with elastic properties are called elastomers. Polymer chains are held together in these materials by relatively weak intermolecular bonds, which permit the polymers to stretch in response to macroscopic stresses. Elastomers are usually thermosets (requiring vulcanization ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Vulcanization Of POLYIsoprene V
Vulcanization (British English: vulcanisation) is a range of processes for hardening rubbers. The term originally referred exclusively to the treatment of natural rubber with sulfur, which remains the most common practice. It has also grown to include the hardening of other (synthetic) rubbers via various means. Examples include silicone rubber via room temperature vulcanizing and chloroprene rubber (neoprene) using metal oxides. Vulcanization can be defined as the curing of elastomers, with the terms 'vulcanization' and 'curing' sometimes used interchangeably in this context. It works by forming cross-links between sections of the polymer chain which results in increased rigidity and durability, as well as other changes in the mechanical and electrical properties of the material. Vulcanization, in common with the curing of other thermosetting polymers, is generally irreversible. The word was suggested by William Brockedon (a friend of Thomas Hancock who attained the Briti ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Poly(p-phenylene Sulfide)
Polyphenylene sulfide (PPS) is an organic polymer consisting of aromatic rings linked by thioether, sulfides. Synthetic fiber and textiles derived from this polymer resist chemical and thermal attack. PPS is used in air filter, filter fabric for coal boilers, papermaking felts, electrical insulation, film capacitor#Polyphenylene sulfide (PPS) film capacitors, film capacitors, specialty synthetic membrane, membranes, gaskets, and seal (mechanical), packings. PPS is the precursor to a conductive polymer of the semi-flexible rod polymer family. The PPS, which is otherwise insulating, can be converted to the semiconductor, semiconducting form by oxidation or use of dopants.David Parker, Jan Bussink, Hendrik T. van de Grampel, Gary W. Wheatley, Ernst-Ulrich Dorf, Edgar Ostlinning, Klaus Reinking, "Polymers, High-Temperature" in Ullmann's Encyclopedia of Industrial Chemistry 2002, Wiley-VCH: Weinheim. Polyphenylene sulfide is an engineering plastic, commonly used today as a high-perf ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sealant
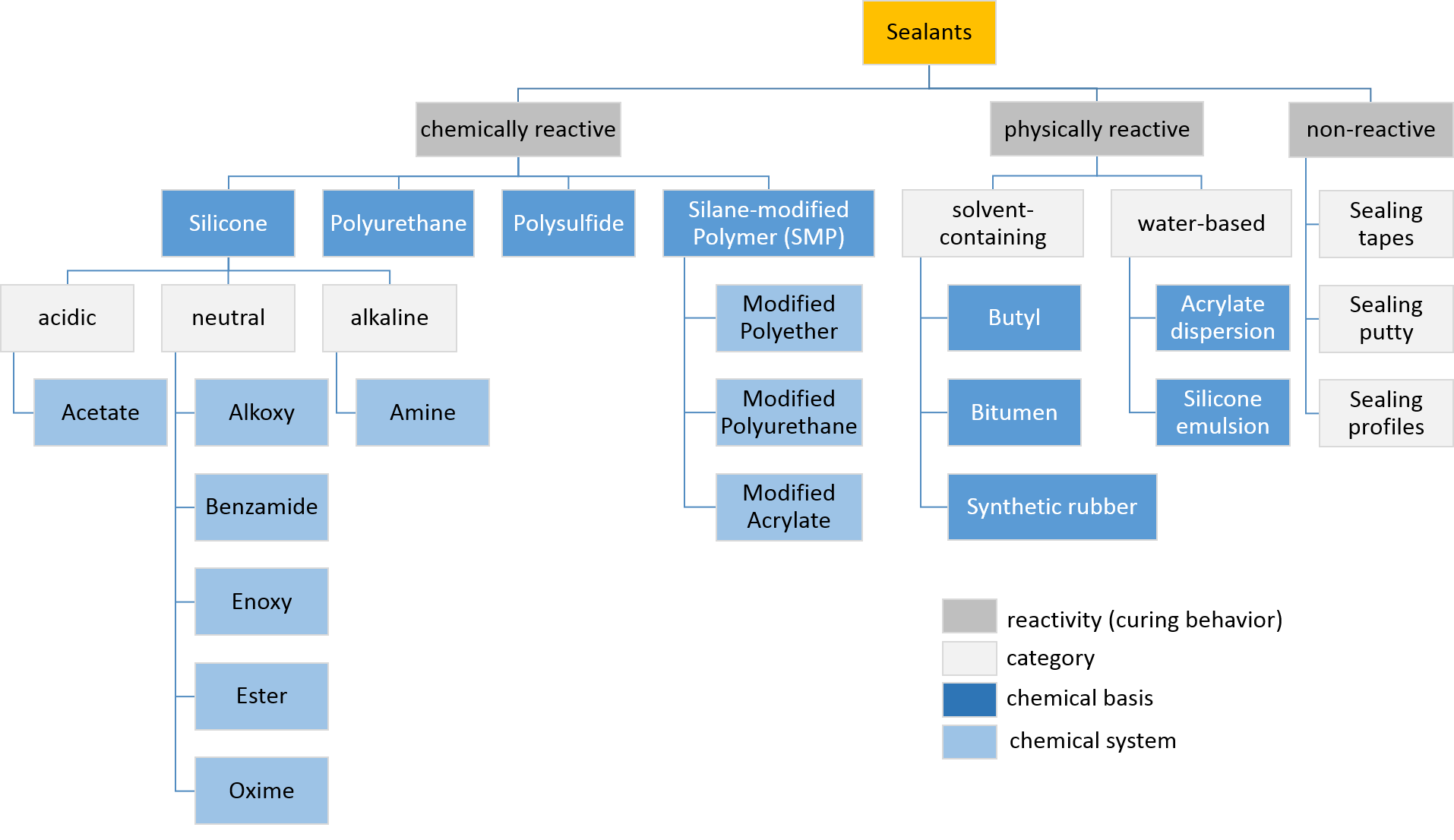
Sealant is a substance used to block the passage of fluids through openings in materials, a type of Seal (mechanical), mechanical seal. In building construction ''sealant'' is sometimes synonymous with ''caulk'' (especially if acrylic latex or polyurethane based) and also serve the purposes of blocking dust, sound and heat transmission. Sealants may be weak or strong, flexible or rigid, permanent or temporary. Sealants are not adhesives but some have adhesive qualities and are called ''adhesive-sealants'' or ''structural sealants''. History Sealants were first used in prehistory in the broadest sense as mud, grass and reeds to seal dwellings from the weather such as the daub in wattle and daub and thatching. Natural sealants and adhesive-sealants included plant resins such as pine pitch and birch pitch, bitumen, wax, tar, natural gum, clay (mud) mortar, lime mortar, lead, blood and egg. In the 17th century glazing putty was first used to seal window glass made with linseed oil a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ring-opening Polymerization
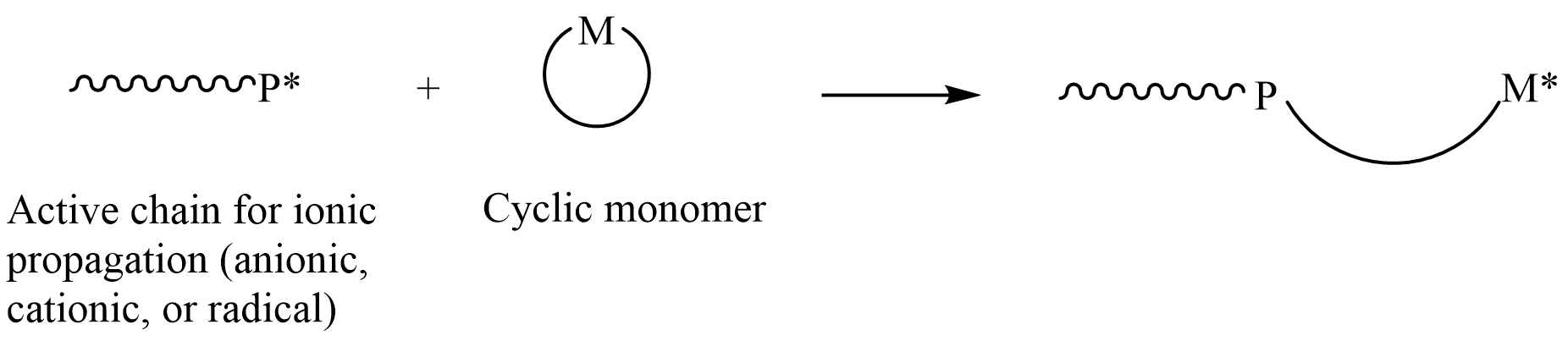
In polymer chemistry, ring-opening polymerization (ROP) is a form of chain-growth polymerization in which the terminus of a polymer chain attacks cyclic monomers to form a longer polymer (see figure). The reactive center can be radical, anionic or cationic. Ring-opening of cyclic monomers is often driven by the relief of bond-angle strain. Thus, as is the case for other types of polymerization, the enthalpy change in ring-opening is negative. Many rings undergo ROP. Monomers Many cyclic monomers are amenable to ROP. These include epoxides, cyclic trisiloxanes, some lactones and lactides, cyclic anhydrides, cyclic carbonates, and amino acid ''N''-carboxyanhydrides. Many strained cycloalkenes, e.g norbornene, are suitable monomers via ring-opening metathesis polymerization. Even highly strained cycloalkane rings, such as cyclopropane and cyclobutane derivatives, can undergo ROP. History Ring-opening polymerization has been used since the beginning of the 1900s to produce ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Thiokol (polymer)
''Thiokol'' is a trade mark for various organic polysulfide polymers. Thiokol polymers are used as an elastomer in seals and sealants. The distinction between the polymers first commercialized by the Thiokol Chemical Company and subsequent polysulfide materials is often unclear. The name Thiokol is a portmanteau of the Greek words for sulfur () and glue () Preparation and structure A variety of thiokols are recognized. Typically they are prepared by the combination of 2-chloroethanol, formaldehyde, and sodium polysulfide (Na2S''x''). The chloroethanol is produced in situ from ethylene oxide and hydrogen chloride. The rank ''x'' of the polysulfide is an important variable. Crosslinking agents are used, such as 1,2,3-trichloropropane. An idealized polymer is represented by this formula HS(CH2CH2OCH2OCH2CH2SS)''n''CH2CH2OCH2OCH2CH2SH. Thiol-terminated resins can be cured oxidatively. History In 1838, Swiss chemists reported the preparation of hydrophobic rubbery materials by the a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |