|
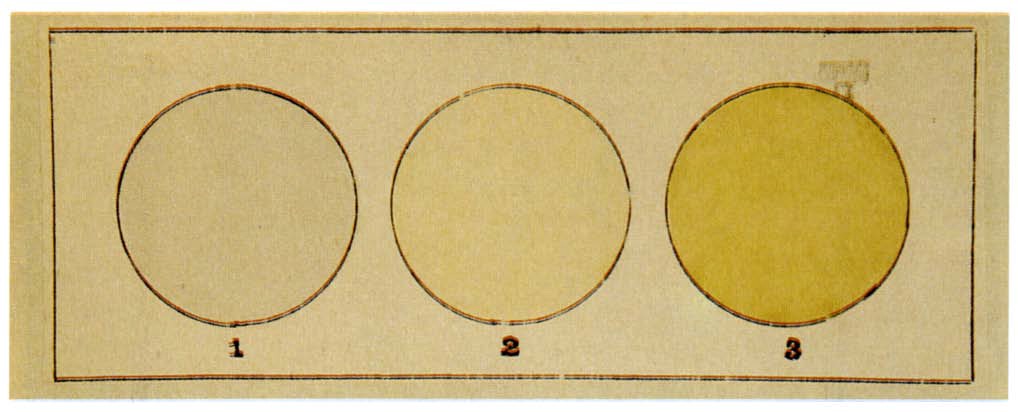
Boron Phosphide
Boron phosphide (BP) (also referred to as boron monophosphide, to distinguish it from boron subphosphide, B12P2) is a chemical compound of boron and phosphorus. It is a semiconductor. History Crystals of boron phosphide were synthesized by Henri Moissan as early as 1891. Appearance Pure BP is almost transparent, n-type crystals are orange-red whereas p-type ones are dark red. Chemical properties BP is not attacked by acids or boiling aqueous alkali water solutions. It is only attacked by molten alkalis. Physical properties BP is known to be chemically inert and exhibit very high thermal conductivity. Some properties of BP are listed below: *lattice constant 0.45383 nm *coefficient of thermal expansion 3.65 /°C (400 K) *heat capacity CP ~ 0.8 J/(g·K) (300 K) *Debye temperature = 985 K *Bulk modulus 152 GPa *relatively high microhardness of 32 GPa (100 g load). *electron and hole mobilities of a few hundred cm2/(V·s) (up to 500 for holes at 300 K) *high thermal conductivit ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Zincblende (crystal Structure)
In crystallography, the cubic (or isometric) crystal system is a crystal system where the unit cell is in the shape of a cube. This is one of the most common and simplest shapes found in crystals and minerals. There are three main varieties of these crystals: *Primitive cubic (abbreviated ''cP'' and alternatively called simple cubic) *Body-centered cubic (abbreviated ''cI'' or bcc) *Face-centered cubic (abbreviated ''cF'' or fcc, and alternatively called ''cubic close-packed'' or ccp) Each is subdivided into other variants listed below. Although the ''unit cells'' in these crystals are conventionally taken to be cubes, the primitive unit cells often are not. Bravais lattices The three Bravais lattices in the cubic crystal system are: The primitive cubic lattice (cP) consists of one lattice point on each corner of the cube; this means each simple cubic unit cell has in total one lattice point. Each atom at a lattice point is then shared equally between eight adjacent cube ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Boron
Boron is a chemical element with the symbol B and atomic number 5. In its crystalline form it is a brittle, dark, lustrous metalloid; in its amorphous form it is a brown powder. As the lightest element of the '' boron group'' it has three valence electrons for forming covalent bonds, resulting in many compounds such as boric acid, the mineral sodium borate, and the ultra-hard crystals of boron carbide and boron nitride. Boron is synthesized entirely by cosmic ray spallation and supernovae and not by stellar nucleosynthesis, so it is a low-abundance element in the Solar System and in the Earth's crust. It constitutes about 0.001 percent by weight of Earth's crust. It is concentrated on Earth by the water-solubility of its more common naturally occurring compounds, the borate minerals. These are mined industrially as evaporites, such as borax and kernite. The largest known deposits are in Turkey, the largest producer of boron minerals. Elemental boron is a meta ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phosphorus
Phosphorus is a chemical element with the symbol P and atomic number 15. Elemental phosphorus exists in two major forms, white phosphorus and red phosphorus, but because it is highly reactive, phosphorus is never found as a free element on Earth. It has a concentration in the Earth's crust of about one gram per kilogram (compare copper at about 0.06 grams). In minerals, phosphorus generally occurs as phosphate. Elemental phosphorus was first isolated as white phosphorus in 1669. White phosphorus emits a faint glow when exposed to oxygen – hence the name, taken from Greek mythology, meaning 'light-bearer' (Latin ), referring to the "Morning Star", the planet Venus. The term '' phosphorescence'', meaning glow after illumination, derives from this property of phosphorus, although the word has since been used for a different physical process that produces a glow. The glow of phosphorus is caused by oxidation of the white (but not red) phosphorus — a process now called chemi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Semiconductor
A semiconductor is a material which has an electrical conductivity value falling between that of a conductor, such as copper, and an insulator, such as glass. Its resistivity falls as its temperature rises; metals behave in the opposite way. Its conducting properties may be altered in useful ways by introducing impurities ("doping") into the crystal structure. When two differently doped regions exist in the same crystal, a semiconductor junction is created. The behavior of charge carriers, which include electrons, ions, and electron holes, at these junctions is the basis of diodes, transistors, and most modern electronics. Some examples of semiconductors are silicon, germanium, gallium arsenide, and elements near the so-called "metalloid staircase" on the periodic table. After silicon, gallium arsenide is the second-most common semiconductor and is used in laser diodes, solar cells, microwave-frequency integrated circuits, and others. Silicon is a critical element for ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Henri Moissan
Ferdinand Frédéric Henri Moissan (28 September 1852 – 20 February 1907) was a French chemist and pharmacist who won the 1906 Nobel Prize in Chemistry for his work in isolating fluorine from its compounds. Moissan was one of the original members of the International Atomic Weights Committee. Biography Early life and education Moissan was born in Paris on 28 September 1852, the son of a minor officer of the eastern railway company, Francis Ferdinand Moissan, and a seamstress, Joséphine Améraldine (née Mitel). His mother was of Jewish descent, his father was not. In 1864 they moved to Meaux, where he attended the local school. During this time, Moissan became an apprentice clockmaker. However, in 1870, Moissan and his family moved back to Paris due to war against Prussia. Moissan was unable to receive the ''grade universitaire'' necessary to attend university. After spending a year in the army, he enrolled at the Ecole Superieure de Pharmacie de Paris. Scientific career ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Bulk Modulus
The bulk modulus (K or B) of a substance is a measure of how resistant to compression the substance is. It is defined as the ratio of the infinitesimal pressure increase to the resulting ''relative'' decrease of the volume. Other moduli describe the material's response ( strain) to other kinds of stress: the shear modulus describes the response to shear stress, and Young's modulus describes the response to normal (lengthwise stretching) stress. For a fluid, only the bulk modulus is meaningful. For a complex anisotropic solid such as wood or paper, these three moduli do not contain enough information to describe its behaviour, and one must use the full generalized Hooke's law. The reciprocal of the bulk modulus at fixed temperature is called the isothermal compressibility. Definition The bulk modulus K (which is usually positive) can be formally defined by the equation :K=-V\frac , where P is pressure, V is the initial volume of the substance, and dP/dV denotes the derivative ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Boron Arsenide
Boron arsenide is a chemical compound involving boron and arsenic, usually with a chemical formula BAs. Other boron arsenide compounds are known, such as the subarsenide . Chemical synthesis of cubic BAs is very challenging and its single crystal forms usually have defects. Properties BAs is a cubic (sphalerite) semiconductor in the III-V family with a lattice constant of 0.4777 nm and an indirect band gap of 1.82 eV. Cubic BAs is reported to decompose to the subarsenide B12As2 at temperatures above 920 °C. Boron arsenide has a melting point of 2076 °C. The thermal conductivity of BAs is very high: around 1300 W/(m·K) at 300 K. The basic physical properties of cubic BAs have been experimentally measured: Band gap (1.82 eV), optical refractive index (3.29 at wavelength 657 nm), elastic modulus (326 GPa), shear modulus, Poisson's ratio, thermal expansion coefficient (3.85×10−6/K), and heat capacity. It can be alloyed with gallium arsenide to produce te ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Boron Nitride
Boron nitride is a thermally and chemically resistant refractory compound of boron and nitrogen with the chemical formula BN. It exists in various crystalline forms that are isoelectronic to a similarly structured carbon lattice. The hexagonal form corresponding to graphite is the most stable and soft among BN polymorphs, and is therefore used as a lubricant and an additive to cosmetic products. The cubic ( zincblende aka sphalerite structure) variety analogous to diamond is called c-BN; it is softer than diamond, but its thermal and chemical stability is superior. The rare wurtzite BN modification is similar to lonsdaleite but slightly softer than the cubic form. Because of excellent thermal and chemical stability, boron nitride ceramics are used in high-temperature equipment and metal casting. Boron nitride has potential use in nanotechnology. Structure Boron nitride exists in multiple forms that differ in the arrangement of the boron and nitrogen atoms, giving rise to ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Aluminium Phosphide
Aluminium phosphide is a highly toxic inorganic compound with the chemical formula AlP, used as a wide band gap semiconductor and a fumigant. This colorless solid is generally sold as a grey-green-yellow powder due to the presence of impurities arising from hydrolysis and oxidation. Properties AlP crystals are dark grey to dark yellow in color and have a zincblende crystal structure with a lattice constant of 5.4510 Å at 300 K. They are thermodynamically stable up to . Aluminium phosphide reacts with water or acids to release phosphine: :AlP + 3 H2O → Al(OH)3 + PH3 :AlP + 3 H+ → Al3+ + PH3 Preparation AlP is synthesized by combination of the elements: : 4Al + P4 → 4AlP : Caution must be taken to avoid exposing the AlP to any sources of moisture, as this generates toxic phosphine gas. Phosphine also poses fire hazards, as it is a dangerous pyrophoric compound, igniting easily in air. Uses Pesticide AlP is used as a rodenticide, insecticide, and fumi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Gallium Phosphide
Gallium phosphide (GaP), a phosphide of gallium, is a compound semiconductor material with an indirect band gap of 2.24 eV at room temperature. Impure polycrystalline material has the appearance of pale orange or grayish pieces. Undoped single crystals are orange, but strongly doped wafers appear darker due to free-carrier absorption. It is odorless and insoluble in water. GaP has a microhardness of 9450 N/mm2, a Debye temperature of , and a thermal expansion coefficient of 5.3 K−1 at room temperature. Sulfur, silicon or tellurium are used as dopants to produce n-type semiconductors. Zinc is used as a dopant for the p-type semiconductor. Gallium phosphide has applications in optical systems. Its static dielectric constant is 11.1 at room temperature. Its refractive index varies between ~3.2 and 5.0 across the visible range, which is higher than in most other semiconducting materials. In its transparent range, its index is higher than almost any other transparent material, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Boron Compounds
Boron compounds are compounds containing the element boron. In the most familiar compounds, boron has the formal oxidation state +3. These include oxides, sulfides, nitrides, and halides. Halides The trihalides adopt a planar trigonal structure. These compounds are Lewis acids in that they readily form adducts with electron-pair donors, which are called Lewis bases. For example, fluoride (F−) and boron trifluoride (BF3) combined to give the tetrafluoroborate anion, BF4−. Boron trifluoride is used in the petrochemical industry as a catalyst. The halides react with water to form boric acid. Boron is found in nature on Earth almost entirely as various oxides of B(III), often associated with other elements. More than one hundred borate minerals contain boron in oxidation state +3. These minerals resemble silicates in some respect, although boron is often found not only in a tetrahedral coordination with oxygen, but also in a trigonal planar configuration. Unlike silicates, bor ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phosphides
In chemistry, a phosphide is a compound containing the ion or its equivalent. Many different phosphides are known, with widely differing structures. Most commonly encountered on the binary phosphides, i.e. those materials consisting only of phosphorus and a less electronegative element. Numerous are polyphosphides, which are solids consisting of anionic chains or clusters of phosphorus. Phosphides are known with the majority of less electronegative elements with the exception of Hg, Pb, Sb, Bi, Te, and Po.Von Schnering, H.G. and Hönle , W. (1994) "Phosphides - Solid-state Chemistry" in ''Encyclopedia of Inorganic Chemistry''. R. Bruce King (ed.). John Wiley & Sons Finally, some phosphides are molecular. Binary phosphides Binary phosphides include phosphorus and one other element. An example of a group 1 phosphide is sodium phosphide (). Other notable examples include aluminium phosphide () and calcium phosphide (), which are used as pesticides, exploiting their tend ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |