|
Axatilimab
Axatilimab, sold under the brand name Niktimvo, is a monoclonal antibody used for the treatment of chronic graft-versus-host disease. It is a blocker of the colony stimulating factor-1 receptor. It is given by injection into a vein. The most common side effects include infections; increased blood level of liver enzymes; decreased blood level of phosphate; low red blood cell count (anemia); muscle, bone, or joint pain; increased blood level of pancreatic enzymes; low energy; increased blood level of calcium; increased blood level of a muscle enzyme; increased blood level of a bone enzyme; nausea; headache; diarrhea; cough; fever; shortness of breath; and infusion related reactions. Infusion-related reactions are common and can be serious. Axatilimab was approved for medical use in the United States in August 2024. The US Food and Drug Administration (FDA) considers it to be a first-in-class medication. Medical use Axatilimab is indicated for the treatment of chronic graft- ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Graft-versus-host Disease
Graft-versus-host disease (GvHD) is a syndrome, characterized by inflammation in different organs. GvHD is commonly associated with bone marrow transplants and stem cell transplants. White blood cells of the donor's immune system which remain within the donated tissue (the graft) recognize the recipient (the host) as foreign (non-self). The white blood cells present within the transplanted tissue then attack the recipient's body's cells, which leads to GvHD. This should not be confused with a transplant rejection, which occurs when the immune system of the transplant recipient rejects the transplanted tissue; GvHD occurs when the donor's immune system's white blood cells reject the recipient. The underlying principle ( alloimmunity) is the same, but the details and course may differ. GvHD can also occur after a blood transfusion, known as ''Transfusion-associated graft-versus-host disease'' or TA-GvHD if the blood products used have not been gamma irradiated or treated ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
CSF1R
Colony stimulating factor 1 receptor (CSF1R), also known as macrophage colony-stimulating factor receptor (M-CSFR), and CD115 (Cluster of Differentiation 115), is a cell-surface protein encoded by the human ''CSF1R'' gene (known also as c-FMS). CSF1R is a receptor that can be activated by two ligands: colony stimulating factor 1 (CSF-1) and interleukin-34 (IL-34). CSF1R is highly expressed in myeloid cells, and CSF1R signaling is necessary for the survival, proliferation, and differentiation of many myeloid cell types ''in vivo'' and ''in vitro.'' CSF1R signaling is involved in many diseases and is targeted in therapies for cancer, neurodegeneration, and inflammatory bone diseases. Gene In the human genome, the ''CSF1R'' gene is located on chromosome 5 (5q32), and in mice the ''Csf1r'' gene is located on chromosome 18 (18D). ''CSF1R'' is 60.002 kilobases (kbs) in length. Hematopoietic stem cells express ''CSF1R'' at low levels, but ''CSF1R'' is highly expressed in more diffe ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Intravenous
Intravenous therapy (abbreviated as IV therapy) is a medical technique that administers fluids, medications and nutrients directly into a person's vein. The intravenous route of administration is commonly used for rehydration or to provide nutrients for those who cannot, or will not—due to reduced mental states or otherwise—consume food or water per os, by mouth. It may also be used to administer pharmaceutical drug, medications or other medical therapy such as blood transfusion, blood products or electrolytes to correct electrolyte imbalances. Attempts at providing intravenous therapy have been recorded as early as the 1400s, but the practice did not become widespread until the 1900s after the development of techniques for safe, effective use. The intravenous route is the fastest way to deliver medications and fluid replacement throughout the body as they are introduced directly into the circulatory system and thus quickly distributed. For this reason, the intravenous route ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Monoclonal Antibody
A monoclonal antibody (mAb, more rarely called moAb) is an antibody produced from a cell lineage made by cloning a unique white blood cell. All subsequent antibodies derived this way trace back to a unique parent cell. Monoclonal antibodies are identical and can thus have monovalent affinity, binding only to a particular epitope (the part of an antigen that is recognized by the antibody). In contrast, polyclonal antibodies are mixtures of antibodies derived from multiple plasma cell lineages which each bind to their particular target epitope. Artificial antibodies known as bispecific monoclonal antibodies can also be engineered which include two different antigen binding sites ( FABs) on the same antibody. It is possible to produce monoclonal antibodies that specifically bind to almost any suitable substance; they can then serve to detect or purify it. This capability has become an investigative tool in biochemistry, molecular biology, and medicine. Monoclonal antibod ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Colony Stimulating Factor 1 Receptor
Colony stimulating factor 1 receptor (CSF1R), also known as macrophage colony-stimulating factor receptor (M-CSFR), and CD115 (Cluster of Differentiation 115), is a cell-surface protein encoded by the human ''CSF1R'' gene (known also as c-FMS). CSF1R is a Immune receptor, receptor that can be activated by two ligands: Macrophage colony-stimulating factor, colony stimulating factor 1 (CSF-1) and Interleukin 34, interleukin-34 (IL-34). CSF1R is highly expressed in Myeloid tissue, myeloid cells, and CSF1R signaling is necessary for the Cell survival, survival, Cell proliferation, proliferation, and Cell differentiation, differentiation of many myeloid cell types ''in vivo'' and ''in vitro.'' CSF1R signaling is involved in many diseases and is targeted in therapies for cancer, neurodegeneration, and Bone disease, inflammatory bone diseases. Gene In the human genome, the ''CSF1R'' gene is located on chromosome 5 (5q32), and in mice the ''Csf1r'' gene is located on chromosome 18 (18D). ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Food And Drug Administration
The United States Food and Drug Administration (FDA or US FDA) is a List of United States federal agencies, federal agency of the United States Department of Health and Human Services, Department of Health and Human Services. The FDA is responsible for protecting and promoting public health through the control and supervision of food safety, tobacco products, caffeine products, dietary supplements, Prescription drug, prescription and Over-the-counter drug, over-the-counter pharmaceutical drugs (medications), vaccines, biopharmaceuticals, blood transfusions, medical devices, electromagnetic radiation emitting devices (ERED), cosmetics, Animal feed, animal foods & feed and Veterinary medicine, veterinary products. The FDA's primary focus is enforcement of the Federal Food, Drug, and Cosmetic Act (FD&C). However, the agency also enforces other laws, notably Section 361 of the Public Health Service Act as well as associated regulations. Much of this regulatory-enforcement work is ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
First-in-class Medication
A first-in-class medication is a prototype drug that uses a "new and unique mechanism of action" to treat a particular medical condition. While the Food and Drug Administration's Center for Drug Evaluation and Research tracks first-in-class medications and reports on them annually, first-in-class is not considered a regulatory category. Although many first-in-class medications qualify as breakthrough therapies, Regenerative Medicine Advanced Therapies and/or orphan drugs, first-in-class status itself has no regulatory effect. Examples Controversy Safety By definition, a first-in-class drug does not have the safety evidence from analogous products that not-first-in-class drugs would have. However, a study investigating recalls and warnings in relation to first-in-class drugs approved between 1997 and 2012 by Health Canada has found that first-in-class drugs actually have a more favourable benefit-to-harm ratio. Economics First-in-class drugs are often seen as commerc ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Indicated
In medicine, an indication is a valid reason to use a certain test, medication, procedure, or surgery. There can be multiple indications to use a procedure or medication. An indication can commonly be confused with the term diagnosis. A diagnosis is the assessment that a particular medical condition is present while an indication is a reason for use. The opposite of an indication is a contraindication, a reason to withhold a certain medical treatment because the risks of treatment clearly outweigh the benefits. In the United States, indications for prescription drugs are approved by the FDA. Indications are included in the Indications and Usage section of the Prescribing Information. The primary role of this section of labeling is to enable health care practitioners to readily identify appropriate therapies for patients by clearly communicating the drug's approved indication(s). The Indications and Usage section states the disease or condition, or manifestation or symptoms thereof ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Aspartate Transaminase
Aspartate transaminase (AST) or aspartate aminotransferase, also known as AspAT/ASAT/AAT or (serum) glutamic oxaloacetic transaminase (GOT, SGOT), is a pyridoxal phosphate (PLP)-dependent transaminase enzyme () that was first described by Arthur Karmen and colleagues in 1954. AST catalyzes the reversible transfer of an α-amino group between aspartate and glutamate and, as such, is an important enzyme in amino acid metabolism. AST is found in the liver, heart, skeletal muscle, kidneys, brain, red blood cells and gall bladder. Serum AST level, serum ALT (alanine transaminase) level, and their ratio (AST/ALT ratio) are commonly measured clinically as biomarkers for liver health. The tests are part of blood test, blood panels. The biological half-life, half-life of total AST in the circulation approximates 17 hours and, on average, 87 hours for ''mitochondrial'' AST. Aminotransferase is cleared by Liver sinusoidal endothelial cell, sinusoidal cells in the liver. Function Aspartate ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
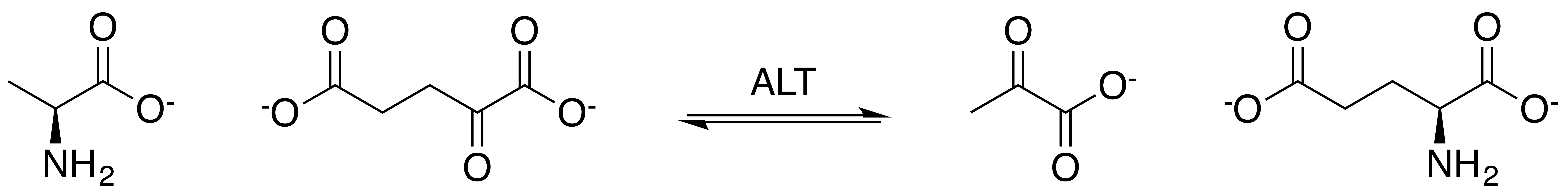
Alanine Transaminase
Alanine aminotransferase (ALT or ALAT), formerly alanine transaminase (ALT), and even earlier referred to as serum glutamate-pyruvate transaminase (GPT) or serum glutamic-pyruvic transaminase (SGPT), is a transaminase enzyme () that was first characterized in the mid-1950s by Arthur Karmen and colleagues. ALT is found in plasma and in various body tissues but is most common in the liver. It catalyzes the two parts of the alanine cycle. Serum ALT level, serum AST ( aspartate transaminase) level, and their ratio ( AST/ALT ratio) are routinely measured clinically as biomarkers for liver health. The half-life of ALT in the circulation approximates 47 hours. Aminotransferase is cleared by sinusoidal cells in the liver. Function ALT catalyzes the transfer of an amino group from L-alanine to α-ketoglutarate, the products of this reversible transamination reaction being pyruvate and L-glutamate. :L-alanine + α-ketoglutarate ⇌ pyruvate + L-glutamate ALT (and all aminotran ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Creatine Kinase
Creatine kinase (CK), also known as creatine phosphokinase (CPK) or phosphocreatine kinase, is an enzyme () expressed by various tissues and cell types. CK catalyses the conversion of creatine and uses adenosine triphosphate (ATP) to create phosphocreatine (PCr) and adenosine diphosphate (ADP). This CK enzyme reaction is reversible and thus ATP can be generated from PCr and ADP. In tissues and cells that consume ATP rapidly, especially skeletal muscle, but also brain, photoreceptor cells of the retina, hair cells of the inner ear, spermatozoa and smooth muscle, PCr serves as an energy reservoir for the rapid buffering and regeneration of ATP ''in situ'', as well as for intracellular energy transport by the PCr shuttle or circuit. Thus creatine kinase is an important enzyme in such tissues. Clinically, creatine kinase is assayed in blood tests as a marker of damage of CK-rich tissue such as in myocardial infarction (heart attack), rhabdomyolysis (severe muscle breakdown), muscu ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |