|
Metallate
Metallate or metalate is the name given to any complex anion containing a metal ligated to several atoms or small groups. Typically, the metal will be one of the transition elements and the ligand will be oxygen or another chalcogenide or a cyanide group (though others are known). The chalcogenide metallates are known as oxometallates, thiometallates, selenometallates and tellurometallates; the cyanide metallates are known as cyanometallates. Oxometallates include permanganate (), chromate () and vanadate ( or ). Thiometallates include tetrathiovanadate (), tetrathiomolybdate (), tetrathiotungstate () and similar ions. Cyanometallates include ferricyanide and ferrocyanide. Metallate is also used as a verb by bioinorganic chemistry Bioinorganic chemistry is a field that examines the role of metals in biology Biology is the scientific study of life. It is a natural science with a broad scope but has several unifying themes that tie it together as a single, cohe ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Anion
An ion () is an atom or molecule with a net electrical charge. The charge of an electron is considered to be negative by convention and this charge is equal and opposite to the charge of a proton, which is considered to be positive by convention. The net charge of an ion is not zero because its total number of electrons is unequal to its total number of protons. A cation is a positively charged ion with fewer electrons than protons while an anion is a negatively charged ion with more electrons than protons. Opposite electric charges are pulled towards one another by electrostatic force, so cations and anions attract each other and readily form ionic compounds. Ions consisting of only a single atom are termed atomic or monatomic ions, while two or more atoms form molecular ions or polyatomic ions. In the case of physical ionization in a fluid (gas or liquid), "ion pairs" are created by spontaneous molecule collisions, where each generated pair consists of a free electron ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cyanometallate
Cyanometallates or cyanometalates are a class of coordination compounds, most often consisting only of cyanide ligands. Most are anions. Cyanide is a highly basic and small ligand, hence it readily saturates the coordination sphere of metal ions. The resulting cyanometallate anions are often used as building blocks for more complex structures called coordination polymers, the best known example of which is Prussian blue, a common dyestuff.*Dunbar, K. R. and Heintz, R. A., "Chemistry of Transition Metal Cyanide Compounds: Modern Perspectives", Progress in Inorganic Chemistry, 1997, 45, 283-391. Examples Homoleptic cyanometallates ''Homoleptic'' cyanometallates are complexes where the only ligand is cyanide. For transition metals, well known homoleptic cyanometallates are the hexacyanides. Hexacyanometalates are known for Ti(III), V(III), Cr(III), Cr(II), Mn(IV), Mn(III), Mn(II), Fe(II), Fe(III), Co(III), Ru(III), Ru(II), Os(III), and Os(II). Other more labile derivatives are al ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Bioinorganic Chemistry
Bioinorganic chemistry is a field that examines the role of metals in biology Biology is the scientific study of life. It is a natural science with a broad scope but has several unifying themes that tie it together as a single, coherent field. For instance, all organisms are made up of cells that process hereditar .... Bioinorganic chemistry includes the study of both natural phenomena such as the behavior of metalloproteins as well as artificially introduced metals, including those that are essential element, non-essential, in medicine and toxicology. Many biological processes such as cellular respiration, respiration depend upon molecules that fall within the realm of inorganic chemistry. The discipline also includes the study of inorganic models or mimics that imitate the behaviour of metalloproteins. As a mix of biochemistry and inorganic chemistry, bioinorganic chemistry is important in elucidating the implications of electron-transfer proteins, substrate bindin ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ferrocyanide
Ferrocyanide is the name of the anion cyanide.html" ;"title="e(cyanide">CN)6sup>4−. Salts of this coordination complex give yellow solutions. It is usually available as the salt potassium ferrocyanide, which has the formula K4Fe(CN)6. e(CN)6sup>4− is a diamagnetic species, featuring low-spin iron(II) center in an octahedral ligand environment. Although many salts of cyanide are highly toxic, ferro- and ferricyanides are less toxic because they tend not to release free cyanide. It is of commercial interest as a precursor to the pigment Prussian blue and, as its potassium salt, an anticaking agent. Reactions Treatment of ferrocyanide with ferric-containing salts gives the intensely coloured pigment Prussian blue (sometimes called ferric ferrocyanide and ferrous ferricyanide). Ferrocyanide reversibly oxidized by one electron, giving ferricyanide: : e(CN)6sup>4− ⇌ e(CN)6sup>3− + e− This conversion can be followed spectroscopically at 420 nm, since ferroc ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ferricyanide
Ferricyanide is the anion e(CN)6sup>3−. It is also called hexacyanoferrate(III) and in rare, but systematic nomenclature, hexacyanidoferrate(III). The most common salt of this anion is potassium ferricyanide, a red crystalline material that is used as an oxidant in organic chemistry. Properties e(CN)6sup>3− consists of a Fe3+ center bound in octahedral geometry to six cyanide ligands. The complex has Oh symmetry. The iron is low spin and easily reduced to the related ferrocyanide ion e(CN)6sup>4−, which is a ferrous (Fe2+) derivative. This redox couple is reversible and entails no making or breaking of Fe–C bonds: : e(CN)6sup>3− + e− ⇌ e(CN)6sup>4− This redox couple is a standard in electrochemistry. Compared to main group cyanides like potassium cyanide, ferricyanides are much less toxic because of the strong bond between the cyanide ion (CN− ) and the Fe3+. They do react with mineral acids, however, to release highly toxic hydrogen cyanide gas ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tetrathiomolybdate
Tetrathiomolybdate, also spelled tiomolibdate (USAN 280px, "Samguk Sagi" Book 04. Silla's Records. In 512, Usan-guk(于山國)was Ulleungdo(鬱陵島) Usan-guk, or the State of Usan, occupied Ulleung-do and the adjacent islands during the Korean Three Kingdoms period. According to the ...), is the anion of the following salts: * Ammonium tetrathiomolybdate, a building block in bioinorganic chemistry * Bis-choline tetrathiomolybdate, a drug for the treatment of Wilson's disease {{sia Thiometallates ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Vanadate
In chemistry, a vanadate is an anionic coordination complex of vanadium. Often vanadate refers to oxoanions of vanadium, most of which exist in its highest oxidation state of +5. The complexes and are referred to as hexacyanovanadate(III) and nonachlorodivanadate(III), respectively. A simple vanadate ion is the tetrahedral orthovanadate anion, (which is also called vanadate(V)), which is present in e.g. sodium orthovanadate and in solutions of in strong base ( pH > 13). Conventionally this ion is represented with a single double bond, however this is a resonance form as the ion is a regular tetrahedron with four equivalent oxygen atoms. Additionally a range of polyoxovanadate ions exist which include discrete ions and "infinite" polymeric ions. There are also vanadates, such as rhodium vanadate, , which has a statistical rutile structure where the and ions randomly occupy the positions in the rutile lattice, that do not contain a lattice of cations and balancing ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
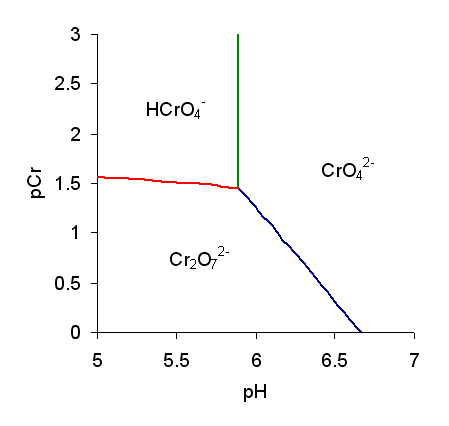
Chromate Ion
Chromate salts contain the chromate anion, . Dichromate salts contain the dichromate anion, . They are oxyanions of chromium in the +6 oxidation state and are moderately strong oxidizing agents. In an aqueous solution, chromate and dichromate ions can be interconvertible. Chemical properties Potassium-chromate-sample.jpg, potassium chromate Potassium-dichromate-sample.jpg, potassium dichromate Chromates react with hydrogen peroxide, giving products in which peroxide, , replaces one or more oxygen atoms. In acid solution the unstable blue peroxo complex Chromium(VI) oxide peroxide, CrO(O2)2, is formed; it is an uncharged covalent molecule, which may be extracted into ether. Addition of pyridine results in the formation of the more stable complex CrO(O2)2py. Acid–base properties In aqueous solution, chromate and dichromate anions exist in a chemical equilibrium. :2 + 2 H+ + H2O The predominance diagram shows that the position of the equilibrium depends ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Permanganate
A permanganate () is a chemical compound containing the manganate(VII) ion, , the conjugate base of permanganic acid. Because the manganese atom is in the +7 oxidation state, the permanganate(VII) ion is a strong oxidizing agent. The ion is a transition metal oxo complex with tetrahedral geometry. Permanganate solutions are purple in color and are stable in neutral or slightly alkaline media. The exact chemical reaction is dependent upon the organic contaminants present and the oxidant utilized. For example, trichloroethane (C2H3Cl3) is oxidized by permanganate ions to form carbon dioxide (CO2), manganese dioxide (MnO2), hydrogen ions (H+), and chloride ions (Cl−). :8 + 3 → 6 + 8 + + 4 + 9 In an acidic solution, permanganate(VII) is reduced to the pale pink +2 oxidation state of the manganese(II) (Mn2+) ion. :8 + + 5 e− → Mn2+ + 4 H2O In a strongly basic solution, permanganate(VII) is reduced to the green +6 oxidation state of the manganate ion, . ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |