|
Vicarious Nucleophilic Substitution
In organic chemistry, the vicarious nucleophilic substitution is a special type of nucleophilic aromatic substitution in which a nucleophile replaces a hydrogen atom on the aromatic ring and not leaving groups such as halogen substituents which are ordinarily encountered in SNAr. This reaction type was reviewed in 1987 by Polish chemists Mieczysław Mąkosza and Jerzy Winiarski.''Synthesis of heterocyclic compounds via vicarious nucleophilic substitution of hydrogen'' Mieczysław Mąkosza Pure Appl. Chem., Vol. 69, No. 3, pp. 559-564, 1997''Article link It is typically encountered with nitroarenes and especially with nucleophiles, resulting in alkylated arenes: the new substituent can take the ''ortho'' or ''para'' positions, reversing the selectivity for the ''meta'' position that is usually observed with such compounds under electrophilic substitution. Carbon nucleophiles carry an electron-withdrawing group and a leaving group: the nucleophile attacks the aromatic ring, an ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Organic Chemistry
Organic chemistry is a subdiscipline within chemistry involving the science, scientific study of the structure, properties, and reactions of organic compounds and organic matter, organic materials, i.e., matter in its various forms that contain carbon atoms.Clayden, J.; Greeves, N. and Warren, S. (2012) ''Organic Chemistry''. Oxford University Press. pp. 1–15. . Study of structure determines their structural formula. Study of properties includes Physical property, physical and Chemical property, chemical properties, and evaluation of Reactivity (chemistry), chemical reactivity to understand their behavior. The study of organic reactions includes the organic synthesis, chemical synthesis of natural products, drugs, and polymers, and study of individual organic molecules in the laboratory and via theoretical (in silico) study. The range of chemicals studied chemistry includes hydrocarbons (compounds containing only carbon and hydrogen) as well as compounds based on carbon, but a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nitrobenzene
Nitrobenzene is an aromatic nitro compound and the simplest of the nitrobenzenes, with the chemical formula C6H5 NO2. It is a water-insoluble pale yellow oil with an almond-like odor. It freezes to give greenish-yellow crystals. It is produced on a large scale from benzene as a precursor to aniline. In the laboratory, it is occasionally used as a solvent, especially for electrophilic reagents. As confirmed by X-ray crystallography, nitrobenzene is a planar molecule. Production Nitrobenzene is prepared by nitration of benzene with a mixture of concentrated sulfuric acid, water, and nitric acid. This mixture is sometimes called "mixed acid." The production of nitrobenzene is one of the most dangerous processes conducted in the chemical industry because of the exothermicity of the reaction (Δ''H'' = −117 kJ/mol). World capacity for nitrobenzene in 1985 was about 1,700,000 tonnes. The nitration process involves formation of the nitronium ion (NO2+), followed by an e ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
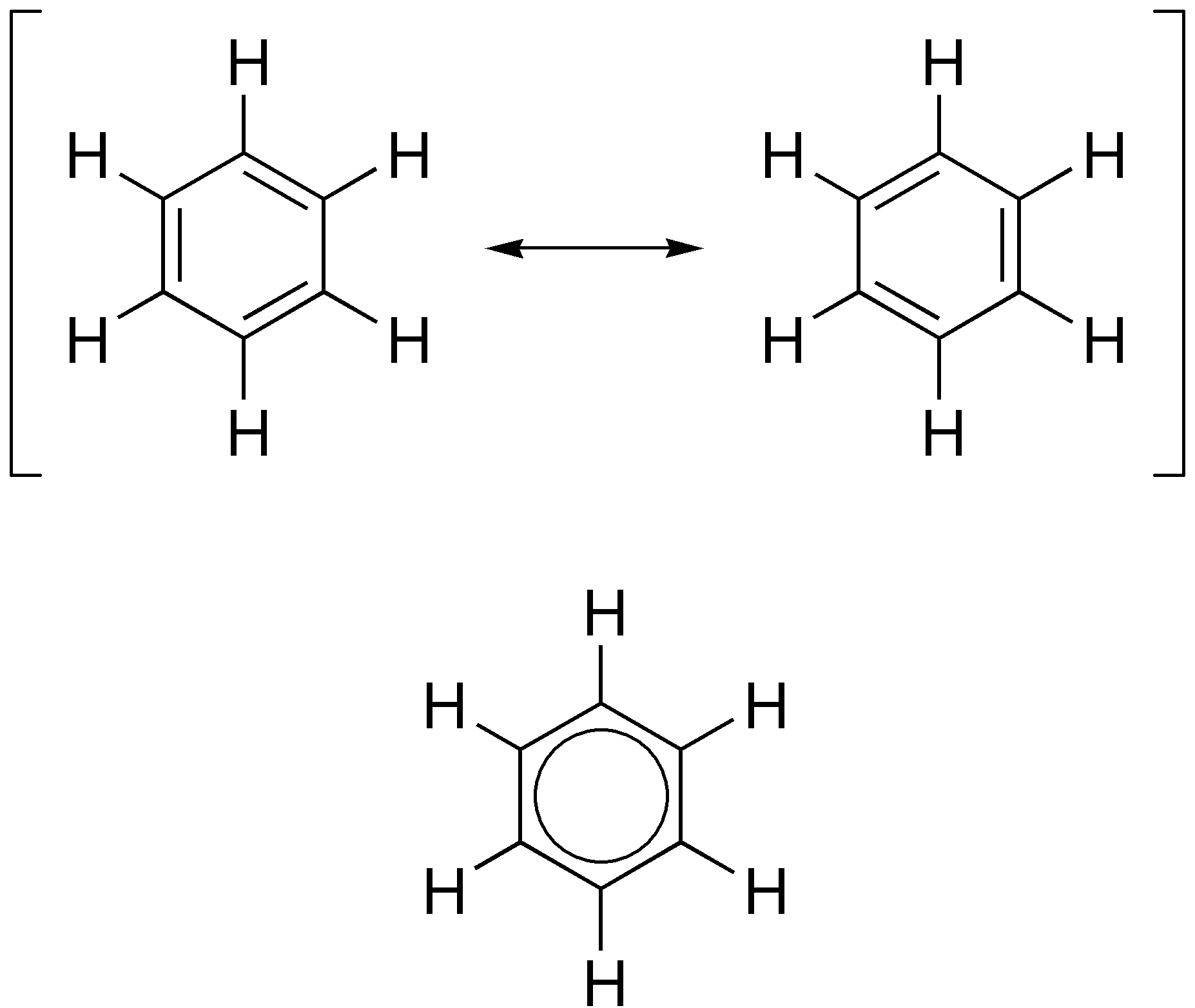
Aromaticity
In organic chemistry, aromaticity is a chemical property describing the way in which a conjugated ring of unsaturated bonds, lone pairs, or empty orbitals exhibits a stabilization stronger than would be expected from conjugation alone. The earliest use of the term was in an article by August Wilhelm Hofmann in 1855. There is no general relationship between aromaticity as a chemical property and the olfactory properties of such compounds. Aromaticity can also be considered a manifestation of cyclic delocalization and of resonance. This is usually considered to be because electrons are free to cycle around circular arrangements of atoms that are alternately single- and double- bonded to one another. This commonly seen model of aromatic rings, namely the idea that benzene was formed from a six-membered carbon ring with alternating single and double bonds (cyclohexatriene), was developed by Kekulé (see History section below). Each bond may be seen as a hybrid of a single bo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Elimination Reaction
An elimination reaction is a type of organic reaction in which two substituents are removed from a molecule in either a one- or two-step mechanism. The one-step mechanism is known as the E2 reaction, and the two-step mechanism is known as the E1 reaction. The numbers refer not to the number of steps in the mechanism, but rather to the kinetics of the reaction: E2 is bimolecular (second-order) while E1 is unimolecular (first-order). In cases where the molecule is able to stabilize an anion but possesses a poor leaving group, a third type of reaction, E1cB-elimination reaction, E1CB, exists. Finally, the pyrolysis of xanthate and acetate esters proceed through an "internal" elimination mechanism, the Ei mechanism, Ei mechanism. E2 mechanism The E2 mechanism, where E2 stands for bimolecular elimination, involves a one-step mechanism in which ''carbon-hydrogen'' and ''carbon-halogen'' bonds break to form a double bond (''C=C molecular geometry, Pi bond''). The specifics of the re ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electron-withdrawing Group
An electron-withdrawing group (EWG) is a Functional group, group or atom that has the ability to draw electron density toward itself and away from other adjacent atoms. This electron density transfer is often achieved by resonance or inductive effects. Electron-withdrawing groups have significant impacts on fundamental chemical processes such as acid-base reactions, redox potentials, and substitution reactions. Consequences of EWGs Effects on Brønsted–Lowry acidity Electron-withdrawing groups exert an "Inductive effect, inductive" or "electron-pulling" effect on covalent bonds. The strength of the electron-withdrawing group is inversely proportional to the Acid dissociation constant, pKa of the carboxylic acid. : The inductive effect is cumulative: trichloroacetic acid is 1000× stronger than chloroacetic acid. : :The impact of the EWG on pKa decreases with distances from the carboxylic group. For benzoic acids, the effect is quantified by the Hammett equation: :\log ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electrophilic Aromatic Substitution
Electrophilic aromatic substitution (SEAr) is an organic reaction in which an atom that is attached to an aromatic ring, aromatic system (usually hydrogen) is replaced by an electrophile. Some of the most important electrophilic aromatic substitutions are aromatic nitration, aromatic halogenation, aromatic sulfonation, alkylation Friedel–Crafts reaction and acylation Friedel–Crafts reaction. Illustrative reactions The most widely practised example of this reaction is the ethylation of benzene. :: Approximately 24,700,000 tons were produced in 1999. (After dehydrogenation and polymerization, the commodity plastic polystyrene is produced.) In this process, acids are used as catalyst to generate the incipient carbocation. Many other electrophilic reactions of benzene are conducted, although on a much smaller scale; they are valuable routes to key intermediates. The nitration of benzene is achieved via the action of the nitronium ion as the electrophile. The Aromatic sulfonation, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Arene Substitution Patterns
Arene substitution patterns are part of organic chemistry IUPAC nomenclature and pinpoint the position of substituents other than hydrogen in relation to each other on an aromatic hydrocarbon. ''Ortho'', ''meta'', and ''para'' substitution * In ''ortho''-substitution, two substituents occupy positions next to each other, which may be numbered 1 and 2. In the diagram, these positions are marked R and ''ortho''. * In ''meta''-substitution, the substituents occupy positions 1 and 3 (corresponding to R and ''meta'' in the diagram). * In ''para''-substitution, the substituents occupy the opposite ends (positions 1 and 4, corresponding to R and ''para'' in the diagram). The toluidines serve as an example for these three types of substitution. Synthesis Electron donating groups, for example amino, hydroxyl, alkyl, and phenyl groups tend to be ''ortho''/''para''-directors, and electron withdrawing groups such as nitro, nitrile, and ketone groups, tend to be ''meta''-directors. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pure Appl
Pure may refer to: Computing * Pure function * PureSystems, a family of computer systems introduced by IBM in 2012 * Pure Software, a company founded in 1991 by Reed Hastings to support the Purify tool * Pure-FTPd, FTP server software * Pure (programming language), functional programming language based on term rewriting * Pure Storage, a company that makes datacenter storage solutions Companies and products * Pure (app), dating app * Pure (company), a British consumer electronics company specialising in digital radios * Pure (restaurant chain), a British fast food chain * Pure Insurance, Privilege Underwriters Reciprocal Exchange * Pure Trading, a Canadian electronic communication network operated by CNQ * Pure Oil, a U.S. chain of gas stations * Propulsion Universelle et Récuperation d'Énergie (PURE), a motorsport engineering company * Pure FM (Portsmouth), a university radio station based in Portsmouth, UK * Pure (Belgian radio station), a former Belgian radio stati ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nucleophilic Aromatic Substitution
A nucleophilic aromatic substitution (SNAr) is a substitution reaction in organic chemistry in which the nucleophile displaces a good leaving group, such as a halide, on an aromatic ring. Aromatic rings are usually nucleophilic, but some aromatic compounds do undergo nucleophilic substitution. Just as normally nucleophilic alkenes can be made to undergo conjugate substitution if they carry electron-withdrawing substituents, so normally nucleophilic aromatic rings also become electrophilic if they have the right substituents.This reaction differs from a common SN2 reaction, because it happens at a trigonal carbon atom (sp2 hybridization). The mechanism of SN2 reaction does not occur due to steric hindrance of the benzene ring. In order to attack the C atom, the nucleophile must approach in line with the C-LG (leaving group) bond from the back, where the benzene ring lies. It follows the general rule for which SN2 reactions occur only at a tetrahedral carbon atom. The SN1 mechani ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Jerzy Winiarski
Jerzy is the Polish version of the masculine given name George. The most common nickname for Jerzy is Jurek (), which may also be used as an official first name. Occasionally the nickname Jerzyk may be used, which means "swift" in Polish. People *Jerzy, ''nom de guerre'' of Ryszard Białous, Polish World War II resistance fighter * Jerzy Andrzejewski, Polish writer * Jerzy Bartmiński, Polish linguist and ethnologist * Jerzy Braun (other), several people * Jerzy Brzęczek, Polish footballer and manager * Jerzy Buzek, Polish politician and former Prime Minister and former President of the European Parliament * Jerzy Dudek, Polish footballer * Jerzy Fedorowicz, Polish actor and theatre director * Jerzy Ficowski, Polish poet and translator * Jerzy Grotowski, Polish theatre director and theorist * Jerzy Hoffman, Polish film director, screenwriter, and producer * Jerzy Jarniewicz, Polish poet, literary critic, translator and essayist * Jerzy Janiszewski, Polish artis ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Mieczysław Mąkosza
Mieczysław Józef Mąkosza (born 16 November 1934) is a Polish chemist specializing in organic synthesis and investigation of organic mechanisms. Along with Jerzy Winiarski he is credited for the discovery of the aromatic vicarious nucleophilic substitution, VNS. He also contributed to the discovery of phase transfer catalysis reactions. From 1979 to 2005 he was director of the Institute of Organic Chemistry of the Polish Academy of Sciences The Polish Academy of Sciences (, PAN) is a Polish state-sponsored institution of higher learning. Headquartered in Warsaw, it is responsible for spearheading the development of science across the country by a society of distinguished scholars a .... References 1934 births 20th-century Polish chemists Living people Recipients of the State Award Badge (Poland) 21st-century Polish chemists {{Poland-scientist-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |