|
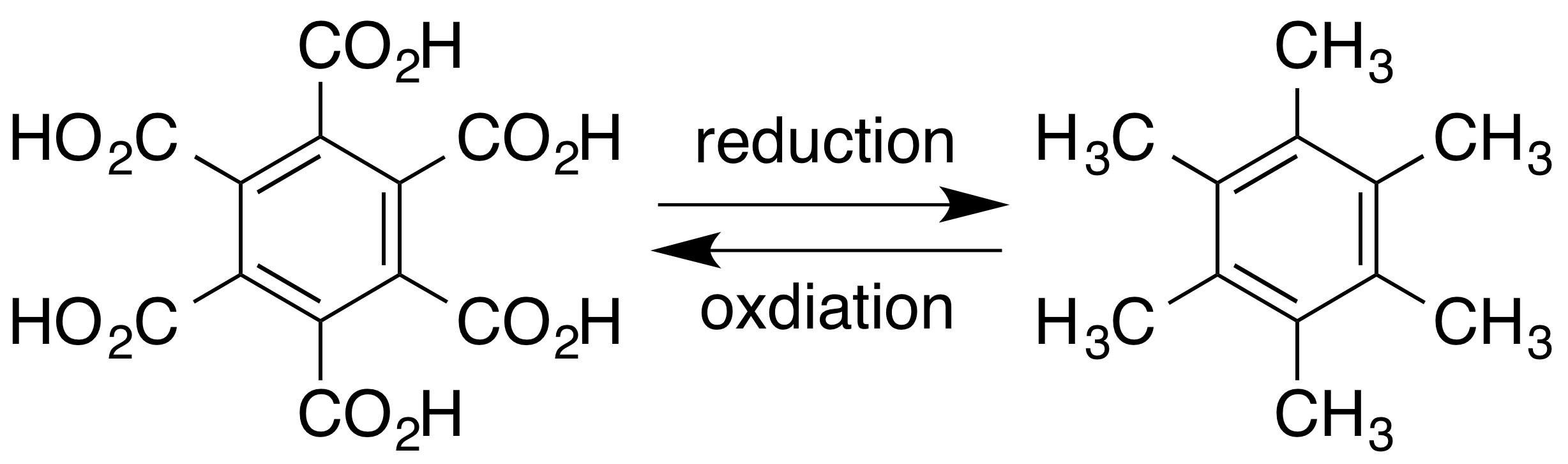
Hexamethylbenzene
Hexamethylbenzene, also known as mellitene, is a hydrocarbon with the molecular formula C12H18 and the condensed structural formula C6(CH3)6. It is an aromatic compound and a derivative of benzene, where benzene's six hydrogen atoms have each been replaced by a methyl group. In 1929 Kathleen Lonsdale reported the crystal structure of hexamethylbenzene, demonstrating that the central ring is hexagonal and flat and thereby ending an ongoing debate about the physical parameters of the benzene system. This was a historically significant result, both for the field of X-ray crystallography and for understanding aromaticity. The compound can be prepared by reacting phenol with methanol at elevated temperatures over a suitable solid catalyst such as alumina. The mechanism of the process has been studied extensively, with several intermediates having been identified. Alkyne trimerisation of dimethylacetylene also yields hexamethylbenzene in the presence of a suitable catalyst. Hexam ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Alkyne Trimerisation
In organic chemistry, an alkyne trimerisation is a +2+2nbsp; cycloaddition reaction in which three alkyne units () react to form a benzene ring. The reaction requires a metal catalyst. The process is of historic interest as well as being applicable to organic synthesis. Being a cycloaddition reaction, it has high atom economy. Many variations have been developed, including cyclisation of mixtures of alkynes and alkenes as well as alkynes and nitriles. Mechanism and stereochemistry Trimerisation of acetylene to benzene is highly exergonic, proceeding with a free energy change of 142 kcal/mol at room temperature. Kinetic barriers however prevent the reaction from proceeding smoothly. The breakthrough came in 1948, when Reppe and Schweckendiek reported their wartime results showing that nickel compounds are effective catalysts: : 3 RC2H -> C6R3H3 Since this discovery, many other cyclotrimerisations have been reported. Mechanism In terms of mechanism, the reactions ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Mellitic Acid
Mellitic acid, also called graphitic acid or benzenehexacarboxylic acid, is an acid first discovered in 1799 by Martin Heinrich Klaproth in the mineral mellite (honeystone), which is the aluminium salt of the acid. It crystallizes in fine silky needles and is soluble in water and alcohol. Preparation Mellitic acid may be prepared by warming mellite with ammonium carbonate, boiling off the excess of the ammonium salt, and adding ammonia to the solution. The precipitated alumina is filtered off, the filtrate evaporated, and the ammonium salt of the acid purified by recrystallization. The ammonium salt is then converted into the lead salt by precipitation with lead acetate, and the lead salt is then decomposed by hydrogen sulfide. The acid may also be prepared by the oxidation of pure carbon, graphite or hexamethylbenzene, by alkaline potassium permanganate in the cold, or by hot concentrated nitric acid. Reactions It is a very stable compound; chlorine, concentrated nit ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Benzene
Benzene is an organic chemical compound with the molecular formula C6H6. The benzene molecule is composed of six carbon atoms joined in a planar ring with one hydrogen atom attached to each. Because it contains only carbon and hydrogen atoms, benzene is classed as a hydrocarbon. Benzene is a natural constituent of petroleum and is one of the elementary petrochemicals. Due to the cyclic continuous pi bonds between the carbon atoms, benzene is classed as an aromatic hydrocarbon. Benzene is a colorless and highly flammable liquid with a sweet smell, and is partially responsible for the aroma of gasoline. It is used primarily as a precursor to the manufacture of chemicals with more complex structure, such as ethylbenzene and cumene, of which billions of kilograms are produced annually. Although benzene is a major industrial chemical, it finds limited use in consumer items because of its toxicity. History Discovery The word "''benzene''" derives from "''gum benzoin''" ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Organoruthenium Chemistry
Organoruthenium chemistry is the chemistry of organometallic compounds containing a carbon to ruthenium chemical bond. Several organoruthenium catalysts are of commercial interest and organoruthenium compounds have been considered for cancer therapy. The chemistry has some stoichiometric similarities with organoiron chemistry, as iron is directly above ruthenium in group 8 of the periodic table. The most important reagents for the introduction of ruthenium are ruthenium(III) chloride and triruthenium dodecacarbonyl. In its organometallic compounds, ruthenium is known to adopt oxidation states from -2 ( u(CO)4sup>2−) to +6 (uN(Me)4sup>−). Most common are those in the 2+ oxidation state, as illustrated below. File:Grubbs Catalyst 1st Generation.svg, 1st generation Grubbs catalyst File:ShvoCat.png, Shvo catalyst File:RuCymCl2.png, (cymene)ruthenium dichloride dimer File:Trirutheniumdodecacarbonyl.svg, triruthenium dodecacarbonyl. File:Chloro(cyclopentadienyl)bis(triphenylp ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
X-ray Crystallography
X-ray crystallography is the experimental science determining the atomic and molecular structure of a crystal, in which the crystalline structure causes a beam of incident X-rays to diffract into many specific directions. By measuring the angles and intensities of these diffracted beams, a crystallographer can produce a three-dimensional picture of the density of electrons within the crystal. From this electron density, the mean positions of the atoms in the crystal can be determined, as well as their chemical bonds, their crystallographic disorder, and various other information. Since many materials can form crystals—such as salts, metals, minerals, semiconductors, as well as various inorganic, organic, and biological molecules—X-ray crystallography has been fundamental in the development of many scientific fields. In its first decades of use, this method determined the size of atoms, the lengths and types of chemical bonds, and the atomic-scale differences among vari ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Kathleen Lonsdale
Dame Kathleen Lonsdale ( Yardley; 28 January 1903 – 1 April 1971) was an Irish-born British pacifist, prison reformer and crystallographer. She proved, in 1929, that the benzene ring is flat by using X-ray diffraction methods to elucidate the structure of hexamethylbenzene. She was the first to use Fourier spectral methods while solving the structure of hexachlorobenzene in 1931. During her career she attained several firsts for female scientists, including being one of the first two women elected a Fellow of the Royal Society (FRS) in 1945 (along with Marjory Stephenson), first woman tenured professor at University College London, first woman president of the International Union of Crystallography, and first woman president of the British Association for the Advancement of Science. Early life and education She was born Kathleen Yardley at Newbridge, County Kildare, Ireland, the tenth child of Harry Yardley, the town postmaster, and Jessie Cameron. Kathleen's family was ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
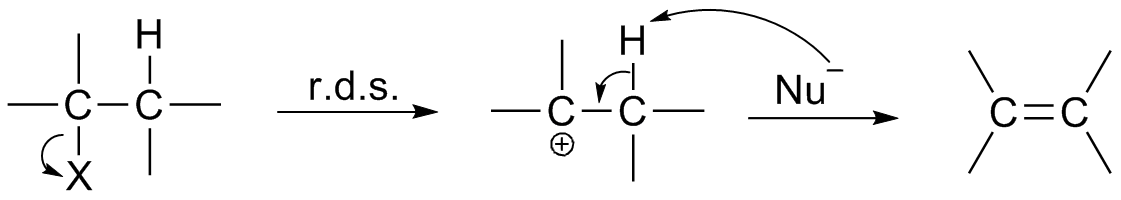
Reaction Mechanism
In chemistry, a reaction mechanism is the step by step sequence of elementary reactions by which overall chemical change occurs. A chemical mechanism is a theoretical conjecture that tries to describe in detail what takes place at each stage of an overall chemical reaction. The detailed steps of a reaction are not observable in most cases. The conjectured mechanism is chosen because it is thermodynamically feasible, and has experimental support in isolated intermediates (see next section) or other quantitative and qualitative characteristics of the reaction. It also describes each reactive intermediate, activated complex, and transition state, and which bonds are broken (and in what order), and which bonds are formed (and in what order). A complete mechanism must also explain the reason for the reactants and catalyst used, the stereochemistry observed in reactants and products, all products formed and the amount of each. The electron or arrow pushing method is often used ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Reaction Intermediate
In chemistry, a reaction intermediate or an intermediate is a molecular entity that is formed from the reactants (or preceding intermediates) but is consumed in further reactions in stepwise chemical reactions that contain multiple elementary steps. Intermediates are the reaction product of one elementary step, but do not appear in the chemical equation for an overall chemical equation. For example, consider this hypothetical stepwise reaction: :A + B -> C + D The reaction includes two elementary steps: :A + B -> X :X -> C + D In this example, X is a reaction intermediate. IUPAC definition The IUPAC Gold Book defines an ''intermediate'' as a compound that has a lifetime greater than a molecular vibration that is formed (directly or indirectly) from the reactants and reacts further to give (either directly or indirectly) the products of a chemical reaction. The lifetime condition distinguishes true, chemically distinct intermediates from vibrational states or such transiti ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Acetic Acid
Acetic acid , systematically named ethanoic acid , is an acidic, colourless liquid and organic compound with the chemical formula (also written as , , or ). Vinegar is at least 4% acetic acid by volume, making acetic acid the main component of vinegar apart from water and other trace elements. Acetic acid is the second simplest carboxylic acid (after formic acid). It is an important chemical reagent and industrial chemical, used primarily in the production of cellulose acetate for photographic film, polyvinyl acetate for wood glue, and synthetic fibres and fabrics. In households, diluted acetic acid is often used in descaling agents. In the food industry, acetic acid is controlled by the food additive code E260 as an acidity regulator and as a condiment. In biochemistry, the acetyl group, derived from acetic acid, is fundamental to all forms of life. When bound to coenzyme A, it is central to the metabolism of carbohydrates and fats. The global demand for aceti ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dimethylacetylene
2-Butyne (dimethylacetylene, crotonylene or but-2-yne) is an alkyne with chemical formula CH3C≡CCH3. Produced artificially, it is a colorless, volatile, pungent liquid at standard temperature and pressure. 2-Butyne is of interest to physical chemists because of its very low torsional barrier and the problem of determining that barrier using high-resolution infrared spectroscopy. Analysis of its spectrum leads to a determination that the torsional barrier is only 6 cm−1 (1.2 J or 72 J mol−1). However, it has not been determined whether the equilibrium structure is eclipsed (D3h) or staggered (D3d). Symmetry analysis using the Molecular Symmetry Group G36 shows that one would need to analyse its high resolution rotation-vibration Raman spectrum to determine its equilibrium structure. 2-Butyne (''dimethylethyne'') forms with 5-decyne (''dibutylethyne''), 4-octyne (''dipropylethyne'') and 3-hexyne (''diethylethyne'') a group of symmetric alkynes. Synthesis 2-Butyne can b ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Methanol
Methanol (also called methyl alcohol and wood spirit, amongst other names) is an organic chemical and the simplest aliphatic alcohol, with the formula C H3 O H (a methyl group linked to a hydroxyl group, often abbreviated as MeOH). It is a light, volatile, colourless, flammable liquid with a distinctive alcoholic odour similar to that of ethanol (potable alcohol). A polar solvent, methanol acquired the name wood alcohol because it was once produced chiefly by the destructive distillation of wood. Today, methanol is mainly produced industrially by hydrogenation of carbon monoxide. Methanol consists of a methyl group linked to a polar hydroxyl group. With more than 20 million tons produced annually, it is used as a precursor to other commodity chemicals, including formaldehyde, acetic acid, methyl tert-butyl ether, methyl benzoate, anisole, peroxyacids, as well as a host of more specialised chemicals. Occurrence Small amounts of methanol are present in normal, h ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |

2.png)
2redox.png)