|
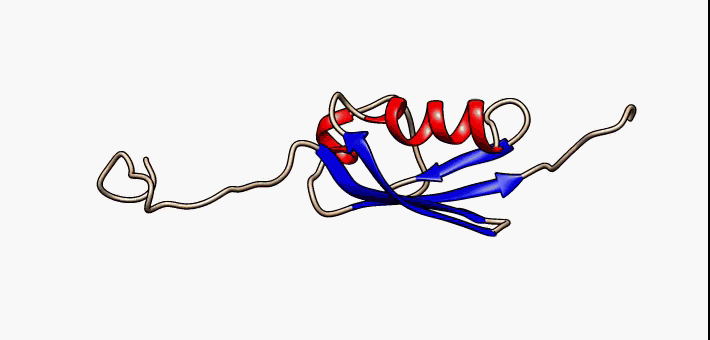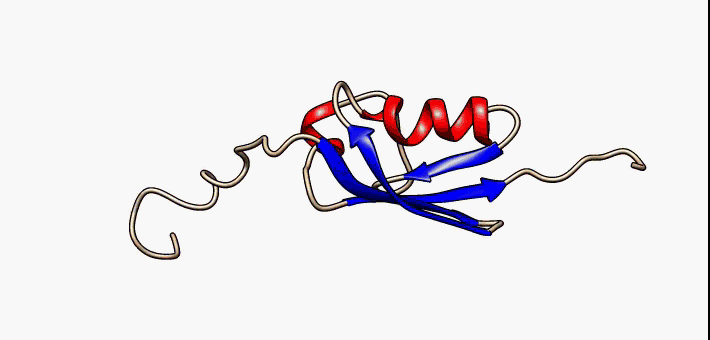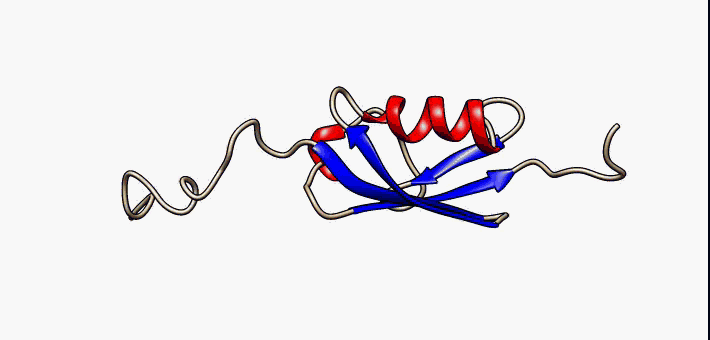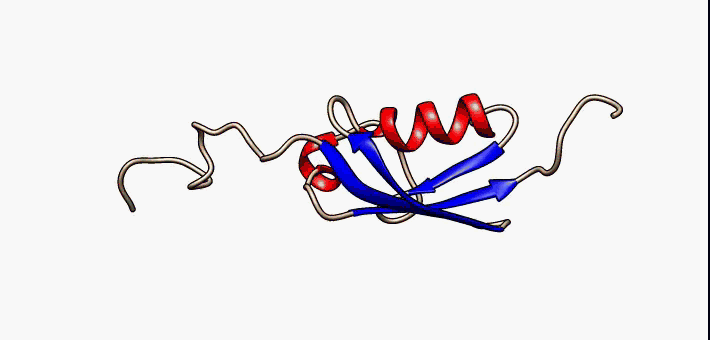
Molten Globule
In molecular biology, the term molten globule (MG) refers to protein states that are more or less compact (hence the "globule"), but are lacking the specific tight packing of amino acid residues which creates the solid state-like tertiary structure of completely folded proteins (hence the "molten"). Protein folding is navigated by a dynamic interplay of secondary and tertiary interactions. Two extreme folding pathway models have been formulated. In the first - the framework model - rapidly formed secondary structure elements assemble into a native tertiary structure. In the second - the hydrophobic collapse model - the formation of a loosely packed tertiary structure precedes secondary structure acquisition. A nucleation-condensation mechanism involving concomitant formation of short and long-range interactions combines features of both extreme models and thereby represents a unifying mechanism of protein folding. During folding, proteins span a continuum of conformers starting ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Molecular Biology
Molecular biology is a branch of biology that seeks to understand the molecule, molecular basis of biological activity in and between Cell (biology), cells, including biomolecule, biomolecular synthesis, modification, mechanisms, and interactions. Though cells and other microscopic structures had been observed in living organisms as early as the 18th century, a detailed understanding of the mechanisms and interactions governing their behavior did not emerge until the 20th century, when technologies used in physics and chemistry had advanced sufficiently to permit their application in the biological sciences. The term 'molecular biology' was first used in 1945 by the English physicist William Astbury, who described it as an approach focused on discerning the underpinnings of biological phenomena—i.e. uncovering the physical and chemical structures and properties of biological molecules, as well as their interactions with other molecules and how these interactions explain observ ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Folding Funnel
The folding funnel hypothesis is a specific version of the energy landscape theory of protein folding, which assumes that a protein's native state corresponds to its free energy minimum under the solution conditions usually encountered in cells. Although energy landscapes may be "rough", with many non-native local minima in which partially folded proteins can become trapped, the folding funnel hypothesis assumes that the native state is a deep free energy minimum with steep walls, corresponding to a single well-defined tertiary structure. The term was introduced by Ken A. Dill in a 1987 article discussing the stabilities of globular proteins. The folding funnel hypothesis is closely related to the hydrophobic collapse hypothesis, under which the driving force for protein folding is the stabilization associated with the sequestration of hydrophobic amino acid side chains in the interior of the folded protein. This allows the water solvent to maximize its entropy, lowering the ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Protein Structure
Protein structure is the three-dimensional arrangement of atoms in an amino acid-chain molecule. Proteins are polymers specifically polypeptides formed from sequences of amino acids, which are the monomers of the polymer. A single amino acid monomer may also be called a ''residue'', which indicates a repeating unit of a polymer. Proteins form by amino acids undergoing condensation reactions, in which the amino acids lose one water molecule per reaction in order to attach to one another with a peptide bond. By convention, a chain under 30 amino acids is often identified as a peptide, rather than a protein. To be able to perform their biological function, proteins fold into one or more specific spatial conformations driven by a number of non-covalent interactions, such as hydrogen bonding, ionic interactions, Van der Waals forces, and hydrophobic packing. To understand the functions of proteins at a molecular level, it is often necessary to determine their three-dimensiona ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Biomolecular Condensate
In biochemistry, biomolecular condensates are a class of membrane-less organelles and organelle subdomains, which carry out specialized functions within the cell. Unlike many organelles, biomolecular condensate composition is not controlled by a bounding membrane. Instead, condensates can form and maintain organization through a range of different processes, the most well-known of which is phase separation of proteins, RNA, and other biopolymers into either colloidal emulsions, gels, liquid crystals, solid crystals, or aggregates within cells. History Micellar theory The micellar theory of Carl Nägeli was developed from his detailed study of starch granules in 1858. Amorphous substances such as starch and cellulose were proposed to consist of building blocks, packed in a loosely crystalline array to form what he later termed "micelles". Water could penetrate between the micelles, and new micelles could form in the interstices between old micelles. The swelling of st ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrophobic Collapse
Hydrophobic collapse is a proposed process for the production of the 3-D conformation adopted by polypeptides and other molecules in polar solvents. The theory states that the nascent polypeptide forms initial secondary structure ( ɑ-helices and β-strands) creating localized regions of predominantly hydrophobic residues. The polypeptide interacts with water, thus placing thermodynamic pressures on these regions which then aggregate or "collapse" into a tertiary conformation with a hydrophobic core. Incidentally, polar residues interact favourably with water, thus the solvent-facing surface of the peptide is usually composed of predominantly hydrophilic regions. Hydrophobic collapse may also reduce the affinity of conformationally flexible drugs to their protein targets by reducing the net hydrophobic contribution to binding by self association of different parts of the drug while in solution. Conversely rigid scaffolds (also called privileged structures) that resist hydro ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Fuzzy Complex
Fuzzy or Fuzzies may refer to: Music * Fuzzy (band) Fuzzy was an American indie pop band based in Boston during the 1990s. The band was composed of singer-guitarists Hilken Mancini and Chris Toppin, and bassist Winston Braman. The drummer role was filled by David Ryan of The Lemonheads, Lemonhead ..., a 1990s Boston indie pop band * Fuzzy (composer), Danish composer Jens Vilhelm Pedersen (born 1939) * ''Fuzzy'' (album), 1993 debut album of American rock band Grant Lee Buffalo * "Fuzzy", a song from the 2009 '' Collective Soul'' album by Collective Soul * "Fuzzy", a song from '' Poppy.Computer'', the debut 2017 album by Poppy * Fuzzy, an Australian events company that organises Listen Out, a multi-city Australian music festival Nickname * Faustina Agolley (born 1984), Australian television presenter, host of the Australian television show ''Video Hits'' * Fuzzy Haskins (1941–2023), American singer and guitarist with the doo-wop group Parliament-Funkadelic * Fuzzy Hufft ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Folding Funnel
The folding funnel hypothesis is a specific version of the energy landscape theory of protein folding, which assumes that a protein's native state corresponds to its free energy minimum under the solution conditions usually encountered in cells. Although energy landscapes may be "rough", with many non-native local minima in which partially folded proteins can become trapped, the folding funnel hypothesis assumes that the native state is a deep free energy minimum with steep walls, corresponding to a single well-defined tertiary structure. The term was introduced by Ken A. Dill in a 1987 article discussing the stabilities of globular proteins. The folding funnel hypothesis is closely related to the hydrophobic collapse hypothesis, under which the driving force for protein folding is the stabilization associated with the sequestration of hydrophobic amino acid side chains in the interior of the folded protein. This allows the water solvent to maximize its entropy, lowering the ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Intrinsically Disordered Proteins
In molecular biology, an intrinsically disordered protein (IDP) is a protein that lacks a fixed or ordered protein tertiary structure, three-dimensional structure, typically in the absence of its macromolecular interaction partners, such as other proteins or RNA. IDPs range from fully unstructured to partially structured and include random coil, molten globule-like Protein aggregation, aggregates, or flexible linkers in large multi-Protein domain, domain proteins. They are sometimes considered as a separate class of proteins along with globular protein, globular, fibrous protein, fibrous and membrane proteins. IDPs are a very large and functionally important class of proteins. They are most numerous in Eukaryote, eukaryotes, with an estimated 30-40% of residues in the eukaryotic proteome located in disordered regions. Disorder is present in around 70% of proteins, either in the form of disordered tails or flexible linkers. Proteins can also be entirely disordered and lack a define ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dead-end Elimination
The dead-end elimination algorithm (DEE) is a method for minimizing a function over a discrete set of independent variables. The basic idea is to identify "dead ends", i.e., combinations of variables that are not necessary to define a global minimum because there is always a way of replacing such a combination with a better or equivalent one. Then we can refrain from further searching such combinations. Hence, dead-end elimination is the mirror image of dynamic programming, in which "good" combinations are identified and explored further. Although the method itself is general, it has been developed and applied mainly to the problems of predicting and designing the structures of proteins (and in this wise was cited in the Scientific Background to the 2024 Nobel Prize in Chemistry).https://www.nobelprize.org/uploads/2024/10/advanced-chemistryprize2024.pdf It is closely related to the notion of dominance in optimization also known as substitutability in a Constraint Satisfactio ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Protein Design
Protein design is the rational design of new protein molecules to design novel activity, behavior, or purpose, and to advance basic understanding of protein function. Proteins can be designed from scratch (''de novo'' design) or by making calculated variants of a known protein structure and its sequence (termed ''protein redesign''). Rational protein design approaches make protein-sequence predictions that will fold to specific structures. These predicted sequences can then be validated experimentally through methods such as peptide synthesis, site-directed mutagenesis, or artificial gene synthesis. Rational protein design dates back to the mid-1970s. Recently, however, there were numerous examples of successful rational design of water-soluble and even transmembrane peptides and proteins, in part due to a better understanding of different factors contributing to protein structure stability and development of better computational methods. Overview and history The goal in ratio ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
De Novo Synthesis
In chemistry, ''de novo'' synthesis () is the synthesis of complex molecules from simple molecules such as sugars or amino acids, as opposed to recycling after partial degradation. For example, nucleotides are not needed in the diet as they can be constructed from small precursor molecules such as formate and aspartate. Methionine, on the other hand, is needed in the diet because while it can be degraded to and then regenerated from homocysteine, it cannot be synthesized ''de novo''. Nucleotide ''De novo'' pathways of nucleotides do not use free bases: adenine (abbreviated as A), guanine (G), cytosine (C), thymine (T), or uracil (U). The purine ring is built up one atom or a few atoms at a time and attached to ribose throughout the process. Pyrimidine ring is synthesized as orotate and attached to ribose phosphate and later converted to common pyrimidine nucleotides. Cholesterol Cholesterol is an essential structural component of animal cell membranes. Cholesterol a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Kinetic Energy
In physics, the kinetic energy of an object is the form of energy that it possesses due to its motion. In classical mechanics, the kinetic energy of a non-rotating object of mass ''m'' traveling at a speed ''v'' is \fracmv^2.Resnick, Robert and Halliday, David (1960) ''Physics'', Section 7-5, Wiley International Edition The kinetic energy of an object is equal to the work, or force ( F) in the direction of motion times its displacement ( s), needed to accelerate the object from rest to its given speed. The same amount of work is done by the object when decelerating from its current speed to a state of rest. The SI unit of energy is the joule, while the English unit of energy is the foot-pound. In relativistic mechanics, \fracmv^2 is a good approximation of kinetic energy only when ''v'' is much less than the speed of light. History and etymology The adjective ''kinetic'' has its roots in the Greek word κίνησις ''kinesis'', meaning "motion". The dichoto ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |