|
Xylene
In organic chemistry, xylene or xylol (; IUPAC name: dimethylbenzene) are any of three organic compounds with the formula . They are derived from the substitution of two hydrogen atoms with methyl groups in a benzene ring; which hydrogens are substituted determines which of three structural isomers results. It is a colorless, flammable, slightly greasy liquid of great industrial value. The mixture is referred to as both xylene and, more precisely, xylenes. Mixed xylenes refers to a mixture of the xylenes plus ethylbenzene. The four compounds have identical empirical formulas . Typically the four compounds are produced together by various catalytic reforming and pyrolysis methods. Occurrence and production Xylenes are an important petrochemical produced by catalytic reforming and also by coal carbonisation in the manufacture of coke fuel. They also occur in crude oil in concentrations of about 0.5–1%, depending on the source. Small quantities occur in gasoline and aircra ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
P-Xylene
''p''-Xylene ( ''para''-xylene) is an aromatic hydrocarbon. It is one of the three isomers of dimethylbenzene known collectively as xylenes. The ''p-'' stands for ''para-'', indicating that the two methyl groups in ''p''-xylene occupy the diametrically opposite substituent positions 1 and 4. It is in the positions of the two methyl groups, their arene substitution pattern, that it differs from the other isomers, ''o''-xylene and ''m''-xylene. All have the same chemical formula C6H4(CH3)2. All xylene isomers are colorless and highly flammable. The odor threshold of ''p''-xylene is 0.62 parts per million (ppm). Production The production of ''p''-xylene is industrially significant, with annual demand estimated at 37 million tons in 2014, and still on the increase. ''p''-Xylene is produced by catalytic reforming of petroleum naphtha as part of the BTX aromatics (benzene, toluene and the xylene isomers) extracted from the catalytic reformate. The ''p''-xylene is then separated out ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
M-Xylene
''m''-Xylene ( ''meta''-xylene) is an aromatic hydrocarbon. It is one of the three isomers of dimethylbenzene known collectively as xylenes. The ''m-'' stands for ''meta-'', indicating that the two methyl groups in ''m''-xylene occupy positions 1 and 3 on a benzene ring. It is in the positions of the two methyl groups, their arene substitution pattern, that it differs from the other isomers, ''o''-xylene and ''p''-xylene. All have the same chemical formula C6H4(CH3)2. All xylene isomers are colorless and highly flammable. Production and use Petroleum contains about 1 weight percent xylenes. The ''meta'' isomer can be isolated from a mix of xylenes by the partial sulfonation (to which other isomers are less prone) followed by removal of unsulfonated oils and steam distillation of the sulfonated product. The major use of ''meta''-xylene is in the production of isophthalic acid, which is used as a copolymerizing monomer to alter the properties of polyethylene terephthalate. The ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Transalkylation
In organic chemistry, transalkylation is a chemical reaction involving the transfer of an alkyl group from one organic compound to another. The reaction is used for the transfer of methyl and ethyl groups between benzene rings. This is of particular value in the petrochemical industry to manufacture p-xylene, styrene, and other aromatic compounds. Motivation for using transalkylation reactions is based on a difference in production and demand for benzene, toluene, and xylenes. Transalkylation can convert toluene, which is overproduced, into benzene and xylene, which are under-produced. Zeolites are often used as catalysts in transalkylation reactions. Disproportionation Transalkylation, as used by the petrochemical industry, is often used to convert toluene into benzene and xylenes. This is achieved through a disproportionation reaction of toluene in which one toluene molecule transfers its methyl group to another one. The reaction is not selective, and the xylene produced ca ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
BTX (chemistry)
In the petroleum refining and petrochemical industries, the initialism BTX refers to mixtures of benzene, toluene, and the three xylene isomers, all of which are aromatic hydrocarbons. The xylene isomers are distinguished by the designations ''ortho'' – (or ''o'' –), ''meta'' – (or ''m'' –), and ''para'' – (or ''p'' –) as indicated in the adjacent diagram. If ethylbenzene is included, the mixture is sometimes referred to as BTEX. The BTX aromatics are very important petrochemical materials. Global consumption of benzene, estimated at more than 40,000,000 tons in 2010, showed an unprecedented growth of more than 3,000,000 tons from the level seen in 2009. Likewise, the para-xylene consumption showed unprecedented growth in 2010, growing by 2,800,000 tons, a full ten percent growth from 2009. [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Benzene
Benzene is an organic chemical compound with the molecular formula C6H6. The benzene molecule is composed of six carbon atoms joined in a planar ring with one hydrogen atom attached to each. Because it contains only carbon and hydrogen atoms, benzene is classed as a hydrocarbon. Benzene is a natural constituent of petroleum and is one of the elementary petrochemicals. Due to the cyclic continuous pi bonds between the carbon atoms, benzene is classed as an aromatic hydrocarbon. Benzene is a colorless and highly flammable liquid with a sweet smell, and is partially responsible for the aroma of gasoline. It is used primarily as a precursor to the manufacture of chemicals with more complex structure, such as ethylbenzene and cumene, of which billions of kilograms are produced annually. Although benzene is a major industrial chemical, it finds limited use in consumer items because of its toxicity. History Discovery The word "''benzene''" derives from "''gum benzoin''" (benz ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Petrochemical
Petrochemicals (sometimes abbreviated as petchems) are the chemical products obtained from petroleum by refining. Some chemical compounds made from petroleum are also obtained from other fossil fuels, such as coal or natural gas, or renewable sources such as maize, palm fruit or sugar cane. The two most common petrochemical classes are olefins (including ethylene and propylene) and aromatics (including benzene, toluene and xylene isomers). Oil refineries produce olefins and aromatics by fluid catalytic cracking of petroleum fractions. Chemical plants produce olefins by steam cracking of natural gas liquids like ethane and propane. Aromatics are produced by catalytic reforming of naphtha. Olefins and aromatics are the building-blocks for a wide range of materials such as solvents, detergents, and adhesives. Olefins are the basis for polymers and oligomers used in plastics, resins, fibers, elastomers, lubricants, and gels. Global ethylene production was ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
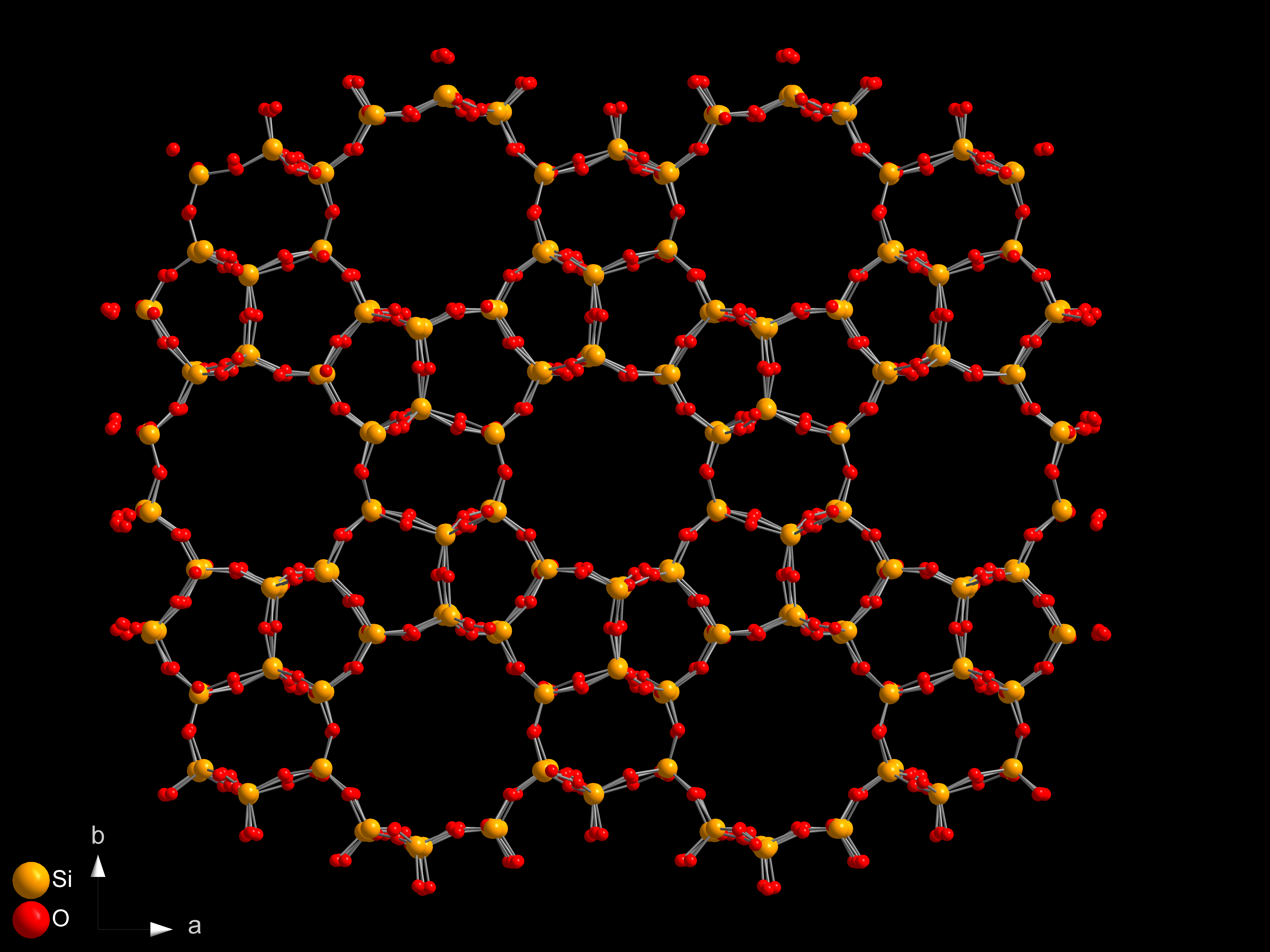
ZSM-5
ZSM-5, Zeolite Socony Mobil–5 (framework type MFI from ZSM-5 (five)), is an aluminosilicate zeolite belonging to the pentasil family of zeolites. Its chemical formula is NanAlnSi96–nO192·16H2O (0 Structure [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Toluene
Toluene (), also known as toluol (), is a substituted aromatic hydrocarbon. It is a colorless, water-insoluble liquid with the smell associated with paint thinners. It is a mono-substituted benzene derivative, consisting of a methyl group (CH3) attached to a phenyl group. As such, its systematic IUPAC name is methylbenzene. Toluene is predominantly used as an industrial feedstock and a solvent. As the solvent in some types of paint thinner, permanent markers, contact cement and certain types of glue, toluene is sometimes used as a recreational inhalant and has the potential of causing severe neurological harm. History The compound was first isolated in 1837 through a distillation of pine oil by the Polish chemist Filip Walter, who named it ''rétinnaphte''. In 1841, French chemist Henri Étienne Sainte-Claire Deville isolated a hydrocarbon from balsam of Tolu (an aromatic extract from the tropical Colombian tree ''Myroxylon balsamum''), which Deville recognized as simila ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Gasoline
Gasoline (; ) or petrol (; ) (see ) is a transparent, petroleum-derived flammable liquid that is used primarily as a fuel in most spark-ignited internal combustion engines (also known as petrol engines). It consists mostly of organic compounds obtained by the fractional distillation of petroleum, enhanced with a variety of additives. On average, U.S. refineries produce, from a barrel of crude oil, about 19 to 20 gallons of gasoline; 11 to 13 gallons of distillate fuel (most of which is sold as diesel fuel); and 3 to 4 gallons of jet fuel. The product ratio depends on the processing in an oil refinery and the crude oil assay. A barrel of oil is defined as holding 42 US gallons, which is about 159 liters or 35 imperial gallons. The characteristic of a particular gasoline blend to resist igniting too early (which causes knocking and reduces efficiency in reciprocating engines) is measured by its octane rating, which is produced in several grades. Tetraethyl lea ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Auguste André Thomas Cahours
August André Thomas Cahours (1813–1891) was a French chemist and scientist whose contribution to organic chemistry was one of the greatest in history. He discovered, among other things, the processes of synthesis of several chemical molecules, including toluene, xylene, several organo-magnesiums, and derivatives of phosphine and arsine. He was a professor at the École Polytechnique and the École Centrale des Arts et Manufactures. In 1868 he was elected as a member of the Académie des sciences (chemistry section) and in 1880 became a commander of the Légion d'honneur. Sociétés savantes de France Early life Auguste Cahours was born in on 2 October 1813. He was the first of the two children of Rose Adelaide Cartron ...[...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Jet Fuel
Jet fuel or aviation turbine fuel (ATF, also abbreviated avtur) is a type of aviation fuel designed for use in aircraft powered by gas-turbine engines. It is colorless to straw-colored in appearance. The most commonly used fuels for commercial aviation are Jet A and Jet A-1, which are produced to a standardized international specification. The only other jet fuel commonly used in civilian turbine-engine powered aviation is Jet B, which is used for its enhanced cold-weather performance. Jet fuel is a mixture of a variety of hydrocarbons. Because the exact composition of jet fuel varies widely based on petroleum source, it is impossible to define jet fuel as a ratio of specific hydrocarbons. Jet fuel is therefore defined as a performance specification rather than a chemical compound. Furthermore, the range of molecular mass between hydrocarbons (or different carbon numbers) is defined by the requirements for the product, such as the freezing point or smoke point. Kerosene-type jet ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Catalytic Reforming
Catalytic reforming is a chemical process used to convert petroleum refinery naphthas distilled from crude oil (typically having low octane ratings) into high-octane liquid products called reformates, which are premium blending stocks for high-octane gasoline. The process converts low-octane linear hydrocarbons (paraffins) into branched alkanes (isoparaffins) and cyclic naphthenes, which are then partially dehydrogenated to produce high-octane aromatic hydrocarbons. The dehydrogenation also produces significant amounts of byproduct hydrogen gas, which is fed into other refinery processes such as hydrocracking. A side reaction is hydrogenolysis, which produces light hydrocarbons of lower value, such as methane, ethane, propane and butanes. In addition to a gasoline blending stock, reformate is the main source of aromatic bulk chemicals such as benzene, toluene, xylene and ethylbenzene which have diverse uses, most importantly as raw materials for conversion into plastics. However, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |

2.png)