|
Europium(II) Fluoride
Europium(II) fluoride is an inorganic compound with a chemical formula EuF2. It was first synthesized in 1937.Zhao Yongzhi, Ma Ying, Hou Shaochun, Zhang Wenjuan, Wang Jingjing, Ding Yanrong, Hao Yifan氟化亚铕研究现状 (''lit''. Research Progress of Europium(II) Fluoride). ''Chinese Rare Earths'', 2017. 38 (5): 134-140. (in Chinese) Production Europium(II) fluoride can be produced by reducing europium(III) fluoride with metallic europium or hydrogen gas.Georg Brauer: ''Handbuch der Präparativen Anorganischen Chemie''. 3., umgearb. Auflage. Band I. Enke, Stuttgart 1975, , S. 255. :\mathrm :\mathrm Properties Europium(II) fluoride is a bright yellowish solid with a fluorite structure. EuF2 can be used to dope a trivalent rare-earth fluoride, such as lanthanum fluoride, LaF3, to create a vacancy-filled structure with increased conductivity over a pure crystal. Such a crystal can be used as a fluoride-specific semipermeable membrane in a fluoride selective electrode to detect t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Fluorite Structure
The fluorite structure refers to a common motif for compounds with the formula MX2. The X ions occupy the eight tetrahedral interstitial sites whereas M ions occupy the regular sites of a face-centered cubic In crystallography, the cubic (or isometric) crystal system is a crystal system where the unit cell is in the shape of a cube. This is one of the most common and simplest shapes found in crystals and minerals. There are three main varieties o ... (FCC) structure. Many compounds, notably the common mineral fluorite (CaF2), adopt this structure. Many compounds with formula M2X have an antifluorite structure. In these the locations of the anions and cations are reversed relative to fluorite (an anti-structure); the anions occupy the FCC regular sites whereas the cations occupy the tetrahedral interstitial sites. For example, magnesium silicide, Mg2Si, has a lattice parameter of 6.338 Å with magnesium cations occupying the tetrahedral interstitial sites, in which each s ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrogen Gas
Hydrogen is a chemical element; it has symbol H and atomic number 1. It is the lightest and most abundant chemical element in the universe, constituting about 75% of all normal matter. Under standard conditions, hydrogen is a gas of diatomic molecules with the formula , called dihydrogen, or sometimes hydrogen gas, molecular hydrogen, or simply hydrogen. Dihydrogen is colorless, odorless, non-toxic, and highly combustible. Stars, including the Sun, mainly consist of hydrogen in a plasma state, while on Earth, hydrogen is found as the gas (dihydrogen) and in molecular forms, such as in water and organic compounds. The most common isotope of hydrogen (H) consists of one proton, one electron, and no neutrons. Hydrogen gas was first produced artificially in the 17th century by the reaction of acids with metals. Henry Cavendish, in 1766–1781, identified hydrogen gas as a distinct substance and discovered its property of producing water when burned; hence its name means 'wa ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Lanthanide Halides
The lanthanide () or lanthanoid () series of chemical elements comprises at least the 14 metallic chemical elements with atomic numbers 57–70, from lanthanum through ytterbium. In the periodic table, they fill the 4f orbitals. Lutetium (element 71) is also sometimes considered a lanthanide, despite being a d-block element and a transition metal. The informal chemical symbol Ln is used in general discussions of lanthanide chemistry to refer to any lanthanide. All but one of the lanthanides are f-block elements, corresponding to the filling of the 4f electron shell. Lutetium is a d-block element (thus also a transition metal), and on this basis its inclusion has been questioned; however, like its congeners scandium and yttrium in group 3, it behaves similarly to the other 14. The term rare-earth element or rare-earth metal is often used to include the stable group 3 elements Sc, Y, and Lu in addition to the 4f elements. All lanthanide elements form trivalent cations, Ln3+, wh ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Fluorides
Fluoride (). According to this source, is a possible pronunciation in British English. is an inorganic, monatomic anion of fluorine, with the chemical formula (also written ), whose salts are typically white or colorless. Fluoride salts typically have distinctive bitter tastes, and are odorless. Its salts and minerals are important chemical reagents and industrial chemicals, mainly used in the production of hydrogen fluoride for fluorocarbons. Fluoride is classified as a weak base since it only partially associates in solution, but concentrated fluoride is corrosive and can attack the skin. Fluoride is the simplest fluorine anion. In terms of charge and size, the fluoride ion resembles the hydroxide ion. Fluoride ions occur on Earth in several minerals, particularly fluorite, but are present only in trace quantities in bodies of water in nature. Nomenclature Fluorides include compounds that contain ionic fluoride and those in which fluoride does not dissociate. The nome ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Europium(II) Compounds
Europium is a chemical element; it has symbol Eu and atomic number 63. It is a silvery-white metal of the lanthanide series that reacts readily with air to form a dark oxide coating. Europium is the most chemically reactive, least dense, and softest of the lanthanides. It is soft enough to be cut with a knife. Europium was discovered in 1896, provisionally designated as Σ; in 1901, it was named after the continent of Europe. Europium usually assumes the oxidation state +3, like other members of the lanthanide series, but compounds having oxidation state +2 are also common. All europium compounds with oxidation state +2 are slightly reducing. Europium has no significant biological role and is relatively non-toxic compared to other heavy metals. Most applications of europium exploit the phosphorescence of europium compounds. Europium is one of the rarest of the rare-earth elements on Earth.Stwertka, Albert. ''A Guide to the Elements'', Oxford University Press, 1996, p. 156. Etymol ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Fluoride Selective Electrode
A fluoride selective electrode is a type of ion selective electrode sensitive to the concentration of the fluoride ion. A common example is the lanthanum fluoride electrode. Lanthanum fluoride electrode In the lanthanum fluoride electrode, the sensing element is a crystal of lanthanum fluoride (LaF3), doped with europium(II) fluoride (EuF2) to create lattice vacancies. Such a crystal is an ionic conductor by virtue of the mobility of fluoride ions which jump between lattice vacancies. An electrochemical cell may be constructed using such a crystal as a membrane separating two fluoride solutions. This cell acts as a concentration cell with transference where the fluoride transport number is 1. As transference of charge through the crystal is almost exclusively due to fluoride, the electrode is highly specific to fluoride. The only ion which significantly interferes is hydroxide (OH−). Generally such "alkaline error" can be avoided by buffering the sample to a pH below 7. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
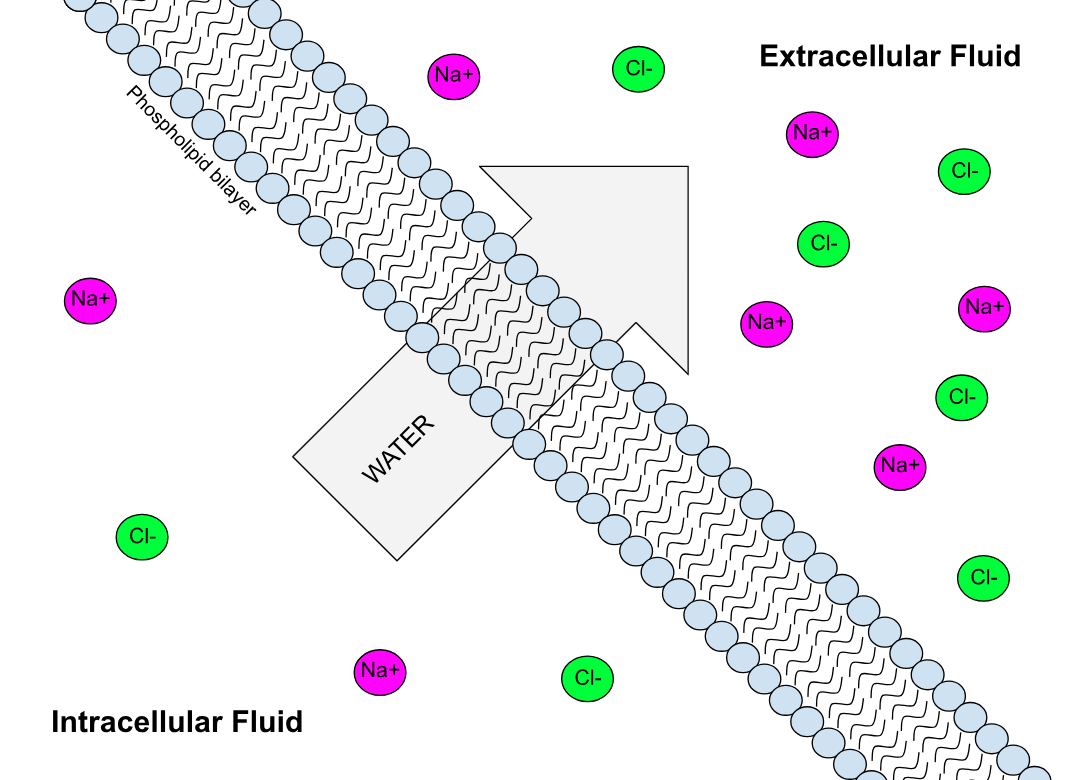
Semipermeable Membrane
Semipermeable membrane is a type of synthetic or biologic, polymeric membrane that allows certain molecules or ions to pass through it by osmosis. The rate of passage depends on the pressure, concentration, and temperature of the molecules or solutes on either side, as well as the permeability of the membrane to each solute. Depending on the membrane and the solute, permeability may depend on solute size, solubility, properties, or chemistry. How the membrane is constructed to be selective in its permeability will determine the rate and the permeability. Many natural and synthetic materials which are rather thick are also semipermeable. One example of this is the thin film on the inside of an egg. Biological membranes are selectively permeable, with the passage of molecules controlled by facilitated diffusion, passive transport or active transport regulated by proteins embedded in the membrane. Biological membranes Phospholipid bilayer A phospholipid bilayer is an ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Lanthanum Fluoride
Lanthanum trifluoride is a refractory ionic compound of lanthanum and fluorine. The chemical formula is . The LaF3 structure Bonding is ionic with lanthanum highly coordinated. The cation sits at the center of a trigonal prism. Nine fluorine atoms are close: three at the bottom corners of the trigonal prism, three in the faces of the trigonal prism, and three at top corners of the trigonal prism. There are also two fluorides a little further away above and below the prism. The cation can be considered 9-coordinate or 11-coordinate. At 300 K, the structure allows the formation of Schottky defects with an activation energy of 0.07 eV, and free flow of fluoride ions with an activation energy of 0.45 eV, making the crystal unusually electrically conductive. The larger sized rare earth elements (lanthanides), which are those with smaller atomic number, also form trifluorides with the LaF3 structure. Some actinides do as well. Applications This white salt is somet ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Fluorite Structure
The fluorite structure refers to a common motif for compounds with the formula MX2. The X ions occupy the eight tetrahedral interstitial sites whereas M ions occupy the regular sites of a face-centered cubic In crystallography, the cubic (or isometric) crystal system is a crystal system where the unit cell is in the shape of a cube. This is one of the most common and simplest shapes found in crystals and minerals. There are three main varieties o ... (FCC) structure. Many compounds, notably the common mineral fluorite (CaF2), adopt this structure. Many compounds with formula M2X have an antifluorite structure. In these the locations of the anions and cations are reversed relative to fluorite (an anti-structure); the anions occupy the FCC regular sites whereas the cations occupy the tetrahedral interstitial sites. For example, magnesium silicide, Mg2Si, has a lattice parameter of 6.338 Å with magnesium cations occupying the tetrahedral interstitial sites, in which each s ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Europium
Europium is a chemical element; it has symbol Eu and atomic number 63. It is a silvery-white metal of the lanthanide series that reacts readily with air to form a dark oxide coating. Europium is the most chemically reactive, least dense, and softest of the lanthanides. It is soft enough to be cut with a knife. Europium was discovered in 1896, provisionally designated as Σ; in 1901, it was named after the continent of Europe. Europium usually assumes the oxidation state +3, like other members of the lanthanide series, but compounds having oxidation state +2 are also common. All europium compounds with oxidation state +2 are slightly reducing. Europium has no significant biological role and is relatively non-toxic compared to other heavy metals. Most applications of europium exploit the phosphorescence of europium compounds. Europium is one of the rarest of the rare-earth elements on Earth.Stwertka, Albert. ''A Guide to the Elements'', Oxford University Press, 1996, p. 156. Ety ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Europium(II) Bromide
Europium(II) bromide is a crystalline compound of one europium atom and two bromine atoms. Europium(II) bromide is a white powder at room temperature, and odorless. Europium dibromide is hygroscopic Hygroscopy is the phenomenon of attracting and holding water molecules via either absorption (chemistry), absorption or adsorption from the surrounding Natural environment, environment, which is usually at normal or room temperature. If water mol .... Reactions Europium(II) bromide is known to be involved in three reactions: :2 EuBr3 + Eu → 3 EuBr2 (requires a temperature of 800-900 °C) :2 EuBr3 → 2 EuBr2 + Br2 (requires a temperature of 900-1000 °C) :Eu + HgBr2 → EuBr2 + Hg (requires a temperature of 700-800 °C) References {{Lanthanide halides Bromides Europium(II) compounds Lanthanide halides ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Europium(III) Fluoride
Europium(III) fluoride is an inorganic compound with a chemical formula EuF3. Production Europium(III) fluoride can be produced by reacting europium(III) nitrate and ammonium fluoride: : Eu(NO3)3 + 3 NH4F → EuF3 + 3 NH4NO3 Europium(III) fluoride nanoparticles can be synthesized by microwave irradiation of europium(III) acetate in an ionic liquid that has tetrafluoroborate Tetrafluoroborate is the anion . This tetrahedral species is isoelectronic with tetrafluoroberyllate (), tetrafluoromethane (CF4), and tetrafluoroammonium () and is valence isoelectronic with many stable and important species including the perc ... as the anion. References {{Lanthanide halides Europium(III) compounds Fluorides Lanthanide halides ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |