|
Binapacryl
Binapacryl was used as a miticide and fungicide. Chemically, it is an ester derivative of dinoseb. Although binapacryl has low toxicity itself, it is readily metabolized to form dinoseb, which is highly toxic. International trade in binapacryl is regulated by the Rotterdam Convention The Rotterdam Convention (formally, the Rotterdam Convention on the Prior Informed Consent Procedure for Certain Hazardous Chemicals and Pesticides in International Trade) is a multilateral treaty to promote shared responsibilities in relatio ...; it has been withdrawn as a pesticide, since products were highly toxic to mammals, fish and aquatic invertebrates. References {{reflist Carboxylate esters Obsolete pesticides Nitrobenzene derivatives Alkene derivatives Phenol esters Sec-Butyl compounds ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Rotterdam Convention
The Rotterdam Convention (formally, the Rotterdam Convention on the Prior Informed Consent Procedure for Certain Hazardous Chemicals and Pesticides in International Trade) is a multilateral treaty to promote shared responsibilities in relation to importation of hazardous chemicals. The convention promotes open exchange of information and calls on exporters of hazardous chemicals to use proper labeling, include directions on safe handling, and inform purchasers of any known restrictions or bans. Signatory nations can decide whether to allow or ban the importation of chemicals listed in the treaty, and exporting countries are obliged to make sure that producers within their jurisdiction comply. In 2012, the Secretariats of the Basel Convention, Basel and Stockholm Convention on Persistent Organic Pollutants, Stockholm conventions, as well as the United Nations Environment Programme, UNEP-part of the Rotterdam Convention Secretariat, merged to a single Secretariat with a matrix s ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Miticide
Acaricides are pesticides that kill members of the arachnid subclass ''Acari'', which includes ticks and mites. Acaricides are used both in medicine and agriculture, although the desired selective toxicity differs between the two fields. Terminology More specific words are sometimes used, depending upon the targeted group: * "Ixodicides" are substances that kill ticks. * "Miticides" are substances that kill mites. *The term scabicide is more narrow, and refers to agents specifically targeting '' Sarcoptes''. *The term "arachnicide" is more general, and refers to agents that target arachnids. This term is used much more rarely, but occasionally appears in informal writing. As a practical matter, mites are a paraphyletic grouping, and mites and ticks are usually treated as a single group. Examples Examples include: * Permethrin can be applied as a spray. The effects are not limited to mites: lice, cockroaches, fleas, mosquitos, and other insects will be affected. * Ivermectin ca ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Fungicide
Fungicides are pesticides used to kill parasitic fungi or their spores. Fungi can cause serious damage in agriculture, resulting in losses of yield and quality. Fungicides are used both in agriculture and to fight fungal infections in animals, including humans. Fungicides are also used to control oomycetes, which are not taxonomically/genetically fungi, although sharing similar methods of infecting plants. Fungicides can either be contact, translaminar or systemic. Contact fungicides are not taken up into the plant tissue and protect only the plant where the spray is deposited. Translaminar fungicides redistribute the fungicide from the upper, sprayed leaf surface to the lower, unsprayed surface. Systemic fungicides are taken up and redistributed through the xylem vessels. Few fungicides move to all parts of a plant. Some are locally systemic, and some move upward. Most fungicides that can be bought retail are sold in liquid form, the active ingredient being present at 0.08% i ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ester
In chemistry, an ester is a compound derived from an acid (either organic or inorganic) in which the hydrogen atom (H) of at least one acidic hydroxyl group () of that acid is replaced by an organyl group (R). These compounds contain a distinctive functional group. Analogues derived from oxygen replaced by other chalcogens belong to the ester category as well. According to some authors, organyl derivatives of acidic hydrogen of other acids are esters as well (e.g. amides), but not according to the IUPAC. Glycerides are fatty acid esters of glycerol; they are important in biology, being one of the main classes of lipids and comprising the bulk of animal fats and vegetable oils. Lactones are cyclic carboxylic esters; naturally occurring lactones are mainly 5- and 6-membered ring lactones. Lactones contribute to the aroma of fruits, butter, cheese, vegetables like celery and other foods. Esters can be formed from oxoacids (e.g. esters of acetic acid, carbonic acid ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
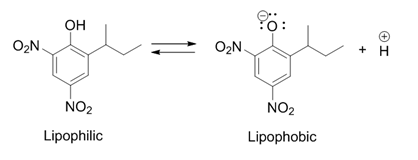
Dinoseb
Dinoseb is a common industry name for 6-sec-butyl-2,4-dinitrophenol, a herbicide in the dinitrophenol family. It is a crystalline orange solid which does not readily dissolve in water. Dinoseb is banned as an herbicide in the European Union (EU) and the United States because of its toxicity. It also finds use as a polymerisation inhibitor, where it is often referred to as DNBP. It is used to prevent the thermally induced polymerisation of styrene and other unsaturated monomers when they are being purified by distillation. History In 1892, dinitro-''ortho''-cresol (2,4-dinitro-6-methylphenol), a chemical compound closely related to dinoseb, was discovered in Germany and first used as an insecticide. It was later also used as an herbicide and also fungicide after those characteristics were discovered. In 1945 the ''ortho''-methyl group was replaced by a ''sec''-butyl group, producing dinoseb. This compound had a superior contact and stomach activity on insects and mites. Dinoseb ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Metabolism
Metabolism (, from ''metabolē'', "change") is the set of life-sustaining chemical reactions in organisms. The three main functions of metabolism are: the conversion of the energy in food to energy available to run cellular processes; the conversion of food to building blocks of proteins, lipids, nucleic acids, and some carbohydrates; and the elimination of metabolic wastes. These enzyme-catalyzed reactions allow organisms to grow and reproduce, maintain their Structures#Biological, structures, and respond to their environments. The word ''metabolism'' can also refer to the sum of all chemical reactions that occur in living organisms, including digestion and the transportation of substances into and between different cells, in which case the above described set of reactions within the cells is called intermediary (or intermediate) metabolism. Metabolic reactions may be categorized as ''catabolic''—the ''breaking down'' of compounds (for example, of glucose to pyruvate by c ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Carboxylate Esters
In organic chemistry, a carboxylate is the conjugate base of a carboxylic acid, (or ). It is an anion, an ion with negative charge. Carboxylate salts are salts that have the general formula , where M is a metal and ''n'' is 1, 2,.... Carboxylate esters have the general formula (also written as ), where R and R′ are organic groups. Synthesis Carboxylate ions can be formed by deprotonation of carboxylic acids. Such acids typically have p''K''a of less than 5, meaning that they can be deprotonated by many bases, such as sodium hydroxide or sodium bicarbonate. : Resonance stabilization of the carboxylate ion Carboxylic acids easily dissociate into a carboxylate anion and a positively charged hydrogen ion (proton), much more readily than alcohols do (into an alkoxide ion and a proton), because the carboxylate ion is stabilized by resonance. The negative charge that is left after deprotonation of the carboxyl group is delocalized between the two electronegativ ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Obsolete Pesticides
Obsolescence is the process of becoming antiquated, out of date, old-fashioned, no longer in general use, or no longer useful, or the condition of being in such a state. When used in a biological sense, it means imperfect or rudimentary when compared with the corresponding part of other organisms. The international standard IEC 62402:2019 Obsolescence Management defines obsolescence as the "transition from available to unavailable from the manufacturer in accordance with the original specification". Obsolescence frequently occurs because a replacement has become available that has, in sum, more advantages compared to the disadvantages incurred by maintaining or repairing the original. Obsolete also refers to something that is already disused or discarded, or antiquated. Typically, obsolescence is preceded by a gradual decline in popularity. Consequences Driven by rapid technological changes, new components are developed and launched on the market with increasing speed. The resul ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nitrobenzene Derivatives
Nitrobenzene is an aromatic nitro compound and the simplest of the nitrobenzenes, with the chemical formula C6H5 NO2. It is a water-insoluble pale yellow oil with an almond-like odor. It freezes to give greenish-yellow crystals. It is produced on a large scale from benzene as a precursor to aniline. In the laboratory, it is occasionally used as a solvent, especially for electrophilic reagents. As confirmed by X-ray crystallography, nitrobenzene is a planar molecule. Production Nitrobenzene is prepared by nitration of benzene with a mixture of concentrated sulfuric acid, water, and nitric acid. This mixture is sometimes called "mixed acid." The production of nitrobenzene is one of the most dangerous processes conducted in the chemical industry because of the exothermicity of the reaction (Δ''H'' = −117 kJ/mol). World capacity for nitrobenzene in 1985 was about 1,700,000 tonnes. The nitration process involves formation of the nitronium ion (NO2+), followed by an electrop ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Alkene Derivatives
In organic chemistry, an alkene, or olefin, is a hydrocarbon containing a carbon–carbon double bond. The double bond may be internal or at the terminal position. Terminal alkenes are also known as α-olefins. The International Union of Pure and Applied Chemistry (IUPAC) recommends using the name "alkene" only for acyclic hydrocarbons with just one double bond; alkadiene, alkatriene, etc., or polyene for acyclic hydrocarbons with two or more double bonds; cycloalkene, cycloalkadiene, etc. for cyclic ones; and "olefin" for the general class – cyclic or acyclic, with one or more double bonds. Acyclic alkenes, with only one double bond and no other functional groups (also known as mono-enes) form a homologous series of hydrocarbons with the general formula with ''n'' being a >1 natural number (which is two hydrogens less than the corresponding alkane). When ''n'' is four or more, isomers are possible, distinguished by the position and conformation of the double bond. A ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phenol Esters
Phenol (also known as carbolic acid, phenolic acid, or benzenol) is an aromatic organic compound with the molecular formula . It is a white crystalline solid that is volatile and can catch fire. The molecule consists of a phenyl group () bonded to a hydroxy group (). Mildly acidic, it requires careful handling because it can cause chemical burns. It is acutely toxic and is considered a health hazard. Phenol was first extracted from coal tar, but today is produced on a large scale (about 7 million tonnes a year) from petroleum-derived feedstocks. It is an important industrial commodity as a precursor to many materials and useful compounds, and is a liquid when manufactured. It is primarily used to synthesize plastics and related materials. Phenol and its chemical derivatives are essential for production of polycarbonates, epoxies, explosives such as picric acid, Bakelite, nylon, detergents, herbicides such as phenoxy herbicides, and numerous pharmaceutical drugs. Propertie ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |