|
Interatomic Potential
Interatomic potentials are mathematical functions to calculate the potential energy of a system of atoms with given positions in space.M. P. Allen and D. J. Tildesley. Computer Simulation of Liquids. Oxford University Press, Oxford, England, 1989.R. Lesar. Introduction to Computational Materials Science. Cambridge University Press, 2013. Interatomic potentials are widely used as the physical basis of molecular mechanics and molecular dynamics simulations in computational chemistry, computational physics and computational materials science to explain and predict materials properties. Examples of quantitative properties and qualitative phenomena that are explored with interatomic potentials include lattice parameters, surface energies, interfacial energies, adsorption, cohesion, thermal expansion, and elastic and plastic material behavior, as well as chemical reactions.N. W. Ashcroft and N. D. Mermin. Solid State Physics.Saunders College, Philadelphia, 1976.Charles Kittel. Introduct ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ewald Summation
Ewald summation, named after Paul Peter Ewald, is a method for computing long-range interactions (e.g. electrostatic interactions) in periodic systems. It was first developed as the method for calculating electrostatic energies of ionic crystals, and is now commonly used for calculating long-range interactions in computational chemistry. Ewald summation is a special case of the Poisson summation formula, replacing the summation of interaction energies in real space with an equivalent summation in Fourier space. In this method, the long-range interaction is divided into two parts: a short-range contribution, and a long-range contribution which does not have a singularity. The short-range contribution is calculated in real space, whereas the long-range contribution is calculated using a Fourier transform. The advantage of this method is the rapid convergence of the energy compared with that of a direct summation. This means that the method has high accuracy and reasonable speed when co ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Radiation Material Science
Radiation materials science is a subfield of materials science which studies the interaction of radiation with matter: a broad subject covering many forms of irradiation and of matter. Main aim of radiation material science Some of the most profound effects of irradiation on materials occur in the core of nuclear power reactors where atoms comprising the structural components are displaced numerous times over the course of their engineering lifetimes. The consequences of radiation to core components includes changes in shape and volume by tens of percent, increases in hardness by factors of five or more, severe reduction in ductility and increased embrittlement, and susceptibility to environmentally induced cracking. For these structures to fulfill their purpose, a firm understanding of the effect of radiation on materials is required in order to account for irradiation effects in design, to mitigate its effect by changing operating conditions, or to serve as a guide for creat ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Elasticity (physics)
In physics and materials science, elasticity is the ability of a body to resist a distorting influence and to return to its original size and shape when that influence or force is removed. Solid objects will deform when adequate loads are applied to them; if the material is elastic, the object will return to its initial shape and size after removal. This is in contrast to ''plasticity'', in which the object fails to do so and instead remains in its deformed state. The physical reasons for elastic behavior can be quite different for different materials. In metals, the atomic lattice changes size and shape when forces are applied (energy is added to the system). When forces are removed, the lattice goes back to the original lower energy state. For rubbers and other polymers, elasticity is caused by the stretching of polymer chains when forces are applied. Hooke's law states that the force required to deform elastic objects should be directly proportional to the distance of ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Coulomb Potential
The electric potential (also called the ''electric field potential'', potential drop, the electrostatic potential) is defined as the amount of work energy needed to move a unit of electric charge from a reference point to the specific point in an electric field. More precisely, it is the energy per unit charge for a test charge that is so small that the disturbance of the field under consideration is negligible. Furthermore, the motion across the field is supposed to proceed with negligible acceleration, so as to avoid the test charge acquiring kinetic energy or producing radiation. By definition, the electric potential at the reference point is zero units. Typically, the reference point is earth or a point at infinity, although any point can be used. In classical electrostatics, the electrostatic field is a vector quantity expressed as the gradient of the electrostatic potential, which is a scalar quantity denoted by or occasionally , equal to the electric potential energy ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Buckingham Potential
In theoretical chemistry, the Buckingham potential is a formula proposed by Richard Buckingham which describes the Pauli exclusion principle and van der Waals energy \Phi_(r) for the interaction of two atoms that are not directly bonded as a function of the interatomic distance r. It is a variety of interatomic potentials. :\Phi_(r) = A \exp \left(-Br\right) - \frac Here, A, B and C are constants. The two terms on the right-hand side constitute a repulsion and an attraction, because their first derivatives with respect to r are negative and positive, respectively. Buckingham proposed this as a simplification of the Lennard-Jones potential, in a theoretical study of the equation of state for gaseous helium, neon and argon. As explained in Buckingham's original paper and, e.g., in section 2.2.5 of Jensen's text,F. Jensen, ''Introduction to Computational Chemistry'', 2nd ed., Wiley, 2007, the repulsion is due to the interpenetration of the closed electron shells. "There is ther ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
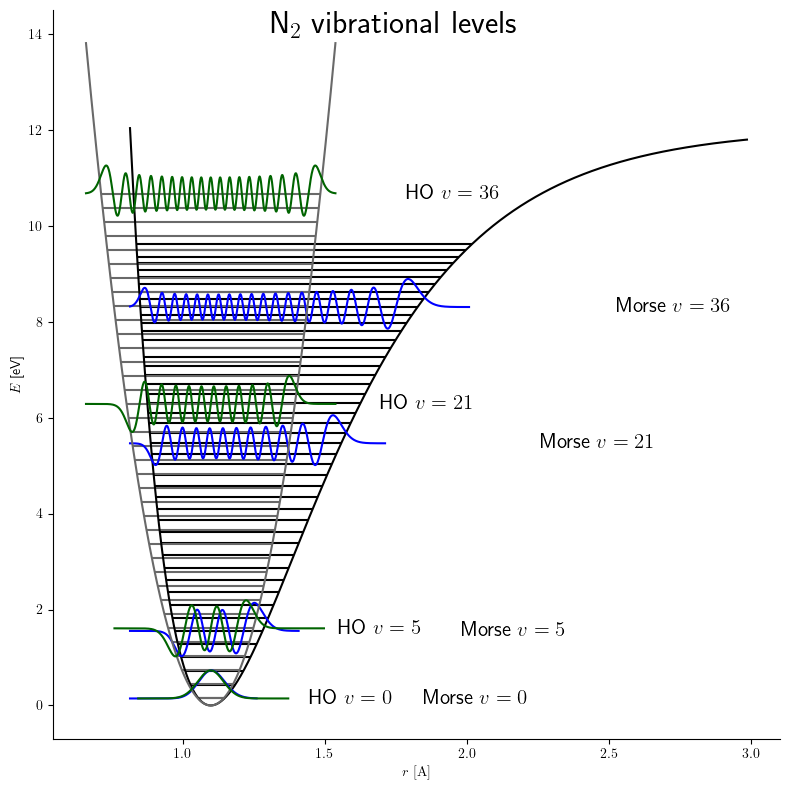
Morse Potential
The Morse potential, named after physicist Philip M. Morse, is a convenient interatomic interaction model for the potential energy of a diatomic molecule. It is a better approximation for the vibrational structure of the molecule than the quantum harmonic oscillator because it explicitly includes the effects of bond breaking, such as the existence of unbound states. It also accounts for the anharmonicity of real bonds and the non-zero transition probability for overtone and combination bands. The Morse potential can also be used to model other interactions such as the interaction between an atom and a surface. Due to its simplicity (only three fitting parameters), it is not used in modern spectroscopy. However, its mathematical form inspired the MLR ( Morse/Long-range) potential, which is the most popular potential energy function used for fitting spectroscopic data. Potential energy function The Morse potential energy function is of the form :V(r) = D_e ( 1-e^ )^2 Here r is ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Force Field (chemistry)
In the context of chemistry and molecular modelling, a force field is a computational method that is used to estimate the forces between atoms within molecules and also between molecules. More precisely, the force field refers to the functional form and parameter sets used to calculate the potential energy of a system of atoms or coarse-grained particles in molecular mechanics, molecular dynamics, or Monte Carlo simulations. The parameters for a chosen energy function may be derived from experiments in physics and chemistry, calculations in quantum mechanics, or both. Force fields are interatomic potentials and utilize the same concept as force fields in classical physics, with the difference that the force field parameters in chemistry describe the energy landscape, from which the acting forces on every particle are derived as a gradient of the potential energy with respect to the particle coordinates. ''All-atom'' force fields provide parameters for every type of atom in ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Van Der Waals Forces
In molecular physics, the van der Waals force is a distance-dependent interaction between atoms or molecules. Unlike ionic bond, ionic or covalent bonds, these attractions do not result from a Chemical bond, chemical electronic bond; they are comparatively weak and therefore more susceptible to disturbance. The van der Waals force quickly vanishes at longer distances between interacting molecules. Named after Dutch physicist Johannes Diderik van der Waals, the van der Waals force plays a fundamental role in fields as diverse as supramolecular chemistry, structural biology, polymer science, nanotechnology, surface science, and condensed matter physics. It also underlies many properties of Organic chemistry, organic compounds and molecular solids, including their solubility in Chemical polarity, polar and non-polar media. If no other force is present, the distance between atoms at which the force becomes repulsive rather than attractive as the atoms approach one another is called ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Potential Well
A potential well is the region surrounding a local minimum of potential energy. Energy captured in a potential well is unable to convert to another type of energy (kinetic energy in the case of a gravitational potential well) because it is captured in the local minimum of a potential well. Therefore, a body may not proceed to the global minimum of potential energy, as it would naturally tend to do due to entropy. Overview Energy may be released from a potential well if sufficient energy is added to the system such that the local maximum is surmounted. In quantum physics, potential energy may escape a potential well without added energy due to the probabilistic characteristics of quantum particles; in these cases a particle may be imagined to tunnel ''through'' the walls of a potential well. The graph of a 2D potential energy function is a potential energy surface that can be imagined as the Earth's surface in a landscape of hills and valleys. Then a potential well would be a val ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Lennard-Jones Potential
The Lennard-Jones potential (also termed the LJ potential or 12-6 potential) is an intermolecular pair potential. Out of all the intermolecular potentials, the Lennard-Jones potential is probably the one that has been the most extensively studied. It is considered an archetype model for simple yet realistic intermolecular interactions. The Lennard-Jones potential models soft repulsive and attractive ( van der Waals) interactions. Hence, the Lennard-Jones potential describes electronically neutral atoms or molecules. It is named after John Lennard-Jones. The commonly used expression for the Lennard-Jones potential is V_\text(r) = 4\varepsilon \left \left(\frac\right)^ - \left(\frac\right)^6 \right, where r is the distance between two interacting particles, \varepsilon is the depth of the potential well (usually referred to as 'dispersion energy'), and \sigma is the distance at which the particle-particle potential energy V is zero (often referred to as 'size of the particle'). The ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Approximation
An approximation is anything that is intentionally similar but not exactly equality (mathematics), equal to something else. Etymology and usage The word ''approximation'' is derived from Latin ''approximatus'', from ''proximus'' meaning ''very near'' and the prefix ''ad-'' (''ad-'' before ''p'' becomes ap- by assimilation (phonology), assimilation) meaning ''to''. Words like ''approximate'', ''approximately'' and ''approximation'' are used especially in technical or scientific contexts. In everyday English, words such as ''roughly'' or ''around'' are used with a similar meaning. It is often found abbreviated as ''approx.'' The term can be applied to various properties (e.g., value, quantity, image, description) that are nearly, but not exactly correct; similar, but not exactly the same (e.g., the approximate time was 10 o'clock). Although approximation is most often applied to numbers, it is also frequently applied to such things as Function (mathematics), mathematical functio ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |