|
Phase Transitions
In physics, chemistry, and other related fields like biology, a phase transition (or phase change) is the physical process of transition between one state of a medium and another. Commonly the term is used to refer to changes among the basic State of matter, states of matter: solid, liquid, and gas, and in rare cases, plasma (physics), plasma. A phase of a thermodynamic system and the states of matter have uniform physical property, physical properties. During a phase transition of a given medium, certain properties of the medium change as a result of the change of external conditions, such as temperature or pressure. This can be a discontinuous change; for example, a liquid may become gas upon heating to its boiling point, resulting in an abrupt change in volume. The identification of the external conditions at which a transformation occurs defines the phase transition point. Types of phase transition States of matter Phase transitions commonly refer to when a substance tran ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phase Change - En
Phase or phases may refer to: Science *State of matter, or phase, one of the distinct forms in which matter can exist *Phase (matter), a region of space throughout which all physical properties are essentially uniform *Phase space, a mathematical space in which each possible state of a physical system is represented by a point also referred to as a "microscopic state" ** Phase space formulation, a formulation of quantum mechanics in phase space *Phase (waves), the position of a point in time (an instant) on a waveform cycle **Instantaneous phase, generalization for both cyclic and non-cyclic phenomena * AC phase, the phase offset between alternating current electric power in multiple conducting wires ** Single-phase electric power, distribution of AC electric power in a system where the voltages of the supply vary in unison **Three-phase electric power, a common method of AC electric power generation, transmission, and distribution * Phase problem, the loss of information (the pha ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Freezing Point
The melting point (or, rarely, liquefaction point) of a substance is the temperature at which it changes state of matter, state from solid to liquid. At the melting point the solid and liquid phase (matter), phase exist in Thermodynamic equilibrium, equilibrium. The melting point of a substance depends on pressure and is usually specified at a Standard temperature and pressure, standard pressure such as 1 Atmosphere (unit), atmosphere or 100 Pascal (unit), kPa. When considered as the temperature of the reverse change from liquid to solid, it is referred to as the freezing point or crystallization point. Because of the ability of substances to Supercooling, supercool, the freezing point can easily appear to be below its actual value. When the "characteristic freezing point" of a substance is determined, in fact, the actual methodology is almost always "the principle of observing the disappearance rather than the formation of ice, that is, the #Melting point measurements, melting ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Displacive Phase Transformations
A diffusionless transformation, commonly known as displacive transformation, denotes solid-state alterations in crystal structures that do not hinge on the diffusion of atoms across extensive distances. Rather, these transformations manifest as a result of synchronized shifts in atomic positions, wherein atoms undergo displacements of distances smaller than the spacing between adjacent atoms, all while preserving their relative arrangement. An example of such a phenomenon is the martensitic transformation, a notable occurrence observed in the context of steel materials. The term "martensite" was originally coined to describe the rigid and finely dispersed constituent that emerges in steels subjected to rapid cooling. Subsequent investigations revealed that materials beyond ferrous alloys, such as non-ferrous alloys and ceramics, can also undergo diffusionless transformations. Consequently, the term "martensite" has evolved to encompass the resultant product arising from such tr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
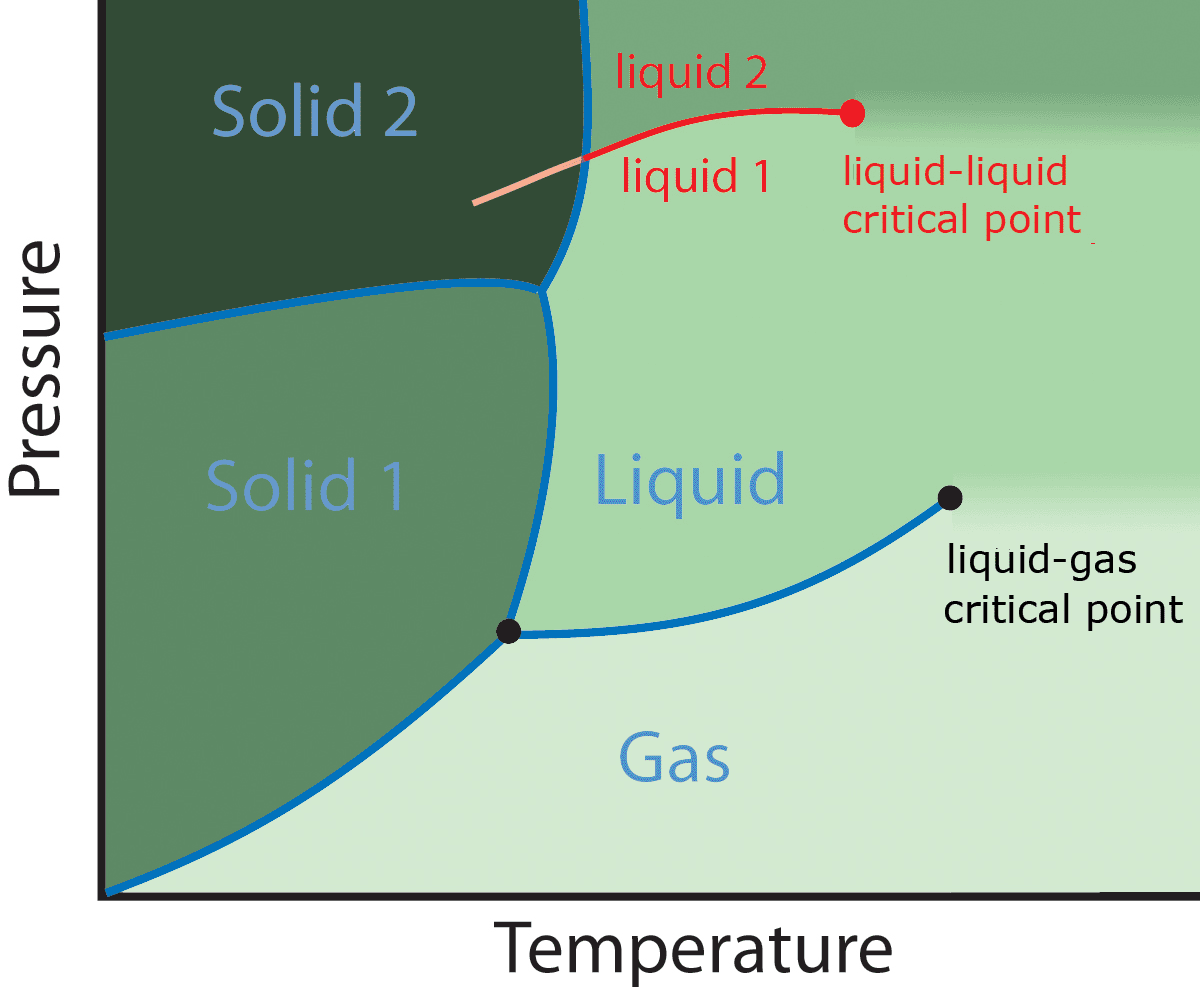
Polyamorphism
Polyamorphism is the ability of a substance to exist in several different amorphous modifications. It is analogous to the polymorphism of crystalline materials. Many amorphous substances can exist with different amorphous characteristics (e.g. polymers). However, polyamorphism requires ''two distinct'' amorphous states with a clear, discontinuous (first-order) phase transition between them. When such a transition occurs between two stable liquid states, a polyamorphic transition may also be referred to as a liquid–liquid phase transition. Overview Even though amorphous materials exhibit no long-range periodic atomic ordering, there is still significant and varied local structure at inter-atomic length scales (see structure of liquids and glasses). Different local structures can produce amorphous phases of the same chemical composition with different physical properties such as density. In several cases sharp transitions have been observed between two different density amorp ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Amorphous Solid
In condensed matter physics and materials science, an amorphous solid (or non-crystalline solid) is a solid that lacks the long-range order that is a characteristic of a crystal. The terms "glass" and "glassy solid" are sometimes used synonymously with amorphous solid; however, these terms refer specifically to amorphous materials that undergo a glass transition. Examples of amorphous solids include glasses, metallic glasses, and certain types of plastics and polymers. Etymology The term "Amorphous" comes from the Greek language, Greek ''a'' ("without"), and ''morphé'' ("shape, form"). Structure Amorphous materials have an internal structure of molecular-scale structural blocks that can be similar to the basic structural units in the crystalline phase of the same compound. Unlike in crystalline materials, however, no long-range regularity exists: amorphous materials cannot be described by the repetition of a finite unit cell. Statistical measures, such as the atomic density ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Crystal Structure
In crystallography, crystal structure is a description of ordered arrangement of atoms, ions, or molecules in a crystalline material. Ordered structures occur from intrinsic nature of constituent particles to form symmetric patterns that repeat along the principal directions of three-dimensional space in matter. The smallest group of particles in a material that constitutes this repeating pattern is the unit cell of the structure. The unit cell completely reflects the symmetry and structure of the entire crystal, which is built up by repetitive translation of the unit cell along its principal axes. The translation vectors define the nodes of the Bravais lattice. The lengths of principal axes/edges, of the unit cell and angles between them are lattice constants, also called ''lattice parameters'' or ''cell parameters''. The symmetry properties of a crystal are described by the concept of space groups. All possible symmetric arrangements of particles in three-dimensional space ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
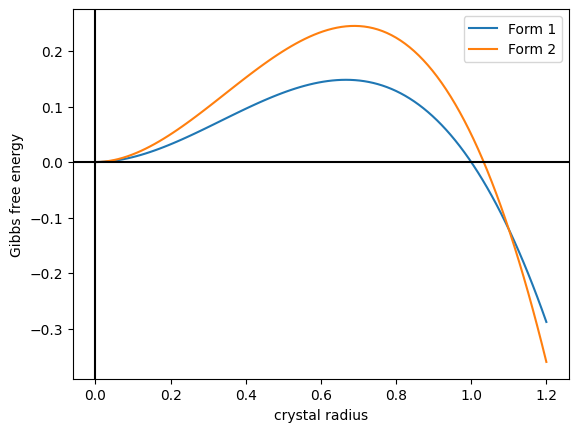
Polymorphism (materials Science)
In crystallography, polymorphism is the phenomenon where a compound or element can crystallize into more than one crystal structure. The preceding definition has evolved over many years and is still under discussion today. Discussion of the defining characteristics of polymorphism involves distinguishing among types of transitions and structural changes occurring in polymorphism versus those in other phenomena. Overview Phase transitions (phase changes) that help describe polymorphism include polymorphic transitions as well as melting and vaporization transitions. According to IUPAC, a polymorphic transition is "A reversible transition of a solid crystalline phase at a certain temperature and pressure (the inversion point) to another phase of the same chemical composition with a different crystal structure." Additionally, Walter McCrone described the phases in polymorphic matter as "different in crystal structure but identical in the liquid or vapor states." McCrone also def ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Allotropy
Allotropy or allotropism () is the property of some chemical elements to exist in two or more different forms, in the same physical State of matter, state, known as allotropes of the elements. Allotropes are different structural modifications of an element: the atoms of the element are Chemical bond, bonded together in different manners. For example, the allotropes of carbon include diamond (the carbon atoms are bonded together to form a Cubic crystal system, cubic lattice of Tetrahedral molecular geometry, tetrahedra), graphite (the carbon atoms are bonded together in sheets of a hexagonal lattice), graphene (single sheets of graphite), and fullerenes (the carbon atoms are bonded together in spherical, tubular, or ellipsoidal formations). The term ''allotropy'' is used for elements only, not for Chemical compound, compounds. The more general term, used for any compound, is Polymorphism (materials science), polymorphism, although its use is usually restricted to solid materials ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pure Iron Phase Diagram (EN)
Pure may refer to: Computing * Pure function * PureSystems, a family of computer systems introduced by IBM in 2012 * Pure Software, a company founded in 1991 by Reed Hastings to support the Purify tool * Pure-FTPd, FTP server software * Pure (programming language), functional programming language based on term rewriting * Pure Storage, a company that makes datacenter storage solutions Companies and products * Pure (app), dating app * Pure (company), a British consumer electronics company specialising in digital radios * Pure (restaurant chain), a British fast food chain * Pure Insurance, Privilege Underwriters Reciprocal Exchange * Pure Trading, a Canadian electronic communication network operated by CNQ * Pure Oil, a U.S. chain of gas stations * Propulsion Universelle et Récuperation d'Énergie (PURE), a motorsport engineering company * Pure FM (Portsmouth), a university radio station based in Portsmouth, UK * Pure (Belgian radio station), a former Belgian radio sta ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Supercooling
Supercooling, also known as undercooling, is the process of lowering the temperature of a liquid below its freezing point without it becoming a solid. Per the established international definition, supercooling means ''‘cooling a substance below the normal freezing point without solidification’.'' IIR International Dictionary of Refrigeration, http://dictionary.iifiir.org/search.php ASHRAE Terminology, https://www.ashrae.org/technical-resources/free-resources/ashrae-terminology While it can be achieved by different physical means, the postponed solidification is most often due to the absence of nucleation, seed crystals or nuclei around which a crystal structure can form. The supercooling of water can be achieved without any special techniques other than chemical demineralization, down to . Supercooled water can occur naturally, for example in the atmosphere, animals or plants. This phenomenon was first identified in 1724 by Daniel Gabriel Fahrenheit, while developing Fahren ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Superheating
In thermodynamics, superheating (sometimes referred to as boiling retardation, or boiling delay) is the phenomenon in which a liquid is heated to a temperature higher than its boiling point, without boiling. This is a so-called ''metastable state'' or '' metastate'', where boiling might occur at any time, induced by external or internal effects.Debenedetti, P.G.Metastable Liquids: Concepts and Principles; Princeton University Press: Princeton, NJ, USA, 1996.Maris, H., Balibar, S. (2000"Negative Pressures and Cavitation in Liquid Helium"Physics Today 53, 29 Superheating is achieved by heating a homogeneous substance in a clean container, free of nucleation sites, while taking care not to disturb the liquid. This may occur by microwaving water in a very smooth container. Disturbing the water may cause an unsafe eruption of hot water and result in burns. Cause Water is said to "boil" when bubbles of water vapor grow without bound, bursting at the surface. For a vapor bubble ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |