|
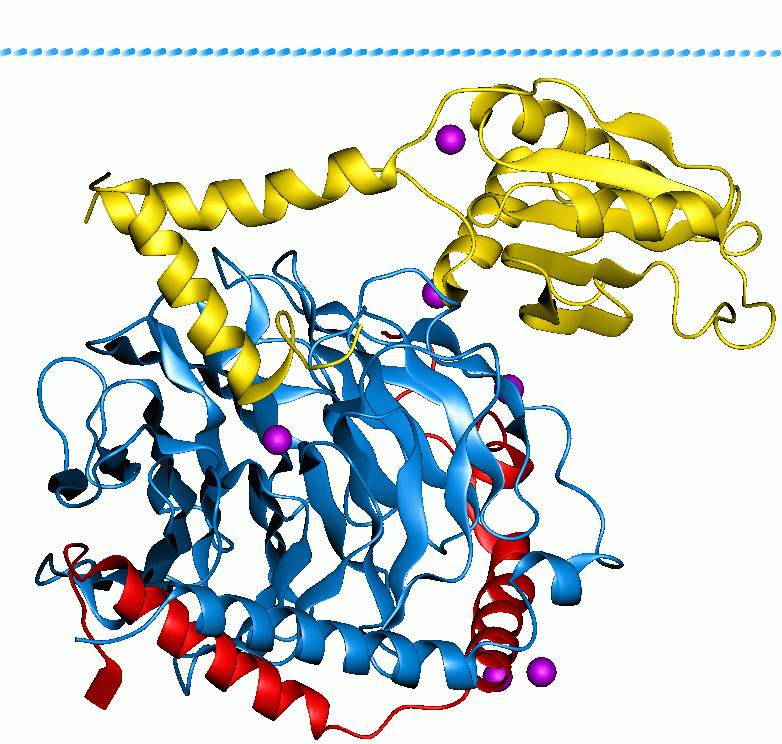
DHR2 Domain
DHR2 (DOCK homology region 2), also known as CZH2 or Docker2, is a protein domain of approximately 450-550 amino acids that is present in the DOCK family of proteins. This domain functions as a guanine nucleotide exchange factor (GEF) domain for small G proteins of the Rho family. DHR2 domains bear no significant similarity to the well described DH domain (Dbl homologous domain) present in other RhoGEFs such as Vav, P-Rex and TRIO. Indeed, the most divergent mammalian DHR2 domains share only 16-17% sequence similarity Sequence homology is the biological homology between DNA, RNA, or protein sequences, defined in terms of shared ancestry in the evolutionary history of life. Two segments of DNA can have shared ancestry because of three phenomena: either a speci .... References Further reading * * * {{refend Protein domains ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Protein Domain
In molecular biology, a protein domain is a region of a protein's Peptide, polypeptide chain that is self-stabilizing and that Protein folding, folds independently from the rest. Each domain forms a compact folded Protein tertiary structure, three-dimensional structure. Many proteins consist of several domains, and a domain may appear in a variety of different proteins. Molecular evolution uses domains as building blocks and these may be recombined in different arrangements to create proteins with different functions. In general, domains vary in length from between about 50 amino acids up to 250 amino acids in length. The shortest domains, such as zinc fingers, are stabilized by metal ions or Disulfide bond, disulfide bridges. Domains often form functional units, such as the calcium-binding EF-hand, EF hand domain of calmodulin. Because they are independently stable, domains can be "swapped" by genetic engineering between one protein and another to make chimera (protein), chimeric ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Amino Acids
Amino acids are organic compounds that contain both amino and carboxylic acid functional groups. Although over 500 amino acids exist in nature, by far the most important are the Proteinogenic amino acid, 22 α-amino acids incorporated into proteins. Only these 22 appear in the genetic code of life. Amino acids can be classified according to the locations of the core structural functional groups (Alpha and beta carbon, alpha- , beta- , gamma- (γ-) amino acids, etc.); other categories relate to Chemical polarity, polarity, ionization, and side-chain group type (aliphatic, Open-chain compound, acyclic, aromatic, Chemical polarity, polar, etc.). In the form of proteins, amino-acid ''Residue (chemistry)#Biochemistry, residues'' form the second-largest component (water being the largest) of human muscles and other tissue (biology), tissues. Beyond their role as residues in proteins, amino acids participate in a number of processes such as neurotransmitter transport and biosynthesi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
DOCK (protein)
DOCK (Dedicator of cytokinesis) is a family of related proteins involved in intracellular signalling networks. DOCK family members contain a RhoGEF domain to function as guanine nucleotide exchange factors to promote GDP release and GTP binding to specific Small GTPases of the Rho family (e.g., Rac and Cdc42), leading to their activation since Rho proteins are inactive when bound to GDP but active when bound to GTP. Subfamilies DOCK family proteins are categorised into four subfamilies based on their sequence homology Sequence homology is the homology (biology), biological homology between DNA sequence, DNA, RNA sequence, RNA, or Protein primary structure, protein sequences, defined in terms of shared ancestry in the evolutionary history of life. Two segments ...: *DOCK-A subfamily ** Dock180 (also known as Dock1) ** Dock2 ** Dock5 *DOCK-B subfamily ** Dock3 (also known as MOCA and PBP) ** Dock4 *DOCK-C subfamily (also known as Zir subfamily) ** Dock6 (also known as Zi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Guanine Nucleotide Exchange Factor
Guanine nucleotide exchange factors (GEFs) are proteins or protein domains that activate monomeric GTPases by stimulating the release of guanosine diphosphate (GDP) to allow binding of guanosine triphosphate (GTP). A variety of unrelated structural domains have been shown to exhibit guanine nucleotide exchange activity. Some GEFs can activate multiple GTPases while others are specific to a single GTPase. Function Guanine nucleotide exchange factors (GEFs) are proteins or protein domains involved in the activation of small GTPases. Small GTPases act as molecular switches in intracellular signaling pathways and have many downstream targets. The most well-known GTPases comprise the Ras superfamily and are involved in essential cell processes such as cell differentiation and proliferation, cytoskeletal organization, vesicle trafficking, and nuclear transport. GTPases are active when bound to GTP and inactive when bound to GDP, allowing their activity to be regulated by GEFs and th ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
G Protein
G proteins, also known as guanine nucleotide-binding proteins, are a Protein family, family of proteins that act as molecular switches inside cells, and are involved in transmitting signals from a variety of stimuli outside a cell (biology), cell to its interior. Their activity is regulated by factors that control their ability to bind to and hydrolyze guanosine triphosphate (GTP) to guanosine diphosphate (GDP). When they are bound to GTP, they are 'on', and, when they are bound to GDP, they are 'off'. G proteins belong to the larger group of enzymes called GTPases. There are two classes of G proteins. The first function as monomeric small GTPases (small G-proteins), while the second function as heterotrimeric G protein protein complex, complexes. The latter class of complexes is made up of ''G alpha subunit, alpha'' (Gα), ''beta'' (Gβ) and ''gamma'' (Gγ) protein subunit, subunits. In addition, the beta and gamma subunits can form a stable Protein dimer, dimeric complex re ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Rho Family
The Rho family of GTPases is a family of small (~21 kDa) signaling G proteins, and is a subfamily of the Ras superfamily. The members of the Rho GTPase family have been shown to regulate many aspects of intracellular actin dynamics, and are found in all eukaryotic kingdoms, including yeasts and some plants. Three members of the family have been studied in detail: Cdc42, Rac1, and RhoA. All G proteins are "molecular switches", and Rho proteins play a role in organelle development, cytoskeletal dynamics, cell movement, and other common cellular functions. History Identification of the Rho family of GTPases began in the mid-1980s. The first identified Rho member was RhoA, isolated serendipitously in 1985 from a low stringency cDNA screening. Rac1 and Rac2 were identified next, in 1989 followed by Cdc42 in 1990. Eight additional mammalian Rho members were identified from biological screenings until the late 1990s, a turning point in biology where availability of complete genome sequenc ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
RhoGEF Domain
RhoGEF domain describes two distinct structural domains with guanine nucleotide exchange factor (GEF) activity to regulate small GTPases in the Rho family. Rho small GTPases are inactive when bound to GDP but active when bound to GTP; RhoGEF domains in proteins are able to promote GDP release and GTP binding to activate specific Rho family members, including RhoA, Rac1 and Cdc42. The largest class of RhoGEFs is composed of proteins containing the " Dbl-homology" (DH) domain, which almost always is found together with a pleckstrin-homology (PH) domain to form a combined DH/PH domain structure. A distinct class of RhoGEFs is those proteins containing the DOCK/CZH/DHR-2 domain. This structure has no sequence similarity with DBL-homology domains. Human proteins containing DH/PH RhoGEF domain ABR; AKAP13/ARHGEF13/Lbc; ALS2; ALS2CL; ARHGEF1/p115-RhoGEF; ARHGEF10; ARHGEF10L; ARHGEF11/PDZ-RhoGEF.; ARHGEF12/LARG; ARHGEF15; ARHGEF16; ARHGEF17; ARHGEF18; ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Vav (protein)
Vav is a family of proteins involved in cell signalling. They act as guanine nucleotide exchange factors (GEFs) for small G protein G proteins, also known as guanine nucleotide-binding proteins, are a Protein family, family of proteins that act as molecular switches inside cells, and are involved in transmitting signals from a variety of stimuli outside a cell (biology), ...s of the Rho family. GEF activity is mediated via module of tandem RhoGEF domain, DH-PH domain, PH domains. Vav proteins also appear to exhibit GEF-independent functions. Although it was originally thought that Vav proteins would only be present in multicellular organisms, Vav family proteins have been observed in Choanoflagellates. Function Some functions of the Vav protein are important for the immune system. Specifically the ability of Vav to change the cytoskeletal structure of lymphocytes, which is particularly used to "aim" cytokines towards bound pathogens or cells. In humans there are three Vav ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
TRIO (gene)
Triple functional domain protein is a protein that in humans is encoded by the ''TRIO'' gene. Interactions TRIO (gene) has been shown to interact with Filamin and RHOA Transforming protein RhoA, also known as Ras homolog family member A (RhoA), is a small GTPase protein in the Rho family of GTPases that in humans is encoded by the ''RHOA'' gene. While the effects of RhoA activity are not all well known, it is .... References Further reading * * * * * * * * * * * * * * * External links * EC 2.7.11 {{gene-5-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sequence Similarity
Sequence homology is the biological homology between DNA, RNA, or protein sequences, defined in terms of shared ancestry in the evolutionary history of life. Two segments of DNA can have shared ancestry because of three phenomena: either a speciation event (orthologs), or a duplication event (paralogs), or else a horizontal (or lateral) gene transfer event (xenologs). Homology among DNA, RNA, or proteins is typically inferred from their nucleotide or amino acid sequence similarity. Significant similarity is strong evidence that two sequences are related by evolutionary changes from a common ancestral sequence. Alignments of multiple sequences are used to indicate which regions of each sequence are homologous. Identity, similarity, and conservation The term "percent homology" is often used to mean "sequence similarity”, that is the percentage of identical residues (''percent identity''), or the percentage of residues conserved with similar physicochemical properties (''p ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |