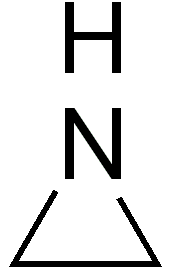|
N-philes
N-philes are group of radical molecules which are specifically attracted to the C=N bonds, defying often the selectivity rules of electrophilic attack. N-philes can often masquerade as electrophiles, where acyl radicals are excellent examples which interact with pi electrons In chemistry, pi bonds (π bonds) are covalent chemical bonds, in each of which two lobes of an orbital on one atom overlap with two lobes of an orbital on another atom, and in which this overlap occurs laterally. Each of these atomic orbitals ... of aryl groups. References * Environmental chemistry Biomolecules Biological processes Free radicals {{organic-chem-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Carbon–nitrogen Bond
A carbon–nitrogen bond is a covalent bond between carbon and nitrogen and is one of the most abundant bonds in organic chemistry and biochemistry. Nitrogen has five valence electrons and in simple amines it is trivalent, with the two remaining electrons forming a lone pair. Through that pair, nitrogen can form an additional bond to hydrogen making it tetravalent and with a positive charge in ammonium salts. Many nitrogen compounds can thus be potentially basic but its degree depends on the configuration: the nitrogen atom in amides is not basic due to delocalization of the lone pair into a double bond and in pyrrole the lone pair is part of an aromatic sextet. Similar to carbon–carbon bonds, these bonds can form stable double bonds, as in imines; and triple bonds, such as nitriles. Bond lengths range from 147.9 pm for simple amines to 147.5 pm for C-N= compounds such as nitromethane to 135.2 pm for partial double bonds in pyridine to 115.8 pm for triple bonds as i ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Radical (chemistry)
In chemistry, a radical, also known as a free radical, is an atom, molecule, or ion that has at least one unpaired valence electron. With some exceptions, these unpaired electrons make radicals highly chemically reactive. Many radicals spontaneously dimerize. Most organic radicals have short lifetimes. A notable example of a radical is the hydroxyl radical (HO·), a molecule that has one unpaired electron on the oxygen atom. Two other examples are triplet oxygen and triplet carbene (꞉) which have two unpaired electrons. Radicals may be generated in a number of ways, but typical methods involve redox reactions. Ionizing radiation, heat, electrical discharges, and electrolysis are known to produce radicals. Radicals are intermediates in many chemical reactions, more so than is apparent from the balanced equations. Radicals are important in combustion, atmospheric chemistry, polymerization, plasma chemistry, biochemistry, and many other chemical processes. A majority ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electrophilic
In chemistry, an electrophile is a chemical species that forms bonds with nucleophiles by accepting an electron pair. Because electrophiles accept electrons, they are Lewis acids. Most electrophiles are positively charged, have an atom that carries a partial positive charge, or have an atom that does not have an octet of electrons. Electrophiles mainly interact with nucleophiles through addition and substitution reactions. Frequently seen electrophiles in organic syntheses include cations such as H+ and NO+, polarized neutral molecules such as HCl, alkyl halides, acyl halides, and carbonyl compounds, polarizable neutral molecules such as Cl2 and Br2, oxidizing agents such as organic peracids, chemical species that do not satisfy the octet rule such as carbenes and radicals, and some Lewis acids such as BH3 and DIBAL. Organic chemistry Addition of halogens These occur between alkenes and electrophiles, often halogens as in halogen addition reactions. Common ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Acyl
In chemistry, an acyl group is a moiety derived by the removal of one or more hydroxyl groups from an oxoacid, including inorganic acids. It contains a double-bonded oxygen atom and an organyl group () or hydrogen in the case of formyl group (). In organic chemistry, the acyl group (IUPAC name alkanoyl if the organyl group is alkyl) is usually derived from a carboxylic acid, in which case it has the formula , where R represents an organyl group or hydrogen. Although the term is almost always applied to organic compounds, acyl groups can in principle be derived from other types of acids such as sulfonic acids and phosphonic acids. In the most common arrangement, acyl groups are attached to a larger molecular fragment, in which case the carbon and oxygen atoms are linked by a double bond. Reactivity trends There are five main types of acyl derivatives. Acid halides are the most reactive towards nucleophiles, followed by anhydrides, esters, and amides. Carboxylate ions ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pi Electrons
In chemistry, pi bonds (π bonds) are covalent chemical bonds, in each of which two lobes of an orbital on one atom overlap with two lobes of an orbital on another atom, and in which this overlap occurs laterally. Each of these atomic orbitals has an electron density of zero at a shared nodal plane that passes through the two bonded nuclei. This plane also is a nodal plane for the molecular orbital of the pi bond. Pi bonds can form in double and triple bonds but do not form in single bonds in most cases. The Greek letter π in their name refers to p orbitals, since the orbital symmetry of the pi bond is the same as that of the p orbital when seen down the bond axis. One common form of this sort of bonding involves p orbitals themselves, though d orbitals also engage in pi bonding. This latter mode forms part of the basis for metal-metal multiple bonding. Properties Pi bonds are usually weaker than sigma bonds. The C–C double bond, composed of one sigma and one p ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Environmental Chemistry
Environmental chemistry is the scientific study of the chemical and biochemical phenomena that occur in natural places. It should not be confused with green chemistry, which seeks to reduce potential pollution at its source. It can be defined as the study of the sources, reactions, transport, effects, and fates of chemical species in the air, soil, and water environments; and the effect of human activity and biological activity on these. Environmental chemistry is an interdisciplinary science that includes atmospheric, aquatic and soil chemistry, as well as heavily relying on analytical chemistry and being related to environmental and other areas of science. Environmental chemistry involves first understanding how the uncontaminated environment works, which chemicals in what concentrations are present naturally, and with what effects. Without this it would be impossible to accurately study the effects humans have on the environment through the release of chemicals. Envir ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Biomolecules
A biomolecule or biological molecule is loosely defined as a molecule produced by a living organism and essential to one or more typically biological processes. Biomolecules include large macromolecules such as proteins, carbohydrates, lipids, and nucleic acids, as well as small molecules such as vitamins and hormones. A general name for this class of material is ''biological materials''. Biomolecules are an important element of living organisms. They are often endogenous, i.e. produced within the organism, but organisms usually also need exogenous biomolecules, for example certain nutrients, to survive. Biomolecules and their reactions are studied in biology and its subfields of biochemistry and molecular biology. Most biomolecules are organic compounds, and just four elements—oxygen, carbon, hydrogen, and nitrogen—make up 96% of the human body's mass. But many other elements, such as the various biometals, are also present in small amounts. The uniformity of bo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Biological Processes
Biological processes are those processes that are necessary for an organism to live and that shape its capacities for interacting with its environment. Biological processes are made of many chemical reactions or other events that are involved in the persistence and transformation of life forms. Regulation of biological processes occurs when any process is modulated in its frequency, rate or extent. Biological processes are regulated by many means; examples include the control of gene expression, protein modification or interaction with a protein or substrate molecule. * Homeostasis: regulation of the internal environment to maintain a constant state; for example, sweating to reduce temperature * Organization: being structurally composed of one or more cells – the basic units of life * Metabolism: transformation of energy by converting chemicals and energy into cellular components (anabolism) and decomposing organic matter ( catabolism). Living things require energy to mai ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |