|
Triethylamine
Triethylamine is the chemical compound with the formula N(CH2CH3)3, commonly abbreviated Et3N. Like triethanolamine and the tetraethylammonium ion, it is often abbreviated TEA. It is a colourless volatile liquid with a strong fishy odor reminiscent of ammonia. Like diisopropylethylamine (Hünig's base), triethylamine is commonly employed in organic synthesis, usually as a base. Synthesis and properties Triethylamine is prepared by the alkylation of ammonia with ethanol: :NH3 + 3 C2H5OH → N(C2H5)3 + 3 H2O The pKa of protonated triethylamine is 10.75,David Evans Research Group and it can be used to prepare buffer solutions at that pH. The hydrochloride |
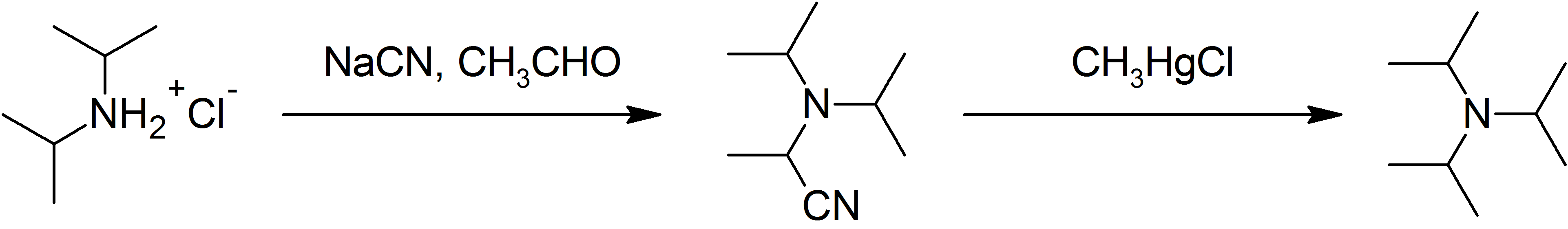
N,N-Diisopropylethylamine
''N'',''N''-Diisopropylethylamine, or Hünig's base, is an organic compound that is a tertiary amine. It is named after the German chemist . It is used in organic chemistry as a non-nucleophilic base. It is commonly abbreviated as DIPEA, DIEA, or ''i''-Pr2NEt. Structure DIPEA consists of a central nitrogen atom that is bonded to an ethyl group and two isopropyl groups. A lone pair of electrons resides on the nitrogen atom, which can react with electrophiles. However, the three alkyl groups on the nitrogen atom create steric hindrance, so only small electrophiles such as Hydrogen ion, protons can react with the nitrogen lone pair. Occurrence and preparation DIPEA is commercially available. It is traditionally prepared by the alkylation of diisopropylamine with diethyl sulfate. Pure DIPEA exists as a colorless liquid, although commercial samples can be slightly yellow. If necessary, the compound can be purified by distillation from potassium hydroxide or calcium hydride. Use ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Diisopropylethylamine
''N'',''N''-Diisopropylethylamine, or Hünig's base, is an organic compound that is a tertiary amine. It is named after the German chemist . It is used in organic chemistry as a non-nucleophilic base. It is commonly abbreviated as DIPEA, DIEA, or ''i''-Pr2NEt. Structure DIPEA consists of a central nitrogen atom that is bonded to an ethyl group and two isopropyl groups. A lone pair of electrons resides on the nitrogen atom, which can react with electrophiles. However, the three alkyl groups on the nitrogen atom create steric hindrance, so only small electrophiles such as protons can react with the nitrogen lone pair. Occurrence and preparation DIPEA is commercially available. It is traditionally prepared by the alkylation of diisopropylamine with diethyl sulfate. Pure DIPEA exists as a colorless liquid, although commercial samples can be slightly yellow. If necessary, the compound can be purified by distillation from potassium hydroxide or calcium hydride. Uses and react ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tris(2-aminoethyl)amine
Tris(2-aminoethyl)amine is the organic compound with the chemical formula, formula N(CH2CH2NH2)3. This colourless liquid is soluble in water and is highly basic, consisting of a tertiary amine center and three pendant primary amine groups. Tris(2-aminoethyl)amine is commonly abbreviated as tren or TREN. It is used a crosslinking agent in the synthesis of polyimine networks and a tripodal ligand in coordination chemistry. Supramolecular and polymer derivatives Tris(2-aminoethyl)amine has been used to prepare molecular capsules and related supramolecular structures. Metal complexes Tren is a C3-symmetric, tetradentate chelating ligand that forms stable complexes with transition metals, especially those in the 2+ and 3+ oxidation states. Tren complexes exist with relatively few isomers, reflecting the constrained connectivity of this tetramine. Thus, only a single achiral stereoisomer exists for [Co(tren)X2]+, where X is halide or pseudohalide. In contrast, for [Co(trien)X2]+ five di ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Triisopropylamine
Triisopropylamine is an organic chemical compound consisting of three isopropyl groups bound to a central nitrogen atom. As a hindered tertiary amine, it can be used as a non-nucleophilic base and as a stabilizer for polymers; however, its applications are limited by its relatively high cost and difficult synthesis. Structure In the early 1990s, theoretical studies and electron diffraction analysis of the 3D structure of the molecule, in the gas phase or in non-polar solvents, indicated that the bonds between the nitrogen atom and the three carbon atoms were nearly coplanar in the ground state, instead of forming a trigonal pyramid as in simpler amines. The average C-N-C angle was claimed to be 119.2°, much closer to the 120° of the flat configuration than to the 111.8° of trimethylamine. This peculiarity was attributed to steric hindrance by the bulky isopropyl radicals. However, in 1998 X-ray diffraction analysis of the crystallized solid showed that the C3N core is ac ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Triethanolamine
Triethanolamine, or TEOA, is an organic compound with the chemical formula . It is a colourless, viscous liquid. It is both a tertiary amine and a triol. A triol is a molecule with three alcohol groups. Approximately 150,000 tonnes were produced in 1999. It is a colourless compound although samples may appear yellow because of impurities. Production Triethanolamine is produced from the reaction of ethylene oxide with aqueous ammonia, also produced are ethanolamine and diethanolamine. The ratio of the products can be controlled by changing the stoichiometry of the reactants. : Applications Triethanolamine is used primarily in making surfactants, such as for emulsifier. It is a common ingredient in formulations used for both industrial and consumer products. The triethanolamine neutralizes fatty acids, adjusts and buffers the pH, and solubilizes oils and other ingredients that are not completely soluble in water. Triethanolammonium salts in some cases are more soluble ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tetraethylammonium
Tetraethylammonium (TEA) is a quaternary ammonium cation with the chemical formula , consisting of four ethyl groups (, denoted Et) attached to a central nitrogen atom. It is a counterion used in the research laboratory to prepare lipophilic salts of inorganic anions. It is used similarly to tetrabutylammonium, the difference being that its salts are less lipophilic, more easily crystallized and more Toxicity, toxic. Preparation The halide salt is prepared by the reaction of triethylamine and an ethyl halide: : This method works well for the preparation of tetraethylammonium iodide (where X = I). Most tetraethylammonium salts are prepared by salt metathesis reactions. For example, the synthesis of tetraethylammonium perchlorate, a salt that has been useful as a supporting electrolyte for polarography, polarographic studies in non-aqueous solvents, is carried out by mixing the water-soluble salts tetraethylammonium bromide and sodium perchlorate in water, from which the water-in ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Diethylamine
Diethylamine is an organic compound with the formula . It is classified as a secondary amine. It is a flammable, volatile weakly alkaline liquid that is miscible with most solvents. It is a colorless liquid, but commercial samples often appear brown due to impurities. It has a strong ammonia-like odor. Production and uses The alumina-catalyzed reaction makes diethylamine from ethanol and ammonia. Diethylamine is obtained together with ethylamine and triethylamine. Annual production of the three ethylamines was estimated in 2000 to be 80,000,000 kg. Diethylamine is used in the production of corrosion inhibitor ''N'',''N''- diethylaminoethanol, by reaction with ethylene oxide. It is also a precursor to a wide variety of other commercial products. It is also sometimes used in the illicit production of LSD. Organic chemistry As the most abundantly available secondary amine that is liquid at room temperature, diethylamine has been extensively deployed in chemical synthesis. It ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
HN3 (nitrogen Mustard)
2-Chloro-''N'',''N''-bis(2-chloroethyl)ethanamine, also known as trichlormethine, tris(2-chloroethyl)amine is the organic compound with the formula N(CH2CH2Cl)3. Often abbreviated HN3 or HN-3, it is a powerful blister agent and a nitrogen mustard used for chemical warfare. HN3 was the last of the nitrogen mustard agents developed. It was designed as a military agent and is the only one of the nitrogen mustards that is still used for military purposes. It is the principal representative of the nitrogen mustards because its vesicant properties are almost equal to those of HD and thus the analogy between the two types of mustard is the strongest.NITROGEN MUSTARD HN-3 Emergency Response Safety and Health Database. |
Volatility (chemistry)
In chemistry, volatility is a material quality which describes how readily a substance vaporizes. At a given temperature and pressure, a substance with high volatility is more likely to exist as a vapour, while a substance with low volatility is more likely to be a liquid or solid. Volatility can also describe the tendency of a vapor to condense into a liquid or solid; less volatile substances will more readily condense from a vapor than highly volatile ones. Differences in volatility can be observed by comparing how fast substances within a group evaporate (or sublimate in the case of solids) when exposed to the atmosphere. A highly volatile substance such as rubbing alcohol (isopropyl alcohol) will quickly evaporate, while a substance with low volatility such as vegetable oil will remain condensed. In general, solids are much less volatile than liquids, but there are some exceptions. Solids that sublimate (change directly from solid to vapor) such as dry ice (solid carbon di ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ammonia
Ammonia is an inorganic chemical compound of nitrogen and hydrogen with the chemical formula, formula . A Binary compounds of hydrogen, stable binary hydride and the simplest pnictogen hydride, ammonia is a colourless gas with a distinctive pungent smell. It is widely used in fertilizers, refrigerants, explosives, cleaning agents, and is a precursor for numeous chemicals. Biologically, it is a common nitrogenous waste, and it contributes significantly to the nutritional needs of terrestrial organisms by serving as a precursor to fertilisers. Around 70% of ammonia produced industrially is used to make fertilisers in various forms and composition, such as urea and diammonium phosphate. Ammonia in pure form is also applied directly into the soil. Ammonia, either directly or indirectly, is also a building block for the synthesis of many chemicals. In many countries, it is classified as an List of extremely hazardous substances, extremely hazardous substance. Ammonia is toxic, cau ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Royal Society Of Chemistry
The Royal Society of Chemistry (RSC) is a learned society and professional association in the United Kingdom with the goal of "advancing the chemistry, chemical sciences". It was formed in 1980 from the amalgamation of the Chemical Society, the Royal Institute of Chemistry, the Faraday Society, and the Society for Analytical Chemistry with a new Royal Charter and the dual role of learned society and professional body. At its inception, the Society had a combined membership of 49,000 in the world. The headquarters of the Society are at Burlington House, Piccadilly, London. It also has offices in Thomas Graham House in Cambridge (named after Thomas Graham (chemist), Thomas Graham, the first president of the Chemical Society) where ''RSC Publishing'' is based. The Society has offices in the United States, on the campuses of The University of Pennsylvania and Drexel University, at the University City Science Center in Philadelphia, Pennsylvania, in both Beijing and Shanghai, People' ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chemical Formula
A chemical formula is a way of presenting information about the chemical proportions of atoms that constitute a particular chemical compound or molecule, using chemical element symbols, numbers, and sometimes also other symbols, such as parentheses, dashes, brackets, commas and ''plus'' (+) and ''minus'' (−) signs. These are limited to a single typographic line of symbols, which may include subscripts and superscripts. A chemical formula is not a chemical name since it does not contain any words. Although a chemical formula may imply certain simple chemical structures, it is not the same as a full chemical structural formula. Chemical formulae can fully specify the structure of only the simplest of molecules and chemical substances, and are generally more limited in power than chemical names and structural formulae. The simplest types of chemical formulae are called '' empirical formulae'', which use letters and numbers indicating the numerical ''proportions'' of atoms ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |