|
Amino Sugars
In organic chemistry, an amino sugar is a sugar molecule in which a hydroxyl group has been replaced with an amine group. More than 60 amino sugars are known, with one of the most abundant being ''N''-acetyl--glucosamine (a 2-amino-2-deoxysugar), which is the main component of chitin Chitin (carbon, C8hydrogen, H13oxygen, O5nitrogen, N)n ( ) is a long-chain polymer of N-Acetylglucosamine, ''N''-acetylglucosamine, an amide derivative of glucose. Chitin is the second most abundant polysaccharide in nature (behind only cell .... Derivatives of amine containing sugars, such as N-acetylglucosamine, ''N''-acetylglucosamine and sialic acid, whose Nitrogen, nitrogens are part of more complex functional groups rather than formally being amines, are also considered amino sugars. Aminoglycosides are a class of antimicrobial compounds that inhibit bacterial protein synthesis. These compounds are conjugates of amino sugars and aminocyclitols. Synthesis From glycals Glycals are cycli ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Regioselectivity
In organic chemistry, regioselectivity is the preference of chemical bonding or breaking in one direction over all other possible directions. It can often apply to which of many possible positions a reagent will affect, such as which proton a strong base will abstract from an organic molecule, or where on a substituted benzene ring a further substituent will be added. A specific example is a halohydrin formation reaction with 2-propenylbenzene: : Because of the preference for the formation of one product over another, the reaction is selective. This reaction is regioselective because it selectively generates one constitutional isomer rather than the other. Various examples of regioselectivity have been formulated as rules for certain classes of compounds under certain conditions, many of which are named. Among the first introduced to chemistry students are Markovnikov's rule for the addition of protic acids to alkenes, and the Fürst-Plattner rule for the addition of ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Galactosamine
Galactosamine is a hexosamine derived from galactose with the molecular formula C6H13NO5. This amino sugar is a constituent of some glycoprotein hormones such as follicle-stimulating hormone (FSH) and luteinizing hormone (LH). Precursors such as uridine diphosphate (UDP), UDP-''N''-acetyl-D-glucosamine, or glucosamine are used to synthesize galactosamine in the human body. A derivative of this compound is ''N''-acetyl-D-galactosamine. Galactosamine is a hepatotoxic, or liver-damaging, agent that is sometimes used in animal models of liver failure. Hepatotoxicity Galactosamine is used to induce hepatitis in rodent liver for research purposes. The result of using galactosamine to induce hepatitis is a disease model in which there is necrosis and inflammation of the liver. This type of tissue damage triggered by galactosamine resembles drug-induced liver disease in humans. Mechanism of hepatotoxicity The proposed mechanism behind galactosamine-induced hepatitis is dep ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Iminosugar
An iminosugar, also known as an iminosaccharide, is any analog of a sugar where a nitrogen atom has replaced the oxygen atom in the ring of the structure. Iminosugars are common components of plants and may be responsible for some of their medicinal properties. The first iminosugar to be isolated from a natural source, 1-deoxynojirimycin (DNJ), found in Mulberry, was reported in 1976, but few others were discovered until many years later. In terms of biochemical activity for medicinal applications, DNJ and 1,4-dideoxy-1,4-imino-D-arabinitol (DAB, another early example of this class of compounds) are alpha-glucosidase inhibitors and were shown to have anti-diabetic and anti-viral activity. DNJ was modified to produce two derivatives now used as medicines, ''N''-hydroxyethyl-DNJ ( miglitol) for diabetes and ''N''-butyl-DNJ ( miglustat) for Gaucher's disease. Anti-cancer and anti-viral activity was subsequently observed for swainsonine—a mannose analogue—and castanospermi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nucleophilic Displacement 2
In chemistry, a nucleophile is a chemical species that forms bonds by donating an electron pair. All molecules and ions with a free pair of electrons or at least one pi bond can act as nucleophiles. Because nucleophiles donate electrons, they are Lewis bases. ''Nucleophilic'' describes the affinity of a nucleophile to bond with positively charged atomic nuclei. Nucleophilicity, sometimes referred to as nucleophile strength, refers to a substance's nucleophilic character and is often used to compare the affinity of atoms. Neutral nucleophilic reactions with solvents such as alcohols and water are named solvolysis. Nucleophiles may take part in nucleophilic substitution, whereby a nucleophile becomes attracted to a full or partial positive charge, and nucleophilic addition. Nucleophilicity is closely related to basicity. The difference between the two is, that basicity is a thermodynamic property (i.e. relates to an equilibrium state), but nucleophilicity is a kinetic property, whic ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Epoxides
In organic chemistry, an epoxide is a cyclic ether, where the ether forms a three-atom Ring (chemistry), ring: two atoms of carbon and one atom of oxygen. This triangular structure has substantial ring strain, making epoxides highly Reactivity (chemistry), reactive, more so than other ethers. They are produced on a large scale for many applications. In general, low molecular weight epoxides are colourless and nonpolar, and often Volatility (chemistry), volatile. Nomenclature A compound containing the epoxide functional group can be called an epoxy, epoxide, oxirane, and ethoxyline. Simple epoxides are often referred to as oxides. Thus, the epoxide of ethylene (C2H4) is ethylene oxide (C2H4O). Many compounds have trivial names; for instance, ethylene oxide is called "oxirane". Some names emphasize the presence of the epoxide functional group, as in the compound ''1,2-epoxyheptane'', which can also be called ''1,2-heptene oxide''. A polymer formed from epoxide precursors is c ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nucleophilic Displacement 1
In chemistry, a nucleophile is a chemical species that forms bonds by donating an electron pair. All molecules and ions with a free pair of electrons or at least one pi bond can act as nucleophiles. Because nucleophiles donate electrons, they are Lewis bases. ''Nucleophilic'' describes the affinity of a nucleophile to bond with positively charged atomic nuclei. Nucleophilicity, sometimes referred to as nucleophile strength, refers to a substance's nucleophilic character and is often used to compare the affinity of atoms. Neutral nucleophilic reactions with solvents such as alcohols and water are named solvolysis. Nucleophiles may take part in nucleophilic substitution, whereby a nucleophile becomes attracted to a full or partial positive charge, and nucleophilic addition. Nucleophilicity is closely related to basicity. The difference between the two is, that basicity is a thermodynamic property (i.e. relates to an equilibrium state), but nucleophilicity is a kinetic property, whic ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Anomeric Centre
In carbohydrate chemistry, a pair of anomers () is a pair of near-identical stereoisomers or diastereomers that differ at only the anomeric carbon, the carbon atom that bears the aldehyde or ketone functional group in the sugar's open-chain form. However, in order for anomers to exist, the sugar must be in its cyclic form, since in open-chain form, the anomeric carbon atom is planar and thus achiral. More formally stated, then, an anomer is an epimer at the hemiacetal/hemiketal carbon atom in a cyclic saccharide. Anomerization is the process of conversion of one anomer to the other. As is typical for stereoisomeric compounds, different anomers have different physical properties, melting points and specific rotations. Nomenclature Every two anomers are designated alpha (α) or beta (β), according to the configurational relationship between the ''anomeric centre'' and the ''anomeric reference atom'', hence they are relative stereodescriptors. The anomeric centre in hemiacetal ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
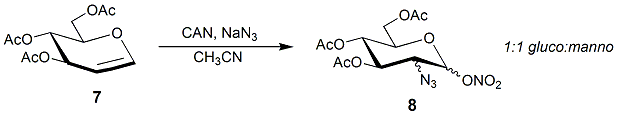
Glycal
Glycal is a name for cyclic enol ether derivatives of sugars having a double bond between carbon atoms 1 and 2 of the ring. The term "glycal" should not be used for an unsaturated sugar that has a double bond in any position other than between carbon atoms 1 and 2. History The first glycal was synthesized by Hermann Emil Fischer and Karl Zach in 1913. They synthesized this 1,2-unsaturated sugar from D-glucose and named their product D-glucal. Fischer believed he had synthesized an aldehyde, and therefore he gave the product a name that suggested this. By the time he discovered his mistake, the name "glycal" was adopted as a general name for all sugars with a double bond between carbon atoms 1 and 2. Conformation Glycals can be formed as pyranose (six-membered) or furanose (five-membered) rings, depending on the monosaccharide used as a starting material to synthesize the glycal. Glycals can also be classified as ''endo''-glycals or ''exo''-glycals. A glycal is an endo-glyc ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Michael Addition
In organic chemistry, the Michael reaction or Michael 1,4 addition is a reaction between a Michael donor (an enolate or other nucleophile) and a Michael acceptor (usually an α,β-unsaturated carbonyl) to produce a Michael adduct by creating a carbon-carbon bond at the acceptor's β-carbon. It belongs to the larger class of conjugate additions and is widely used for the mild formation of carbon–carbon bonds. The Michael addition is an important atom-economical method for diastereoselective and enantioselective C–C bond formation, and many asymmetric variants exist : In this general Michael addition scheme, either or both of R and R' on the nucleophile (the Michael donor) represent electron-withdrawing substituents such as acyl, cyano, nitro, or sulfone groups, which make the adjacent methylene hydrogen acidic enough to form a carbanion when reacted with the base, ''B:''. For the alkene (the Michael acceptor), the R" substituent is usually a carbonyl, which makes the co ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Thiophenol
Thiophenol is an organosulfur compound with the formula C6H5SH, sometimes abbreviated as PhSH. This foul-smelling colorless liquid is the simplest aromatic thiol. The chemical structures of thiophenol and its derivatives are analogous to phenols, where the oxygen atom in the hydroxyl group (−OH) bonded to the aromatic ring in phenol is replaced by a sulfur atom. The prefix thio- implies a sulfur-containing compound and when used before a root word name for a compound which would normally contain an oxygen atom, in the case of 'thiol' that the alcohol oxygen atom is replaced by a sulfur atom. Thiophenols also describes a class of compounds formally derived from thiophenol itself. All have a sulfhydryl group (-SH) covalently bonded to an aromatic ring. The organosulfur ligand in the medicine thiomersal is a thiophenol. Synthesis There are several methods of synthesis for thiophenol and related compounds, although thiophenol itself is usually purchased for laboratory oper ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |