|
Lupinine Graphic 1
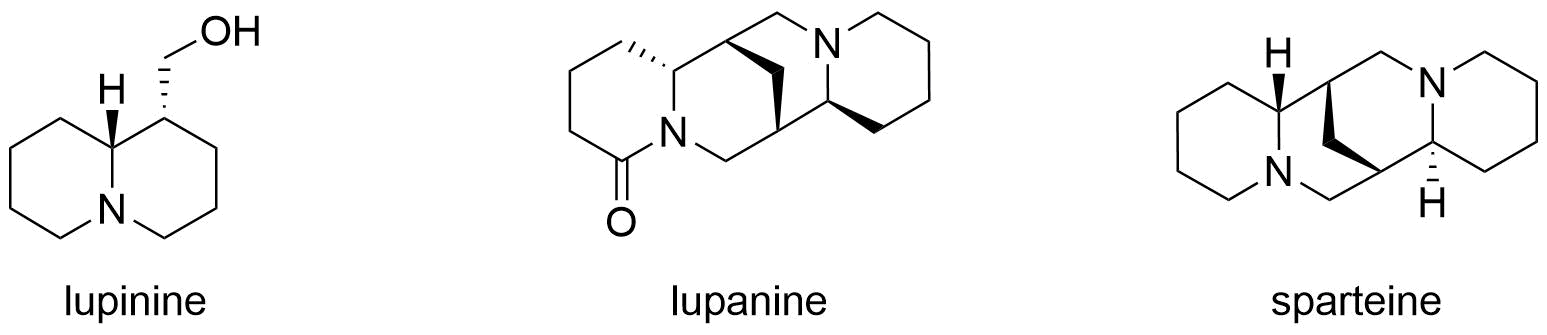
Lupinine is a Quinolizidine alkaloids, quinolizidine alkaloid present in the genus ''Lupinus'' (colloquially referred to as lupins) of the flowering plant family Fabaceae. The scientific literature contains many reports on the isolation and synthesis of this compound as well as a vast number of studies on its biosynthesis from its natural precursor, lysine. Studies have shown that lupinine hydrochloride is a mildly toxic acetylcholinesterase inhibitor and that lupinine has an inhibitory effect on acetylcholine receptors. The characteristically bitter taste of lupin beans, which come from the seeds of ''Lupinus'' plants, is attributable to the quinolizidine alkaloids which they contain, rendering them unsuitable for human and animal consumption unless handled properly. However, because lupin beans have potential nutritional value due to their high protein content, efforts have been made to reduce their alkaloid content through the development of "sweet" varieties of ''Lupinus''. To ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Quinolizidine Alkaloids
Quinolizidine alkaloids are natural products that have a quinolizidine structure; this includes the lupine alkaloids. Occurrence Quinolizidine alkaloids can be found in the plant family of legumes, especially in papilionaceous plants. While the lupine alkaloids (following their name) can be found in lupines, tinctorin, for example, was isolated from the dyer's broom. Examples More than 200 quinolizidine alkaloids are known which can be classified into 6 structural types: * the lupinine type with 34 known structures, including lupinine and its derivatives * the camoensine type with 6 known structures, including camoensin * the spartein type with 66 structures, including sparteine, lupanine, angustifoline * the α-pyridone type with 25 structures, including anagyrine and cytisine * the matrine type with 31 structures, including matrine * and the ormosanin type with 19 structures, including ormosanine. (–)-Lupinine Structural Formula V2.svg, Lupinine, (–)-lupinine (6R,7 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Acetylcholinesterase
Acetylcholinesterase (HUGO Gene Nomenclature Committee, HGNC symbol ACHE; EC 3.1.1.7; systematic name acetylcholine acetylhydrolase), also known as AChE, AChase or acetylhydrolase, is the primary cholinesterase in the body. It is an enzyme that catalysis, catalyzes the breakdown of acetylcholine and some other choline esters that function as neurotransmitters: : acetylcholine + H2O = choline + acetate It is found at mainly neuromuscular junctions and in chemical synapses of the cholinergic type, where its activity serves to terminate cholinergic neurotransmission, synaptic transmission. It belongs to the carboxylesterase family of enzymes. It is the primary target of inhibition by organophosphorus compounds such as nerve agents and pesticides. Enzyme structure and mechanism AChE is a hydrolase that hydrolyzes choline esters. It has a very high catalytic activity—each molecule of AChE degrades about 5,000 molecules of acetylcholine (ACh) per second, approaching the limit ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Alkaloid
Alkaloids are a broad class of natural product, naturally occurring organic compounds that contain at least one nitrogen atom. Some synthetic compounds of similar structure may also be termed alkaloids. Alkaloids are produced by a large variety of organisms including bacteria, fungus, fungi, Medicinal plant, plants, and animals. They can be purified from crude extracts of these organisms by acid-base extraction, or solvent extractions followed by silica-gel column chromatography. Alkaloids have a wide range of pharmacology, pharmacological activities including antimalarial medication, antimalarial (e.g. quinine), asthma, antiasthma (e.g. ephedrine), chemotherapy, anticancer (e.g. omacetaxine mepesuccinate, homoharringtonine), cholinomimetic (e.g. galantamine), vasodilation, vasodilatory (e.g. vincamine), Antiarrhythmic agent, antiarrhythmic (e.g. quinidine), analgesic (e.g. morphine), antibacterial (e.g. chelerythrine), and anti-diabetic, antihyperglycemic activities (e.g. berb ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Quinolizidine
Quinolizidine (norlupinane, octahydro-2''H''-quinolizine) is a nitrogen-containing heterocyclic compound. Some alkaloids (e.g. cytisine and sparteine) are derivatives of quinolizidine. Quinolizidine alkaloids Quinolizidine alkaloids, such as nupharine and related chemicals, can be found in '' Nymphaea lotus'' and other species in the family Nymphaeaceae. Quinolizidine alkaloids are also found in genistoid legumes Legumes are plants in the pea family Fabaceae (or Leguminosae), or the fruit or seeds of such plants. When used as a dry grain for human consumption, the seeds are also called pulses. Legumes are grown agriculturally, primarily for human consu .... External links * * Synthesis: {{heterocyclic-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Lupinine Graphic 2
Lupinine is a quinolizidine alkaloid present in the genus ''Lupinus'' (colloquially referred to as lupins) of the flowering plant family Fabaceae. The scientific literature contains many reports on the isolation and synthesis of this compound as well as a vast number of studies on its biosynthesis from its natural precursor, lysine. Studies have shown that lupinine hydrochloride is a mildly toxic acetylcholinesterase inhibitor and that lupinine has an inhibitory effect on acetylcholine receptors. The characteristically bitter taste of lupin beans, which come from the seeds of ''Lupinus'' plants, is attributable to the quinolizidine alkaloids which they contain, rendering them unsuitable for human and animal consumption unless handled properly. However, because lupin beans have potential nutritional value due to their high protein content, efforts have been made to reduce their alkaloid content through the development of "sweet" varieties of ''Lupinus''. Toxicity Lupinine is a he ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
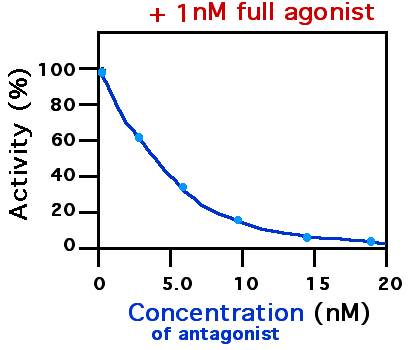
Receptor Antagonist
A receptor antagonist is a type of receptor ligand or drug that blocks or dampens a biological response by binding to and blocking a receptor rather than activating it like an agonist. Antagonist drugs interfere in the natural operation of receptor proteins.Pharmacology Guide: In vitro pharmacology: concentration-response curves ." '' GlaxoWellcome.'' Retrieved on December 6, 2007. They are sometimes called blockers; examples include alpha blockers, beta b ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Agonist
An agonist is a chemical that activates a Receptor (biochemistry), receptor to produce a biological response. Receptors are Cell (biology), cellular proteins whose activation causes the cell to modify what it is currently doing. In contrast, an Receptor antagonist, antagonist blocks the action of the agonist, while an inverse agonist causes an action opposite to that of the agonist. Etymology The word originates from the Ancient Greek, Greek word (''agōnistēs''), "contestant; champion; rival" < (''agōn''), "contest, combat; exertion, struggle" < (''agō''), "I lead, lead towards, conduct; drive." Types of agonists Receptor (biochemistry), Receptors can be activated by either endogenous agonists (such as hormones and neurotransmitters) or exogenous agonists (such as medication, drugs), resulting in a biological response. A physiological agonism an ...[...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
IC50
Half maximal inhibitory concentration (IC50) is a measure of the potency of a substance in inhibiting a specific biological or biochemical function. IC50 is a quantitative measure that indicates how much of a particular inhibitory substance (e.g. drug) is needed to inhibit, ''in vitro'', a given biological process or biological component by 50%. The biological component could be an enzyme, cell, cell receptor or microbe. IC50 values are typically expressed as molar concentration. IC50 is commonly used as a measure of antagonist drug potency in pharmacological research. IC50 is comparable to other measures of potency, such as EC50 for excitatory drugs. EC50 represents the dose or plasma concentration required for obtaining 50% of a maximum effect ''in vivo''. IC50 can be determined with functional assays or with competition binding assays. Sometimes, IC50 values are converted to the pIC50 scale. :\ce = -\log_ \ce Due to the minus sign, higher values of pIC50 indicate ex ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Neurotransmission
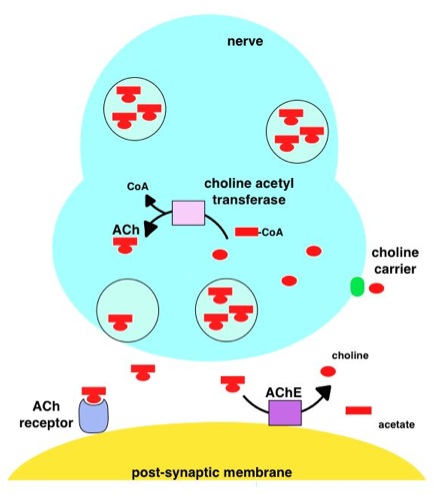
Neurotransmission (Latin: ''transmissio'' "passage, crossing" from ''transmittere'' "send, let through") is the process by which signaling molecules called neurotransmitters are released by the axon terminal of a neuron (the presynaptic neuron), and bind to and react with the receptors on the dendrites of another neuron (the postsynaptic neuron) a short distance away. Changes in the concentration of ions, such as Ca2+, Na+, K+, underlie both chemical and electrical activity in the process. The increase in calcium levels is essential and can be promoted by protons. A similar process occurs in retrograde neurotransmission, where the dendrites of the postsynaptic neuron release retrograde neurotransmitters (e.g., endocannabinoids; synthesized in response to a rise in intracellular calcium levels) that signal through receptors that are located on the axon terminal of the presynaptic neuron, mainly at GABAergic and glutamatergic synapses. Neurotransmission is regulated by severa ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nicotinic Acetylcholine Receptor
Nicotinic acetylcholine receptors, or nAChRs, are Receptor (biochemistry), receptor polypeptides that respond to the neurotransmitter acetylcholine. Nicotinic receptors also respond to drugs such as the agonist nicotine. They are found in the central and peripheral nervous system, muscle, and many other tissues of many organisms. At the neuromuscular junction they are the primary receptor in muscle for motor nerve-muscle communication that controls muscle contraction. In the peripheral nervous system: (1) they transmit outgoing signals from the presynaptic to the postsynaptic cells within the Sympathetic nervous system, sympathetic and parasympathetic nervous system, and (2) they are the receptors found on skeletal muscle that receive acetylcholine released to signal for muscular contraction. In the immune system, nAChRs regulate inflammatory processes and signal through distinct intracellular pathways. In insects, the cholinergic system is limited to the central nervous system. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Muscarinic Acetylcholine Receptor
Muscarinic acetylcholine receptors (mAChRs) are acetylcholine receptors that form G protein-coupled receptor, G protein-coupled receptor complexes in the cell membranes of certain neurons and other Cell (biology), cells. They play several roles, including acting as the main end-receptor stimulated by acetylcholine released from postganglionic fibers. They are mainly found in the parasympathetic nervous system, but also have a role in the sympathetic nervous system in the control of Sweat gland, sweat glands. Muscarinic receptors are so named because they are more sensitive to muscarine than to nicotine. Their counterparts are nicotinic acetylcholine receptors (nAChRs), receptor ion channels that are also important in the autonomic nervous system. Many drugs and other substances (for example pilocarpine and Hyoscine hydrobromide, scopolamine) manipulate these two distinct receptors by acting as selective agonists or receptor antagonist, antagonists. Function acetylcholine, Ac ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Anabasis Aphylla
''Anabasis aphylla'' is a species of flowering plant in the family Amaranthaceae, native to the region surrounding the Caspian Sea, Central Asia, and Xinjiang and western Gansu provinces of China. A many-branched shrub usually found growing in alluvial fans and dune swales, it is sometimes planted to catch blowing soil and stabilize sand dunes. The alkaloid anabasine was named for this toxic species, from which it was first isolated by Orechoff and Menschikoff in the year 1931. Anabasine was widely used as an insecticide in the former Soviet Union until 1970. Anabaseine Anabaseine (3,4,5,6-tetrahydro-2,3′-bipyridine) is an alkaloid toxin produced by ''Nemertines'' worms and ''Aphaenogaster'' ants. It is structurally similar to nicotine and anabasine. Similarly, it has been shown to act as an agonist on most ni ... and anabasamine are other alkaloids found to exist in the plant. References Amaranthaceae Flora of South European Russia Flora of the Caucasus Flora of I ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |