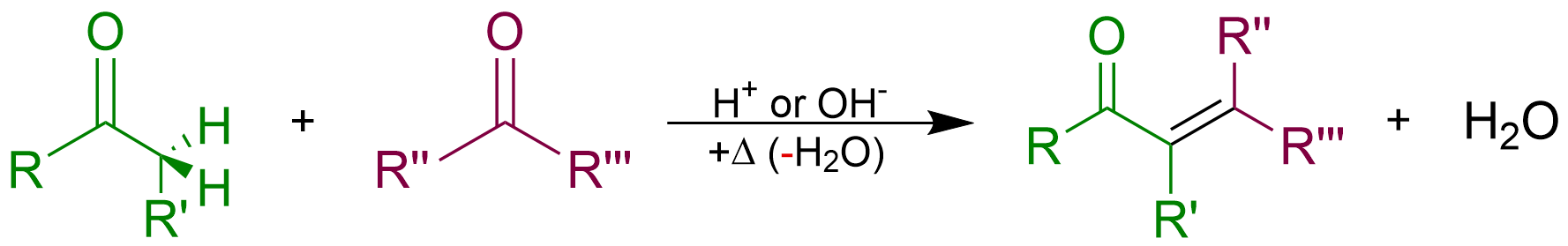|
Dicinnamalacetone Rxn 2
Dicinnamalacetone is a conjugated organic compound. It is used as an indicator for the presence of hydrogen halides in solvents, and its preparation is used as an example of the aldol condensation in organic chemistry teaching labs. Preparation Dicinnamalacetone (specifically the all-''trans'' isomer) may be prepared by the reaction of acetone and '' trans-''cinnamaldehyde catalyzed by a strong base, such as potassium hydroxide. The reaction is typically conducted in a mixture of water and ethanol. Clock reaction The preparation of dicinnamalacetone is an example of a clock reaction. Upon the addition of acetone to a solution of ''trans''-cinnamaldehyde and potassium hydroxide in ethanol and water, a precipitate of dicinnamalacetone forms spontaneously after a delay. The formation of dicinnamalacetone constitutes two separate aldol condensations. In the first, one molar equivalent of ''trans''-cinnamaldehyde and one molar equivalent of acetone condense Condensation ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Conjugated System
In physical organic chemistry, a conjugated system is a system of connected p-orbitals with delocalized electrons in a molecule, which in general lowers the overall energy of the molecule and increases Chemical stability, stability. It is Resonance (chemistry), conventionally represented as having alternating single and multiple covalent bond, bonds. Lone pairs, radical (chemistry), radicals or carbenium ions may be part of the system, which may be Cyclic molecule, cyclic, acyclic, Linear molecular geometry, linear or mixed. The term "conjugated" was coined in 1899 by the German chemist Johannes Thiele (chemist), Johannes Thiele. Conjugation is the orbital overlap, overlap of one p-orbital with another across an adjacent Sigma bond, σ bond (in transition metals, d-orbitals can be involved). A conjugated system has a region of overlapping p-orbitals, bridging the interjacent locations that simple diagrams illustrate as not having a π bond. They allow a delocalization of pi el ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chemical Clock
A chemical clock (or clock reaction) is a complex mixture of reacting chemical compounds in which the onset of an observable property (discoloration or coloration) occurs after a predictable induction time due to the presence of clock species at a detectable amount. In cases where one of the reagents has a visible color, crossing a concentration threshold can lead to an abrupt color change after a reproducible time lapse. Types Clock reactions may be classified into three or four types: Substrate-depletive clock reaction The simplest clock reaction featuring two reactions: :A → C (rate k1) :B + C → products (rate k2, fast) When substrate (B) is present, the clock species (C) is quickly consumed in the second reaction. Only when substrate B is all used up or depleted, species C can build up in amount causing the color to change. An example for this clock reaction is the sulfite/iodate reaction or iodine clock reaction, also known as Landolt's reaction. Sometimes, a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Reaction Rate
The reaction rate or rate of reaction is the speed at which a chemical reaction takes place, defined as proportional to the increase in the concentration of a product per unit time and to the decrease in the concentration of a reactant per unit time. Reaction rates can vary dramatically. For example, the oxidative rusting of iron under Earth's atmosphere is a slow reaction that can take many years, but the combustion of cellulose in a fire is a reaction that takes place in fractions of a second. For most reactions, the rate decreases as the reaction proceeds. A reaction's rate can be determined by measuring the changes in concentration over time. Chemical kinetics is the part of physical chemistry that concerns how rates of chemical reactions are measured and predicted, and how reaction-rate data can be used to deduce probable reaction mechanisms. The concepts of chemical kinetics are applied in many disciplines, such as chemical engineering, enzymology and environmental e ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dicinnamalacetone Rxn 2
Dicinnamalacetone is a conjugated organic compound. It is used as an indicator for the presence of hydrogen halides in solvents, and its preparation is used as an example of the aldol condensation in organic chemistry teaching labs. Preparation Dicinnamalacetone (specifically the all-''trans'' isomer) may be prepared by the reaction of acetone and '' trans-''cinnamaldehyde catalyzed by a strong base, such as potassium hydroxide. The reaction is typically conducted in a mixture of water and ethanol. Clock reaction The preparation of dicinnamalacetone is an example of a clock reaction. Upon the addition of acetone to a solution of ''trans''-cinnamaldehyde and potassium hydroxide in ethanol and water, a precipitate of dicinnamalacetone forms spontaneously after a delay. The formation of dicinnamalacetone constitutes two separate aldol condensations. In the first, one molar equivalent of ''trans''-cinnamaldehyde and one molar equivalent of acetone condense Condensation ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Condensation Reaction
In organic chemistry, a condensation reaction is a type of chemical reaction in which two molecules are combined to form a single molecule, usually with the loss of a small molecule such as water. If water is lost, the reaction is also known as a dehydration synthesis. However other molecules can also be lost, such as ammonia, ethanol, acetic acid and hydrogen sulfide. The addition of the two molecules typically proceeds in a step-wise fashion to the addition product, usually in equilibrium, and with loss of a water molecule (hence the name condensation). The reaction may otherwise involve the functional groups of the molecule, and is a versatile class of reactions that can occur in acidic or basic conditions or in the presence of a catalyst. This class of reactions is a vital part of life as it is essential to the formation of peptide bonds between amino acids and to the biosynthesis of fatty acids. Many variations of condensation reactions exist. Common examples include ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Aldol Condensation
An aldol condensation is a condensation reaction in organic chemistry in which two carbonyl moieties (of aldehydes or ketones) react to form a β-hydroxyaldehyde or β-hydroxyketone (an aldol reaction), and this is then followed by dehydration to give a conjugated enone. The overall reaction equation is as follows (where the Rs can be H) Aldol condensations are important in organic synthesis and biochemistry as ways to form carbon–carbon bonds. In its usual form, it involves the nucleophilic addition of a ketone enolate to an aldehyde to form a β-hydroxy ketone, or aldol (aldehyde + alcohol), a structural unit found in many naturally occurring molecules and pharmaceuticals. The term ''aldol condensation'' is also commonly used, especially in biochemistry, to refer to just the first (addition) stage of the process—the aldol reaction itself—as catalyzed by aldolases. However, the first step is formally an addition reaction rather than a condensation reaction bec ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Precipitation (chemistry)
In an aqueous solution, precipitation is the "sedimentation of a solid material (a precipitate) from a liquid solution". The solid formed is called the precipitate. In case of an inorganic chemical reaction leading to precipitation, the chemical reagent causing the solid to form is called the precipitant. The clear liquid remaining above the precipitated or the centrifuged solid phase is also called the supernate or supernatant. The notion of precipitation can also be extended to other domains of chemistry (organic chemistry and biochemistry) and even be applied to the solid phases (e.g. metallurgy and alloys) when solid impurities segregation (materials science), segregate from a solid phase. Supersaturation The precipitation of a compound may occur when its concentration exceeds its solubility. This can be due to temperature changes, solvent evaporation, or by mixing solvents. Precipitation occurs more rapidly from a strongly supersaturated solution. The formation of a pr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dicinnamalacetone Synth
Dicinnamalacetone is a conjugated organic compound. It is used as an indicator for the presence of hydrogen halides in solvents, and its preparation is used as an example of the aldol condensation in organic chemistry teaching labs. Preparation Dicinnamalacetone (specifically the all-''trans'' isomer) may be prepared by the reaction of acetone and '' trans-''cinnamaldehyde catalyzed by a strong base, such as potassium hydroxide. The reaction is typically conducted in a mixture of water and ethanol. Clock reaction The preparation of dicinnamalacetone is an example of a clock reaction. Upon the addition of acetone to a solution of ''trans''-cinnamaldehyde and potassium hydroxide in ethanol and water, a precipitate of dicinnamalacetone forms spontaneously after a delay. The formation of dicinnamalacetone constitutes two separate aldol condensations. In the first, one molar equivalent of ''trans''-cinnamaldehyde and one molar equivalent of acetone condense Condensation ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Organic Compound
Some chemical authorities define an organic compound as a chemical compound that contains a carbon–hydrogen or carbon–carbon bond; others consider an organic compound to be any chemical compound that contains carbon. For example, carbon-containing compounds such as alkanes (e.g. methane ) and its derivatives are universally considered organic, but many others are sometimes considered inorganic, such as certain compounds of carbon with nitrogen and oxygen (e.g. cyanide ion , hydrogen cyanide , chloroformic acid , carbon dioxide , and carbonate ion ). Due to carbon's ability to catenate (form chains with other carbon atoms), millions of organic compounds are known. The study of the properties, reactions, and syntheses of organic compounds comprise the discipline known as organic chemistry. For historical reasons, a few classes of carbon-containing compounds (e.g., carbonate salts and cyanide salts), along with a few other exceptions (e.g., carbon dioxide, and even ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ethanol
Ethanol (also called ethyl alcohol, grain alcohol, drinking alcohol, or simply alcohol) is an organic compound with the chemical formula . It is an Alcohol (chemistry), alcohol, with its formula also written as , or EtOH, where Et is the pseudoelement symbol for ethyl group, ethyl. Ethanol is a Volatility (chemistry), volatile, flammable, colorless liquid with a characteristic wine-like odor and pungent taste. As a psychoactive depressant, it is the active ingredient in alcoholic beverages, and the second most consumed drug globally behind caffeine. Ethanol is naturally produced by the fermentation process of sugars by yeasts or via petrochemical processes such as ethylene hydration. Historically it was used as a general anesthetic, and has modern medical applications as an antiseptic, disinfectant, solvent for some medications, and antidote for methanol poisoning and ethylene glycol poisoning. It is used as a chemical solvent and in the Chemical synthesis, synthesis of orga ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Water
Water is an inorganic compound with the chemical formula . It is a transparent, tasteless, odorless, and Color of water, nearly colorless chemical substance. It is the main constituent of Earth's hydrosphere and the fluids of all known living organisms (in which it acts as a solvent). It is vital for all known forms of life, despite not providing food energy or organic micronutrients. Its chemical formula, , indicates that each of its molecules contains one oxygen and two hydrogen atoms, connected by covalent bonds. The hydrogen atoms are attached to the oxygen atom at an angle of 104.45°. In liquid form, is also called "water" at standard temperature and pressure. Because Earth's environment is relatively close to water's triple point, water exists on Earth as a solid, a liquid, and a gas. It forms precipitation in the form of rain and aerosols in the form of fog. Clouds consist of suspended droplets of water and ice, its solid state. When finely divided, crystalline ice ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |