|
Peptide Synthesis
In organic chemistry, peptide synthesis is the production of peptides, compounds where multiple amino acids are linked via amide bonds, also known as peptide bonds. Peptides are chemically synthesized by the condensation reaction of the carboxyl group of one amino acid to the amino group of another. Protecting group strategies are usually necessary to prevent undesirable side reactions with the various amino acid side chains. Chemical peptide synthesis most commonly starts at the carboxyl end of the peptide (C-terminus), and proceeds toward the amino-terminus (N-terminus). Protein biosynthesis (long peptides) in living organisms occurs in the opposite direction. The chemical synthesis of peptides can be carried out using classical solution-phase techniques, although these have been replaced in most research and development settings by solid-phase methods (see below). Solution-phase synthesis retains its usefulness in large-scale production of peptides for industrial purposes howe ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Peptide Coupling
In organic chemistry, peptide synthesis is the production of peptides, compounds where multiple amino acids are linked via amide bonds, also known as peptide bonds. Peptides are chemically synthesized by the condensation reaction of the carboxyl group of one amino acid to the amino group of another. Protecting group strategies are usually necessary to prevent undesirable side reactions with the various amino acid side chains. Chemical peptide synthesis most commonly starts at the carboxyl end of the peptide (C-terminus), and proceeds toward the amino-terminus (N-terminus). Protein biosynthesis (long peptides) in living organisms occurs in the opposite direction. The chemical synthesis of peptides can be carried out using classical solution-phase techniques, although these have been replaced in most research and development settings by solid-phase methods (see below). Solution-phase synthesis retains its usefulness in large-scale production of peptides for industrial purposes howe ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Multicolumn Countercurrent Solvent Gradient Purification
Multicolumn Countercurrent Solvent Gradient Purification (MCSGP) is a form of chromatography that is used to separate or purify biomolecules from complex mixtures. It was developed at the Swiss Federal Institute of Technology Zürich by Aumann and Morbidelli. The process consists of two to six chromatographic columns which are connected to one another in such a way that as the mixture moves through the columns the compound is purified into several fractions. Overview The MCSGP process consists of several, at least two, chromatographic columns which are switched in position opposite to the flow direction (seprocess animation. Most of the columns are equipped with a gradient pump to adjust the modifier concentration at the column inlet. Some columns are connected directly, so that non pure product streams are internally recycled. Other columns are short circuited, so that they operate in pure batch mode. The system is split into several sections, from which every section performs a ta ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
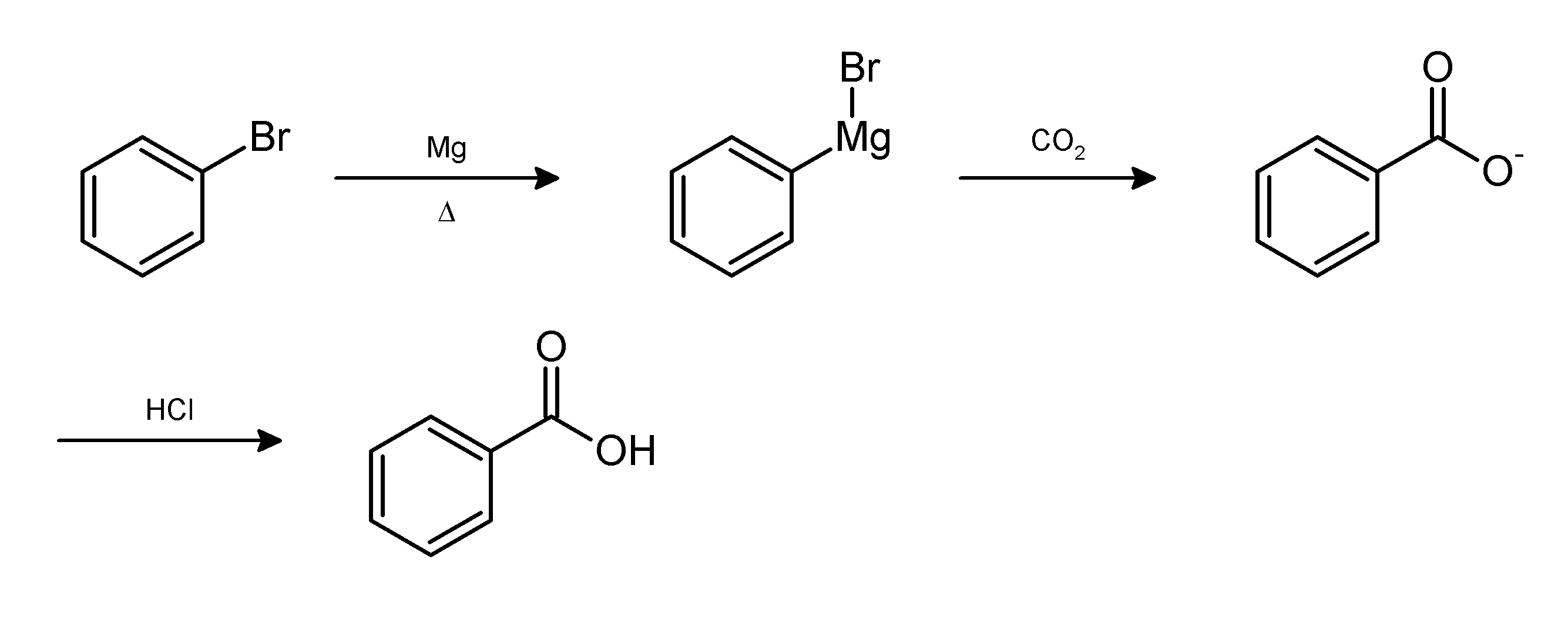
Work-up (chemistry)
In chemistry, work-up refers to the series of manipulations required to isolate and purify the product(s) of a chemical reaction. Typically, these manipulations may include: * quenching a reaction to deactivate any unreacted reagents. * cooling the reaction mixture or adding an '' antisolvent'' to induce precipitation, and collecting or removing the solids by filtration, decantation, or centrifugation * removal of solvents by evaporation * separating the reaction mixture into organic and aqueous layers by liquid-liquid extraction * purification by chromatography, distillation or recrystallization For example, the Grignard reaction between phenylmagnesium bromide and carbon dioxide in the form of dry ice gives the conjugate base of benzoic acid. The desired product, benzoic acid, is obtained by the following work-up:{{cite book , title = Introduction to Organic Laboratory Techniques: A Small Scale Approach , author = Donald L. Pavia , year = 2004 , publisher = Thomson Broo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
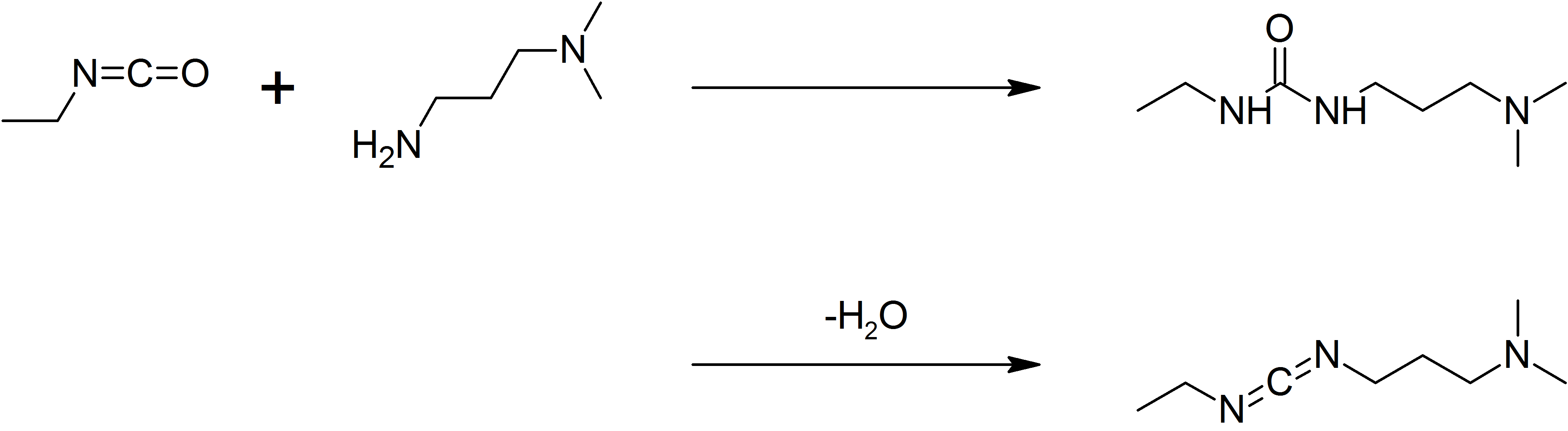
1-Ethyl-3-(3-dimethylaminopropyl)carbodiimide
1-Ethyl-3-(3-dimethylaminopropyl)carbodiimide (EDC, EDAC or EDCI) is a water-soluble carbodiimide usually handled as the hydrochloride. It is typically employed in the 4.0-6.0 pH range. It is generally used as a carboxyl activating agent for the coupling of primary amines to yield amide bonds. While other carbodiimides like dicyclohexylcarbodiimide (DCC) or diisopropylcarbodiimide (DIC) are also employed for this purpose, EDC has the advantage that the urea byproduct formed (often challenging to remove in the case of DCC or DIC) can be washed away from the amide product using dilute acid. Additionally, EDC can also be used to activate phosphate groups in order to form phosphomonoesters and phosphodiesters. Common uses for this carbodiimide include peptide synthesis, protein crosslinking to nucleic acids, but also in the preparation of immunoconjugates. EDC is often used in combination with ''N''-hydroxysuccinimide (NHS) for the immobilisation of large biomolecules. Recent work ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Urea
Urea, also known as carbamide, is an organic compound with chemical formula . This amide has two amino groups (–) joined by a carbonyl functional group (–C(=O)–). It is thus the simplest amide of carbamic acid. Urea serves an important role in the metabolism of nitrogen-containing compounds by animals and is the main nitrogen-containing substance in the urine of mammals. It is a colorless, odorless solid, highly soluble in water, and practically non-toxic ( is 15 g/kg for rats). Dissolved in water, it is neither acidic nor alkaline. The body uses it in many processes, most notably nitrogen excretion. The liver forms it by combining two ammonia molecules () with a carbon dioxide () molecule in the urea cycle. Urea is widely used in fertilizers as a source of nitrogen (N) and is an important raw material for the chemical industry. In 1828 Friedrich Wöhler discovered that urea can be produced from inorganic starting materials, which was an important conceptual miles ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Diisopropylcarbodiimide
''N'',''N′''-Diisopropylcarbodiimide is a carbodiimide used in peptide synthesis. As a liquid, it is easier to handle than the commonly used ''N'',''N′''-dicyclohexylcarbodiimide, a waxy solid. In addition, ''N'',''N′''-diisopropylurea, its byproduct in many chemical reactions, is soluble in most organic solvents, a property that facilitates work-up In chemistry, work-up refers to the series of manipulations required to isolate and purify the product(s) of a chemical reaction. Typically, these manipulations may include: * quenching a reaction to deactivate any unreacted reagents. * coolin .... Further reading * * {{organic-compound-stub Peptide coupling reagents Carbodiimides Reagents for biochemistry Biochemistry Biochemistry methods Isopropyl compounds ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dicyclohexylcarbodiimide
''N'',''N''′-Dicyclohexylcarbodiimide (DCC or DCCD) is an organic compound with the chemical formula (C6H11N)2C. It is a waxy white solid with a sweet odor. Its primary use is to couple amino acids during artificial peptide synthesis. The low melting point of this material allows it to be melted for easy handling. It is highly soluble in dichloromethane, tetrahydrofuran, acetonitrile and dimethylformamide, but insoluble in water. Structure and spectroscopy The C−N=C=N−C core of carbodiimides (N=C=N) is linear, being related to the structure of allene. The molecule has idealized C2 symmetry. The N=C=N moiety gives characteristic IR spectroscopic signature at 2117 cm−1. The 15N NMR spectrum shows a characteristic shift of 275 ppm upfield of nitric acid and the 13C NMR spectrum features a peak at about 139 ppm downfield from TMS. Preparation DCC is produced by the decarboxylation of cyclohexylisocyanate using phosphine oxides as a catalyst: :2 C6H11NCO ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Carbodiimide
In organic chemistry, a carbodiimide (systematic IUPAC name: methanediimine) is a functional group with the formula RN=C=NR. They are exclusively synthetic. A well known carbodiimide is dicyclohexylcarbodiimide, which is used in peptide synthesis. Dialkylcarbodiimides are stable. Some diaryl derivatives tend to convert to dimers and polymers upon standing at room temperature, though this mostly occurs with low melting point carbodiimides that are liquids at room temperature. Solid diaryl carbodiimides are more stable, but can slowly undergo hydrolysis in the presence of water over time. Structure and bonding From the perspective of bonding, carbodiimides are isoelectronic with carbon dioxide. Three principal resonance structures describe carbodiimides: :RN=C=NR ↔ RN+≡C-N−R ↔ RN−-C≡N+R The N=C=N core is relatively linear and the C-N=C angles approach 120°. In the case of C(NCHPh2)2, the central N=C=N angle is 170° and the C-N=C angles are within 1° of 126°. Th ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Epimer
In stereochemistry, an epimer is one of a pair of diastereomers. The two epimers have opposite configuration at only one stereogenic center out of at least two. All other stereogenic centers in the molecules are the same in each. Epimerization is the interconversion of one epimer to the other epimer. Doxorubicin and epirubicin are two epimers that are used as drugs. Examples The stereoisomers β-D-glucopyranose and β-D- mannopyranose are epimers because they differ only in the stereochemistry at the C-2 position. The hydroxy group in β-D-glucopyranose is equatorial (in the "plane" of the ring), while in β-D-mannopyranose the C-2 hydroxy group is axial (up from the "plane" of the ring). These two molecules are epimers but, because they are not mirror images of each other, are not enantiomers. (Enantiomers have the same name, but differ in D and L classification.) They are also not sugar anomers, since it is not the anomeric carbon involved in the stereochemistry. Similarl ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Racemization
In chemistry, racemization is a conversion, by heat or by chemical reaction, of an optically active compound into a racemic (optically inactive) form. This creates a 1:1 molar ratio of enantiomers and is referred too as a racemic mixture (i.e. contain equal amount of (+) and (−) forms). Plus and minus forms are called Dextrorotation and levorotation. The D and L enantiomers are present in equal quantities, the resulting sample is described as a racemic mixture or a racemate. Racemization can proceed through a number of different mechanisms, and it has particular significance in pharmacology as different enantiomers may have different pharmaceutical effects. Stereochemistry Chiral molecules have two forms (at each point of asymmetry), which differ in their optical characteristics: The ''levorotatory form'' (the ''(−)-form'') will rotate counter-clockwise on the plane of polarization of a beam of light, whereas the ''dextrorotatory'' form (the ''(+)-form'') will rotate clockwi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Amide
In organic chemistry, an amide, also known as an organic amide or a carboxamide, is a compound with the general formula , where R, R', and R″ represent organic groups or hydrogen atoms. The amide group is called a peptide bond when it is part of the main chain of a protein, and an isopeptide bond when it occurs in a side chain, such as in the amino acids asparagine and glutamine. It can be viewed as a derivative of a carboxylic acid () with the hydroxyl group () replaced by an amine group (); or, equivalently, an acyl (alkanoyl) group () joined to an amine group. Common examples of amides are acetamide (), benzamide (), and dimethylformamide (). Amides are qualified as primary, secondary, and tertiary according to whether the amine subgroup has the form , , or , where R and R' are groups other than hydrogen. The core of amides is called the amide group (specifically, carboxamide group). Amides are pervasive in nature and technology. Proteins and importa ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |