|
Oxyhydrogen
Oxyhydrogen is a mixture of hydrogen (H2) and oxygen (O2) gases. This gaseous mixture is used for torches to process refractory materials and was the first gaseous mixture used for welding. Theoretically, a ratio of 2:1 hydrogen:oxygen is enough to achieve maximum efficiency; in practice a ratio 4:1 or 5:1 is needed to avoid an oxidizing flame. This mixture may also be referred to as ' (Scandinavian and German '; ), although some authors define knallgas to be a generic term for the mixture of fuel with the precise amount of oxygen required for complete combustion, thus 2:1 oxyhydrogen would be called "hydrogen-knallgas". "Brown's gas" and HHO are terms for oxyhydrogen originating in pseudoscience, although is preferred due to meaning . Properties Oxyhydrogen will combust when brought to its autoignition temperature. For the stoichiometric mixture in air, at normal atmospheric pressure, autoignition occurs at about 570 °C (1065 °F). The minimum energy requi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electrolysis Of Water
Electrolysis of water is using electricity to Water splitting, split water into oxygen () and hydrogen () gas by electrolysis. Hydrogen gas released in this way can be used as hydrogen fuel, but must be kept apart from the oxygen as the mixture would be extremely explosive. Separately pressurised into convenient 'tanks' or 'gas bottles', hydrogen can be used for oxyhydrogen welding and other applications, as the hydrogen / oxygen flame can reach approximately 2,800°C. Water electrolysis requires a minimum potential difference of 1.23 volts, although at that voltage external heat is also required. Typically 1.5 volts is required. Electrolysis is rare in industrial applications since hydrogen can be produced less expensively from fossil fuels. Most of the time, hydrogen is made by splitting methane (CH4) into carbon dioxide (CO2) and hydrogen (H2) via steam reforming. This is a carbon-intensive process that means for every kilogram of “grey” hydrogen produced, approximatel ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Welding
Welding is a fabrication (metal), fabrication process that joins materials, usually metals or thermoplastics, primarily by using high temperature to melting, melt the parts together and allow them to cool, causing Fusion welding, fusion. Common alternative methods include solvent welding (of thermoplastics) using chemicals to melt materials being bonded without heat, and #Solid-state welding, solid-state welding processes which bond without melting, such as pressure, cold welding, and diffusion bonding. Metal welding is distinct from lower temperature bonding techniques such as brazing and soldering, which do not melt the base metal (parent metal) and instead require flowing a filler metal to solidify their bonds. In addition to melting the base metal in welding, a filler material is typically added to the joint to form a pool of molten material (the weld pool) that cools to form a joint that can be stronger than the base material. Welding also requires a form of shield to ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electrolyser 1884
In chemistry and manufacturing, electrolysis is a technique that uses direct electric current (DC) to drive an otherwise non-spontaneous chemical reaction. Electrolysis is commercially important as a stage in the separation of elements from naturally occurring sources such as ores using an electrolytic cell. The voltage that is needed for electrolysis to occur is called the decomposition potential. The word "lysis" means to separate or break, so in terms, electrolysis would mean "breakdown via electricity." Etymology The word "electrolysis" was introduced by Michael Faraday in 1834, using the Greek words "amber", which since the 17th century was associated with electrical phenomena, and ' meaning "dissolution". Nevertheless, electrolysis, as a tool to study chemical reactions and obtain pure elements, precedes the coinage of the term and formal description by Faraday. History In the early nineteenth century, William Nicholson and Anthony Carlisle sought to further Volt ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Flame Temperature
A flame () is the visible, gaseous part of a fire. It is caused by a highly exothermic chemical reaction made in a thin zone. When flames are hot enough to have ionized gaseous components of sufficient density, they are then considered plasma. Mechanism Color and temperature of a flame are dependent on the type of fuel involved in the combustion. For example, when a lighter is held to a candle, the applied heat causes the fuel molecules in the candle wax to vaporize. In this state they can then readily react with oxygen in the air, which gives off enough heat in the subsequent exothermic reaction to vaporize yet more fuel, thus sustaining a consistent flame. The high temperature of the flame causes the vaporized fuel molecules to decompose, forming various incomplete combustion products and free radicals, and these products then react with each other and with the oxidizer involved in the reaction of the following flame (fire). One may investigate different parts of a candle ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Black Body
A black body or blackbody is an idealized physical body that absorbs all incident electromagnetic radiation, regardless of frequency or angle of incidence. The radiation emitted by a black body in thermal equilibrium with its environment is called ''black-body radiation''. The name "black body" is given because it absorbs all colors of light. In contrast, a white body is one with a "rough surface that reflects all incident rays completely and uniformly in all directions." A black body in thermal equilibrium (that is, at a constant temperature) emits electromagnetic black-body radiation. The radiation is emitted according to Planck's law, meaning that it has a spectrum that is determined by the temperature alone (see figure at right), not by the body's shape or composition. An ideal black body in thermal equilibrium has two main properties: #It is an ideal emitter: at every frequency, it emits as much or more thermal radiative energy as any other body at the same temperature. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Calcium Oxide
Calcium oxide (formula: Ca O), commonly known as quicklime or burnt lime, is a widely used chemical compound. It is a white, caustic, alkaline, crystalline solid at room temperature. The broadly used term '' lime'' connotes calcium-containing inorganic compounds, in which carbonates, oxides, and hydroxides of calcium, silicon, magnesium, aluminium, and iron predominate. By contrast, ''quicklime'' specifically applies to the single compound calcium oxide. Calcium oxide that survives processing without reacting in building products, such as cement, is called free lime. Quicklime is relatively inexpensive. Both it and the chemical derivative calcium hydroxide (of which quicklime is the base anhydride) are important commodity chemicals. Preparation Calcium oxide is usually made by the thermal decomposition of materials, such as limestone or seashells, that contain calcium carbonate (CaCO3; mineral calcite) in a lime kiln. This is accomplished by heating the material to above , ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Limelight
Limelight (also known as Drummond light or calcium light)James R. Smith (2004). ''San Francisco's Lost Landmarks'', Quill Driver Books. is a non-electric type of stage lighting that was once used in theatres and music halls. An intense illumination is created when a flame fed by oxygen and hydrogen is directed at a cylinder of quicklime (calcium oxide), due to a combination of incandescence and candoluminescence. Although it has long since been replaced by electric lighting, the term has nonetheless survived, as someone in the public eye is still said to be "in the limelight". The actual lamps are called "limes", a term which has been transferred to electrical equivalents. History Discovery and invention The limelight effect was discovered in the 1820s by Goldsworthy Gurney, based on his work with the "oxy-hydrogen blowpipe", credit for which is normally given to Robert Hare. In 1825, a Scottish engineer, Thomas Drummond (1797–1840), saw a demonstration of the effect ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Gas Lighting
Gas lighting is the production of artificial light from combustion of a fuel gas such as methane, propane, butane, acetylene, ethylene, hydrogen, carbon monoxide, coal gas (town gas) or natural gas. The light is produced either directly by the flame, generally by using special mixes (typically propane or butane) of illuminating gas to increase brightness, or indirectly with other components such as the gas mantle or the limelight, with the gas primarily functioning to heat the mantle or the lime to incandescence. Before electricity became sufficiently widespread and economical to allow for general public use, gas lighting was prevalent for outdoor and indoor use in cities and suburbs where the infrastructure for distribution of gas was practical. At that time, the most common fuels for gas lighting were wood gas, coal gas and, in limited cases, water gas. Early gas lights were ignited manually by lamplighters, although many later designs are self-igniting. Gas light ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Limelight Diagram
Limelight (also known as Drummond light or calcium light)James R. Smith (2004). ''San Francisco's Lost Landmarks'', Quill Driver Books. is a non-electric type of stage lighting that was once used in Theater (structure), theatres and music halls. An intense illumination is created when a flame fed by Oxyhydrogen, oxygen and hydrogen is directed at a cylinder of quicklime (calcium oxide), due to a combination of incandescence and candoluminescence. Although it has long since been replaced by electric lighting, the term has nonetheless survived, as someone in the public eye is still said to be "in the limelight". The actual lamps are called "limes", a term which has been transferred to electrical equivalents. History Discovery and invention The limelight effect was discovered in the 1820s by Goldsworthy Gurney, based on his work with the "oxy-hydrogen blowpipe (tool), blowpipe", credit for which is normally given to Robert Hare (chemist), Robert Hare. In 1825, a Scotland, Scotti ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
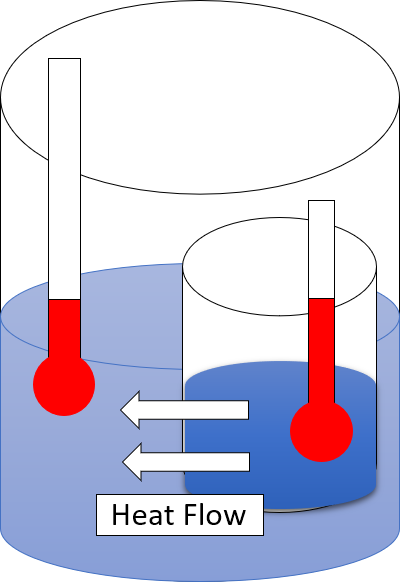
Second Law Of Thermodynamics
The second law of thermodynamics is a physical law based on Universal (metaphysics), universal empirical observation concerning heat and Energy transformation, energy interconversions. A simple statement of the law is that heat always flows spontaneously from hotter to colder regions of matter (or 'downhill' in terms of the temperature gradient). Another statement is: "Not all heat can be converted into Work (thermodynamics), work in a cyclic process."Young, H. D; Freedman, R. A. (2004). ''University Physics'', 11th edition. Pearson. p. 764. The second law of thermodynamics establishes the concept of entropy as a physical property of a thermodynamic system. It predicts whether processes are forbidden despite obeying the requirement of conservation of energy as expressed in the first law of thermodynamics and provides necessary criteria for spontaneous processes. For example, the first law allows the process of a cup falling off a table and breaking on the floor, as well as allowi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
First Law Of Thermodynamics
The first law of thermodynamics is a formulation of the law of conservation of energy in the context of thermodynamic processes. For a thermodynamic process affecting a thermodynamic system without transfer of matter, the law distinguishes two principal forms of energy transfer, heat and thermodynamic work. The law also defines the internal energy of a system, an extensive property for taking account of the balance of heat transfer, thermodynamic work, and matter transfer, into and out of the system. Energy cannot be created or destroyed, but it can be transformed from one form to another. In an externally isolated system, with internal changes, the sum of all forms of energy is constant. An equivalent statement is that perpetual motion machines of the first kind are impossible; work done by a system on its surroundings requires that the system's internal energy be consumed, so that the amount of internal energy lost by that work must be resupplied as heat by an external e ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
William Nicholson (chemist)
William Nicholson (13 December 175321 May 1815) was an English writer, translator, publisher, scientist, inventor, patent agent and civil engineer. He launched the first monthly scientific journal in Britain, ''Journal of Natural Philosophy, Chemistry, and the Arts'', in 1797, and remained its editor until 1814. In 1800, he and Anthony Carlisle were the first to achieve electrolysis, the splitting of water into hydrogen and oxygen, using a voltaic pile. Nicholson also wrote extensively on natural philosophy and chemistry. Early life Nicholson was educated in Yorkshire, and after leaving school, he made two voyages as a midshipman in the service of the British East India Company. His first voyage was to India and the second voyage was to China on board the ''Gatton,'' (1772-1773). Subsequently, having become acquainted with Josiah Wedgwood in 1775, he moved to Amsterdam, where he made a living for a few years as Wedgwood's agent. On his return to England he was persuaded by T ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |