Pure Alcohol on:
[Wikipedia]
[Google]
[Amazon]
Ethanol (also called ethyl alcohol, grain alcohol, drinking alcohol, or simply alcohol) is an
 The largest single use of ethanol is as an engine fuel and fuel additive. Brazil in particular relies heavily upon the use of ethanol as an engine fuel, due in part to its role as one of the world's leading producers of ethanol. Gasoline sold in Brazil contains at least 25% anhydrous ethanol. Hydrous ethanol (about 95% ethanol and 5% water) can be used as fuel in more than 90% of new gasoline-fueled cars sold in the country.
The US and many other countries primarily use E10 (10% ethanol, sometimes known as gasohol) and E85 (85% ethanol) ethanol/gasoline mixtures. Over time, it is believed that a material portion of the ≈ per year market for gasoline will begin to be replaced with fuel ethanol.
The largest single use of ethanol is as an engine fuel and fuel additive. Brazil in particular relies heavily upon the use of ethanol as an engine fuel, due in part to its role as one of the world's leading producers of ethanol. Gasoline sold in Brazil contains at least 25% anhydrous ethanol. Hydrous ethanol (about 95% ethanol and 5% water) can be used as fuel in more than 90% of new gasoline-fueled cars sold in the country.
The US and many other countries primarily use E10 (10% ethanol, sometimes known as gasohol) and E85 (85% ethanol) ethanol/gasoline mixtures. Over time, it is believed that a material portion of the ≈ per year market for gasoline will begin to be replaced with fuel ethanol.
 Australian law limits the use of pure ethanol from sugarcane waste to 10% in automobiles. Older cars (and vintage cars designed to use a slower burning fuel) should have the engine valves upgraded or replaced.
According to an industry advocacy group, ethanol as a fuel reduces harmful tailpipe emissions of carbon monoxide, particulate matter, oxides of nitrogen, and other ozone-forming pollutants. Argonne National Laboratory analyzed greenhouse gas emissions of many different engine and fuel combinations, and found that biodiesel/petrodiesel blend (B20 (biodiesel), B20) showed a reduction of 8%, conventional E85 ethanol blend a reduction of 17% and cellulosic ethanol 64%, compared with pure gasoline. Ethanol has a much greater research octane number (RON) than gasoline, meaning it is less prone to pre-ignition, allowing for better ignition advance which means more torque, and efficiency in addition to the lower carbon emissions.
Ethanol combustion in an internal combustion engine yields many of the products of incomplete combustion produced by gasoline and significantly larger amounts of formaldehyde and related species such as acetaldehyde. This leads to a significantly larger photochemical reactivity and more ground level ozone. This data has been assembled into The Clean Fuels Report comparison of fuel emissions and show that ethanol exhaust generates 2.14 times as much ozone as gasoline exhaust. When this is added into the custom ''Localized Pollution Index'' of The Clean Fuels Report, the local pollution of ethanol (pollution that contributes to smog) is rated 1.7, where gasoline is 1.0 and higher numbers signify greater pollution. The California Air Resources Board formalized this issue in 2008 by recognizing control standards for formaldehydes as an emissions control group, much like the conventional NOx and reactive organic gases (ROGs).
More than 20% of Brazilian cars are able to use 100% ethanol as fuel, which includes ethanol-only engines and flex-fuel engines. Flex-fuel engines in Brazil are able to work with all ethanol, all gasoline or any mixture of both. In the United States, flex-fuel vehicles can run on 0% to 85% ethanol (15% gasoline) since higher ethanol blends are not yet allowed or efficient. Brazil supports this fleet of ethanol-burning automobiles with large national infrastructure that produces ethanol from domestically grown sugarcane.
Ethanol's high miscibility with water makes it unsuitable for shipping through modern Pipeline transport, pipelines like liquid hydrocarbons. Mechanics have seen increased cases of damage to small engines (in particular, the carburetor) and attribute the damage to the increased water retention by ethanol in fuel.
Ethanol was commonly used as fuel in early bipropellant rocket (liquid-propelled) vehicles, in conjunction with an oxidizer such as liquid oxygen. The German A-4 ballistic rocket of World War II (better known by its propaganda name ), which is credited as having begun the space age, used ethanol as the main constituent of . Under such nomenclature, the ethanol was mixed with 25% water to reduce the combustion chamber temperature. The design team helped develop U.S. rockets following World War II, including the ethanol-fueled Redstone (rocket family), Redstone rocket, which launched the first U.S. astronaut on suborbital spaceflight. Alcohols fell into general disuse as more energy-dense rocket fuels were developed, although ethanol was used in recent experimental Light aircraft, lightweight Mark-III X-racer, rocket-powered racing aircraft.
Commercial fuel cells operate on reformed natural gas, hydrogen or methanol. Ethanol is an attractive alternative due to its wide availability, low cost, high purity and low toxicity. There is a wide range of fuel cell concepts that have entered trials including direct-ethanol fuel cells, auto-thermal reforming systems and thermally integrated systems. The majority of work is being conducted at a research level although there are a number of organizations at the beginning of the commercialization of ethanol fuel cells.
Ethanol fireplaces can be used for home heating or for decoration. Ethanol can also be used as stove fuel for cooking.
Australian law limits the use of pure ethanol from sugarcane waste to 10% in automobiles. Older cars (and vintage cars designed to use a slower burning fuel) should have the engine valves upgraded or replaced.
According to an industry advocacy group, ethanol as a fuel reduces harmful tailpipe emissions of carbon monoxide, particulate matter, oxides of nitrogen, and other ozone-forming pollutants. Argonne National Laboratory analyzed greenhouse gas emissions of many different engine and fuel combinations, and found that biodiesel/petrodiesel blend (B20 (biodiesel), B20) showed a reduction of 8%, conventional E85 ethanol blend a reduction of 17% and cellulosic ethanol 64%, compared with pure gasoline. Ethanol has a much greater research octane number (RON) than gasoline, meaning it is less prone to pre-ignition, allowing for better ignition advance which means more torque, and efficiency in addition to the lower carbon emissions.
Ethanol combustion in an internal combustion engine yields many of the products of incomplete combustion produced by gasoline and significantly larger amounts of formaldehyde and related species such as acetaldehyde. This leads to a significantly larger photochemical reactivity and more ground level ozone. This data has been assembled into The Clean Fuels Report comparison of fuel emissions and show that ethanol exhaust generates 2.14 times as much ozone as gasoline exhaust. When this is added into the custom ''Localized Pollution Index'' of The Clean Fuels Report, the local pollution of ethanol (pollution that contributes to smog) is rated 1.7, where gasoline is 1.0 and higher numbers signify greater pollution. The California Air Resources Board formalized this issue in 2008 by recognizing control standards for formaldehydes as an emissions control group, much like the conventional NOx and reactive organic gases (ROGs).
More than 20% of Brazilian cars are able to use 100% ethanol as fuel, which includes ethanol-only engines and flex-fuel engines. Flex-fuel engines in Brazil are able to work with all ethanol, all gasoline or any mixture of both. In the United States, flex-fuel vehicles can run on 0% to 85% ethanol (15% gasoline) since higher ethanol blends are not yet allowed or efficient. Brazil supports this fleet of ethanol-burning automobiles with large national infrastructure that produces ethanol from domestically grown sugarcane.
Ethanol's high miscibility with water makes it unsuitable for shipping through modern Pipeline transport, pipelines like liquid hydrocarbons. Mechanics have seen increased cases of damage to small engines (in particular, the carburetor) and attribute the damage to the increased water retention by ethanol in fuel.
Ethanol was commonly used as fuel in early bipropellant rocket (liquid-propelled) vehicles, in conjunction with an oxidizer such as liquid oxygen. The German A-4 ballistic rocket of World War II (better known by its propaganda name ), which is credited as having begun the space age, used ethanol as the main constituent of . Under such nomenclature, the ethanol was mixed with 25% water to reduce the combustion chamber temperature. The design team helped develop U.S. rockets following World War II, including the ethanol-fueled Redstone (rocket family), Redstone rocket, which launched the first U.S. astronaut on suborbital spaceflight. Alcohols fell into general disuse as more energy-dense rocket fuels were developed, although ethanol was used in recent experimental Light aircraft, lightweight Mark-III X-racer, rocket-powered racing aircraft.
Commercial fuel cells operate on reformed natural gas, hydrogen or methanol. Ethanol is an attractive alternative due to its wide availability, low cost, high purity and low toxicity. There is a wide range of fuel cell concepts that have entered trials including direct-ethanol fuel cells, auto-thermal reforming systems and thermally integrated systems. The majority of work is being conducted at a research level although there are a number of organizations at the beginning of the commercialization of ethanol fuel cells.
Ethanol fireplaces can be used for home heating or for decoration. Ethanol can also be used as stove fuel for cooking.
 Ethanol is a volatile, colorless liquid that has a slight odor. It burns with a smokeless blue flame that is not always visible in normal light. The physical properties of ethanol stem primarily from the presence of its hydroxyl group and the shortness of its carbon chain. Ethanol's hydroxyl group is able to participate in hydrogen bonding, rendering it more viscous and less volatile than less polar organic compounds of similar molecular weight, such as propane. Ethanol's adiabatic flame temperature for combustion in air is 2082 °C or 3779 °F.
Ethanol is slightly more refractive than water, having a refractive index of 1.36242 (at λ=589.3 nm and ). The triple point for ethanol is .
Ethanol is a volatile, colorless liquid that has a slight odor. It burns with a smokeless blue flame that is not always visible in normal light. The physical properties of ethanol stem primarily from the presence of its hydroxyl group and the shortness of its carbon chain. Ethanol's hydroxyl group is able to participate in hydrogen bonding, rendering it more viscous and less volatile than less polar organic compounds of similar molecular weight, such as propane. Ethanol's adiabatic flame temperature for combustion in air is 2082 °C or 3779 °F.
Ethanol is slightly more refractive than water, having a refractive index of 1.36242 (at λ=589.3 nm and ). The triple point for ethanol is .
 Hydrogen bonding causes pure ethanol to be hygroscopic to the extent that it readily absorbs water from the air. The polar nature of the hydroxyl group causes ethanol to dissolve many ionic compounds, notably sodium hydroxide, sodium and potassium hydroxides, magnesium chloride, calcium chloride, ammonium chloride, ammonium bromide, and sodium bromide. Sodium chloride, Sodium and potassium chlorides are slightly soluble in ethanol. Because the ethanol molecule also has a nonpolar end, it will also dissolve nonpolar substances, including most essential oils''Merck Index of Chemicals and Drugs'', 9th ed.; monographs 6575 through 6669 and numerous flavoring, coloring, and medicinal agents.
The addition of even a few percent of ethanol to water sharply reduces the surface tension of water. This property partially explains the "tears of wine" phenomenon. When wine is swirled in a glass, ethanol evaporates quickly from the thin film of wine on the wall of the glass. As the wine's ethanol content decreases, its surface tension increases and the thin film "beads up" and runs down the glass in channels rather than as a smooth sheet.
Hydrogen bonding causes pure ethanol to be hygroscopic to the extent that it readily absorbs water from the air. The polar nature of the hydroxyl group causes ethanol to dissolve many ionic compounds, notably sodium hydroxide, sodium and potassium hydroxides, magnesium chloride, calcium chloride, ammonium chloride, ammonium bromide, and sodium bromide. Sodium chloride, Sodium and potassium chlorides are slightly soluble in ethanol. Because the ethanol molecule also has a nonpolar end, it will also dissolve nonpolar substances, including most essential oils''Merck Index of Chemicals and Drugs'', 9th ed.; monographs 6575 through 6669 and numerous flavoring, coloring, and medicinal agents.
The addition of even a few percent of ethanol to water sharply reduces the surface tension of water. This property partially explains the "tears of wine" phenomenon. When wine is swirled in a glass, ethanol evaporates quickly from the thin film of wine on the wall of the glass. As the wine's ethanol content decreases, its surface tension increases and the thin film "beads up" and runs down the glass in channels rather than as a smooth sheet.
 Ethanol is produced both as a
Ethanol is produced both as a
 Breweries and biofuel plants employ two methods for measuring ethanol concentration. Infrared ethanol sensors measure the vibrational frequency of dissolved ethanol using the C−H band at 2900 cm. This method uses a relatively inexpensive solid-state sensor that compares the C−H band with a reference band to calculate the ethanol content. The calculation makes use of the Beer–Lambert law. Alternatively, by measuring the density of the starting material and the density of the product, using a hydrometer, the change in specific gravity during fermentation indicates the alcohol content. This inexpensive and indirect method has a long history in the beer brewing industry.
Breweries and biofuel plants employ two methods for measuring ethanol concentration. Infrared ethanol sensors measure the vibrational frequency of dissolved ethanol using the C−H band at 2900 cm. This method uses a relatively inexpensive solid-state sensor that compares the C−H band with a reference band to calculate the ethanol content. The calculation makes use of the Beer–Lambert law. Alternatively, by measuring the density of the starting material and the density of the product, using a hydrometer, the change in specific gravity during fermentation indicates the alcohol content. This inexpensive and indirect method has a long history in the beer brewing industry.
Alcohol (Ethanol)
at ''The Periodic Table of Videos'' (University of Nottingham)
International Labour Organization
ethanol safety information
* [http://webbook.nist.gov/cgi/cbook.cgi?Name=ethanol&Units=SI National Institute of Standards and Technology] chemical data on ethanol
Chicago Board of Trade
news and market data on ethanol futures * Calculation o
vapor pressureliquid densitydynamic liquid viscositysurface tension
of ethanol
Ethanol History
A look into the history of ethanol
Industrial ethanol production process flow diagram using ethylene and sulphuric acid
{{Authority control Ethanol, Alcohol solvents Alkanols Anatomical preservation Chemical hazards Commodity chemicals Disinfectants Hepatotoxins Household chemicals Human metabolites IARC Group 1 carcinogens Oxygenates Primary alcohols Rocket fuels Teratogens Alcohol chemistry
organic compound
Some chemical authorities define an organic compound as a chemical compound that contains a carbon–hydrogen or carbon–carbon bond; others consider an organic compound to be any chemical compound that contains carbon. For example, carbon-co ...
with the chemical formula
A chemical formula is a way of presenting information about the chemical proportions of atoms that constitute a particular chemical compound or molecule, using chemical element symbols, numbers, and sometimes also other symbols, such as pare ...
. It is an alcohol
Alcohol may refer to:
Common uses
* Alcohol (chemistry), a class of compounds
* Ethanol, one of several alcohols, commonly known as alcohol in everyday life
** Alcohol (drug), intoxicant found in alcoholic beverages
** Alcoholic beverage, an alco ...
, with its formula also written as , or EtOH, where Et is the pseudoelement symbol
The skeletal formula, line-angle formula, bond-line formula or shorthand formula of an organic compound is a type of minimalist structural formula representing a molecule's atoms, bonds and some details of its geometry. The lines in a skeleta ...
for ethyl. Ethanol is a volatile, flammable
A combustible material is a material that can burn (i.e., sustain a flame) in air under certain conditions. A material is flammable if it ignites easily at ambient temperatures. In other words, a combustible material ignites with some effort ...
, colorless liquid with a characteristic wine
Wine is an alcoholic drink made from Fermentation in winemaking, fermented fruit. Yeast in winemaking, Yeast consumes the sugar in the fruit and converts it to ethanol and carbon dioxide, releasing heat in the process. Wine is most often made f ...
-like odor and pungent
Pungency ( ) is the taste of food commonly referred to as spiciness, hotness or heat, found in foods such as chili peppers. Highly pungent tastes may be experienced as unpleasant. The term piquancy ( ) is sometimes applied to foods with a lower ...
taste. As a psychoactive
A psychoactive drug, psychopharmaceutical, mind-altering drug, consciousness-altering drug, psychoactive substance, or psychotropic substance is a chemical substance that alters psychological functioning by modulating central nervous system acti ...
depressant
Depressants, also known as central nervous system depressants, or colloquially known as "downers", are drugs that lower neurotransmission levels, decrease the electrical activity of brain cells, or reduce arousal or stimulation in various ...
, it is the active ingredient in alcoholic beverage
Drinks containing alcohol (drug), alcohol are typically divided into three classes—beers, wines, and Distilled beverage, spirits—with alcohol content typically between 3% and 50%. Drinks with less than 0.5% are sometimes considered Non-al ...
s, and the second most consumed drug globally behind caffeine
Caffeine is a central nervous system (CNS) stimulant of the methylxanthine chemical classification, class and is the most commonly consumed Psychoactive drug, psychoactive substance globally. It is mainly used for its eugeroic (wakefulness pr ...
.
Ethanol is naturally produced by the fermentation
Fermentation is a type of anaerobic metabolism which harnesses the redox potential of the reactants to make adenosine triphosphate (ATP) and organic end products. Organic molecules, such as glucose or other sugars, are catabolized and reduce ...
process of sugar
Sugar is the generic name for sweet-tasting, soluble carbohydrates, many of which are used in food. Simple sugars, also called monosaccharides, include glucose
Glucose is a sugar with the Chemical formula#Molecular formula, molecul ...
s by yeast
Yeasts are eukaryotic, single-celled microorganisms classified as members of the fungus kingdom (biology), kingdom. The first yeast originated hundreds of millions of years ago, and at least 1,500 species are currently recognized. They are est ...
s or via petrochemical
Petrochemicals (sometimes abbreviated as petchems) are the chemical products obtained from petroleum by refining. Some chemical compounds made from petroleum are also obtained from other fossil fuels, such as coal or natural gas, or renewable s ...
processes such as ethylene
Ethylene (IUPAC name: ethene) is a hydrocarbon which has the formula or . It is a colourless, flammable gas with a faint "sweet and musky" odour when pure. It is the simplest alkene (a hydrocarbon with carbon–carbon bond, carbon–carbon doub ...
hydration. Historically it was used as a general anesthetic
General anaesthetics (or anesthetics) are often defined as compounds that induce a loss of consciousness in humans or loss of righting reflex in animals. Clinical definitions are also extended to include an induced coma that causes lack of aware ...
, and has modern medical applications as an antiseptic
An antiseptic ( and ) is an antimicrobial substance or compound that is applied to living tissue to reduce the possibility of sepsis, infection, or putrefaction. Antiseptics are generally distinguished from ''antibiotics'' by the latter's abil ...
, disinfectant
A disinfectant is a chemical substance or compound used to inactivate or destroy microorganisms on inert surfaces. Disinfection does not necessarily kill all microorganisms, especially resistant bacterial spores; it is less effective than ...
, solvent for some medications, and antidote
An antidote is a substance that can counteract a form of poisoning. The term ultimately derives from the Greek term φάρμακον ἀντίδοτον ''(pharmakon antidoton)'', "(medicine) given as a remedy". An older term in English which is ...
for methanol poisoning
Methanol toxicity (also methanol poisoning) is poisoning from methanol, characteristically via ingestion. Symptoms may include an altered/decreased level of consciousness, poor or no coordination, vomiting, abdominal pain, and a specific smell o ...
and ethylene glycol poisoning
Ethylene glycol poisoning is poisoning caused by drinking ethylene glycol. Early symptoms include intoxication, vomiting and abdominal pain. Later symptoms may include a decreased level of consciousness, headache, and seizures. Long term outco ...
. It is used as a chemical solvent
A solvent (from the Latin language, Latin ''wikt:solvo#Latin, solvō'', "loosen, untie, solve") is a substance that dissolves a solute, resulting in a Solution (chemistry), solution. A solvent is usually a liquid but can also be a solid, a gas ...
and in the synthesis
Synthesis or synthesize may refer to:
Science Chemistry and biochemistry
*Chemical synthesis, the execution of chemical reactions to form a more complex molecule from chemical precursors
**Organic synthesis, the chemical synthesis of organi ...
of organic compounds, and as a fuel source for lamps, stoves, and internal combustion engines. Ethanol also can be dehydrated to make ethylene, an important chemical feedstock. As of 2023, world production of ethanol fuel was , coming mostly from the U.S. (51%) and Brazil (26%).
The term “ethanol,” originates from the ethyl group
In organic chemistry, an ethyl group (abbr. Et) is an alkyl substituent with the formula , derived from ethane (). ''Ethyl'' is used in the International Union of Pure and Applied Chemistry
The International Union of Pure and Applied ...
coined in 1834 and was officially adopted in 1892, while “alcohol”—now referring broadly to similar compounds—originally described a powdered cosmetic and only later came to mean ethanol specifically. Ethanol occurs naturally as a byproduct of yeast
Yeasts are eukaryotic, single-celled microorganisms classified as members of the fungus kingdom (biology), kingdom. The first yeast originated hundreds of millions of years ago, and at least 1,500 species are currently recognized. They are est ...
metabolism in environments like overripe fruit and palm blossoms, during plant germination
Germination is the process by which an organism grows from a seed or spore. The term is applied to the sprouting of a seedling from a seed of an angiosperm or gymnosperm, the growth of a sporeling from a spore, such as the spores of fungi, ...
under anaerobic
Anaerobic means "living, active, occurring, or existing in the absence of free oxygen", as opposed to aerobic which means "living, active, or occurring only in the presence of oxygen." Anaerobic may also refer to:
*Adhesive#Anaerobic, Anaerobic ad ...
conditions, in interstellar space, in human breath, and in rare cases, is produced internally due to auto-brewery syndrome
Auto-brewery syndrome (ABS) (also known as gut fermentation syndrome, endogenous ethanol fermentation or drunkenness disease) is a condition characterized by the fermentation of ingested carbohydrates in the gastrointestinal tract of the body cau ...
.
Ethanol has been used since ancient times as an intoxicant
A psychoactive drug, psychopharmaceutical, mind-altering drug, consciousness-altering drug, psychoactive substance, or psychotropic substance is a chemical substance that alters psychological functioning by modulating central nervous system acti ...
. Production through fermentation
Fermentation is a type of anaerobic metabolism which harnesses the redox potential of the reactants to make adenosine triphosphate (ATP) and organic end products. Organic molecules, such as glucose or other sugars, are catabolized and reduce ...
and distillation
Distillation, also classical distillation, is the process of separating the component substances of a liquid mixture of two or more chemically discrete substances; the separation process is realized by way of the selective boiling of the mixt ...
evolved over centuries across various cultures. Chemical identification and synthetic production began by the 19th century.
Name
''Ethanol'' is thesystematic name
A systematic name is a name given in a systematic way to one unique group, organism, object or chemical substance, out of a specific population or collection. Systematic names are usually part of a nomenclature.
A semisystematic name or semitrivi ...
defined by the International Union of Pure and Applied Chemistry
The International Union of Pure and Applied Chemistry (IUPAC ) is an international federation of National Adhering Organizations working for the advancement of the chemical sciences, especially by developing nomenclature and terminology. It is ...
for a compound consisting of an alkyl group
In organic chemistry, an alkyl group is an alkane missing one hydrogen.
The term ''alkyl'' is intentionally unspecific to include many possible substitutions.
An acyclic alkyl has the general formula of . A cycloalkyl group is derived from a cy ...
with two carbon atom
Atoms are the basic particles of the chemical elements. An atom consists of a atomic nucleus, nucleus of protons and generally neutrons, surrounded by an electromagnetically bound swarm of electrons. The chemical elements are distinguished fr ...
s (prefix "eth-"), having a single bond between them (infix "-an-") and an attached −OH functional group
In organic chemistry, a functional group is any substituent or moiety (chemistry), moiety in a molecule that causes the molecule's characteristic chemical reactions. The same functional group will undergo the same or similar chemical reactions r ...
(suffix "-ol").
The "eth-" prefix and the qualifier "ethyl" in "ethyl alcohol" originally came from the name "ethyl" assigned in 1834 to the group − by Justus Liebig. He coined the word from the German
German(s) may refer to:
* Germany, the country of the Germans and German things
**Germania (Roman era)
* Germans, citizens of Germany, people of German ancestry, or native speakers of the German language
** For citizenship in Germany, see also Ge ...
name ''Aether'' of the compound −O− (commonly called "ether" in English, more specifically called "diethyl ether
Diethyl ether, or simply ether, is an organic compound with the chemical formula , sometimes abbreviated as . It is a colourless, highly Volatility (chemistry), volatile, sweet-smelling ("ethereal odour"), extremely flammable liquid. It belongs ...
"). According to the ''Oxford English Dictionary
The ''Oxford English Dictionary'' (''OED'') is the principal historical dictionary of the English language, published by Oxford University Press (OUP), a University of Oxford publishing house. The dictionary, which published its first editio ...
'', ''Ethyl'' is a contraction of the Ancient Greek αἰθήρ (', "upper air") and the Greek word ὕλη (', "wood, raw material", hence "matter, substance"). ''Ethanol'' was coined as a result of a resolution on naming alcohols and phenols that was adopted at the International Conference on Chemical Nomenclature
Chemical nomenclature is a set of rules to generate systematic name#In chemistry, systematic names for chemical compounds. The nomenclature used most frequently worldwide is the one created and developed by the International Union of Pure and Appli ...
that was held in April 1892 in Geneva
Geneva ( , ; ) ; ; . is the List of cities in Switzerland, second-most populous city in Switzerland and the most populous in French-speaking Romandy. Situated in the southwest of the country, where the Rhône exits Lake Geneva, it is the ca ...
, Switzerland.
The term ''alcohol'' now refers to a wider class of substances in chemistry nomenclature, but in common parlance it remains the name of ethanol. It is a medieval loan from Arabic
Arabic (, , or , ) is a Central Semitic languages, Central Semitic language of the Afroasiatic languages, Afroasiatic language family spoken primarily in the Arab world. The International Organization for Standardization (ISO) assigns lang ...
, a powdered ore of antimony
Antimony is a chemical element; it has chemical symbol, symbol Sb () and atomic number 51. A lustrous grey metal or metalloid, it is found in nature mainly as the sulfide mineral stibnite (). Antimony compounds have been known since ancient t ...
used since antiquity as a cosmetic, and retained that meaning in Middle Latin
Medieval Latin was the form of Literary Latin used in Roman Catholic Western Europe during the Middle Ages. It was also the administrative language in the former Roman Provinces of Mauretania, Numidia and Africa Proconsularis under the Vandals ...
. The use of 'alcohol' for ethanol (in full, "alcohol of wine") was first recorded in 1753. Before the late 18th century the term ''alcohol'' generally referred to any sublimated substance.
Uses
Recreational drug
As acentral nervous system
The central nervous system (CNS) is the part of the nervous system consisting primarily of the brain, spinal cord and retina. The CNS is so named because the brain integrates the received information and coordinates and influences the activity o ...
depressant
Depressants, also known as central nervous system depressants, or colloquially known as "downers", are drugs that lower neurotransmission levels, decrease the electrical activity of brain cells, or reduce arousal or stimulation in various ...
, ethanol is one of the most commonly consumed psychoactive drug
A psychoactive drug, psychopharmaceutical, mind-altering drug, consciousness-altering drug, psychoactive substance, or psychotropic substance is a chemical substance that alters psychological functioning by modulating central nervous system acti ...
s. Despite alcohol's psychoactive, addictive, and carcinogenic
A carcinogen () is any agent that promotes the development of cancer. Carcinogens can include synthetic chemicals, naturally occurring substances, physical agents such as ionizing and non-ionizing radiation, and Biological agent, biologic agent ...
properties, it is readily available and legal for sale in many countries. There are laws regulating the sale, exportation/importation, taxation, manufacturing, consumption, and possession of alcoholic beverages. The most common regulation is prohibition for minors.
In mammals, ethanol is primarily metabolized
Metabolism (, from ''metabolē'', "change") is the set of life-sustaining chemical reactions in organisms. The three main functions of metabolism are: the conversion of the energy in food to energy available to run cellular processes; the co ...
in the liver
The liver is a major metabolic organ (anatomy), organ exclusively found in vertebrates, which performs many essential biological Function (biology), functions such as detoxification of the organism, and the Protein biosynthesis, synthesis of var ...
and stomach
The stomach is a muscular, hollow organ in the upper gastrointestinal tract of Human, humans and many other animals, including several invertebrates. The Ancient Greek name for the stomach is ''gaster'' which is used as ''gastric'' in medical t ...
by ADH enzymes. These enzymes catalyze the oxidation
Redox ( , , reduction–oxidation or oxidation–reduction) is a type of chemical reaction in which the oxidation states of the reactants change. Oxidation is the loss of electrons or an increase in the oxidation state, while reduction is ...
of ethanol into acetaldehyde
Acetaldehyde (IUPAC systematic name ethanal) is an organic compound, organic chemical compound with the chemical formula, formula , sometimes abbreviated as . It is a colorless liquid or gas, boiling near room temperature. It is one of the most ...
(ethanal):
:CH3CH2OH + NAD+ → CH3CHO + NADH
Nicotinamide adenine dinucleotide (NAD) is a coenzyme central to metabolism. Found in all living cells, NAD is called a dinucleotide because it consists of two nucleotides joined through their phosphate groups. One nucleotide contains an ade ...
+ H+
When present in significant concentrations, this metabolism of ethanol is additionally aided by the cytochrome P450
Cytochromes P450 (P450s or CYPs) are a Protein superfamily, superfamily of enzymes containing heme as a cofactor (biochemistry), cofactor that mostly, but not exclusively, function as monooxygenases. However, they are not omnipresent; for examp ...
enzyme CYP2E1
Cytochrome P450 2E1 (abbreviated CYP2E1, ) is a member of the cytochrome P450 mixed-function oxidase system, which is involved in the metabolism of xenobiotics in the body. This class of enzymes is divided up into a number of subcategories, inclu ...
in humans, while trace amounts are also metabolized by catalase
Catalase is a common enzyme found in nearly all living organisms exposed to oxygen (such as bacteria, plants, and animals) which catalyzes the decomposition of hydrogen peroxide to water and oxygen. It is a very important enzyme in protecting ...
. The resulting intermediate, acetaldehyde, is a known carcinogen, and poses significantly greater toxicity in humans than ethanol itself. Many of the symptoms typically associated with alcohol intoxication—as well as many of the health hazards typically associated with the long-term consumption of ethanol—can be attributed to acetaldehyde toxicity in humans.
The subsequent oxidation of acetaldehyde into acetate
An acetate is a salt formed by the combination of acetic acid with a base (e.g. alkaline, earthy, metallic, nonmetallic, or radical base). "Acetate" also describes the conjugate base or ion (specifically, the negatively charged ion called ...
is performed by aldehyde dehydrogenase
Aldehyde dehydrogenases () are a group of enzymes that catalyse the oxidation of aldehydes. They convert aldehydes (R–C(=O)) to carboxylic acids (R–C(=O)). The oxygen comes from a water molecule. To date, nineteen ALDH genes have ...
(ALDH) enzymes. A mutation in the ALDH2 gene that encodes for an inactive or dysfunctional form of this enzyme affects roughly 50% of east Asian populations, contributing to the characteristic alcohol flush reaction
Alcohol flush reaction is a condition in which a person develops flushes or blotches associated with erythema on the face, neck, shoulders, ears, and in some cases, the entire body after consuming alcoholic beverages. The reaction is the resul ...
that can cause temporary reddening of the skin as well as a number of related, and often unpleasant, symptoms of acetaldehyde toxicity. This mutation is typically accompanied by another mutation in the ADH enzyme ADH1B in roughly 80% of east Asians, which improves the catalytic efficiency of converting ethanol into acetaldehyde.
Medical
Ethanol is the oldest knownsedative
A sedative or tranquilliser is a substance that induces sedation by reducing irritability or Psychomotor agitation, excitement. They are central nervous system (CNS) Depressant, depressants and interact with brain activity, causing its decelera ...
, used as an oral general anesthetic
General anaesthetics (or anesthetics) are often defined as compounds that induce a loss of consciousness in humans or loss of righting reflex in animals. Clinical definitions are also extended to include an induced coma that causes lack of aware ...
during surgery in ancient Mesopotamia
Mesopotamia is a historical region of West Asia situated within the Tigris–Euphrates river system, in the northern part of the Fertile Crescent. Today, Mesopotamia is known as present-day Iraq and forms the eastern geographic boundary of ...
and in medieval times
In the history of Europe, the Middle Ages or medieval period lasted approximately from the 5th to the late 15th centuries, similarly to the post-classical period of global history. It began with the fall of the Western Roman Empire and t ...
. Mild intoxication starts at a blood alcohol concentration
Blood alcohol content (BAC), also called blood alcohol concentration or blood alcohol level, is a measurement of alcohol intoxication used for legal or medical purposes.
BAC is expressed as mass of alcohol per volume of blood. In US and many i ...
of 0.05% and induces anesthetic coma at 0.4%. This use carries the high risk of deadly alcohol intoxication
Alcohol intoxication, commonly described in higher doses as drunkenness or inebriation, and known in overdose as alcohol poisoning, is the behavior and physical effects caused by recent consumption of Alcohol (drug), alcohol. The technical ter ...
, pulmonary aspiration
Pulmonary aspiration is the entry of solid or liquid material such as pharyngeal secretions, food, drink, or stomach contents from the oropharynx or gastrointestinal tract, into the trachea and lungs. When pulmonary aspiration occurs during ea ...
and vomiting, which led to use of alternatives in antiquity, such as opium
Opium (also known as poppy tears, or Lachryma papaveris) is the dried latex obtained from the seed Capsule (fruit), capsules of the opium poppy ''Papaver somniferum''. Approximately 12 percent of opium is made up of the analgesic alkaloid mor ...
and cannabis
''Cannabis'' () is a genus of flowering plants in the family Cannabaceae that is widely accepted as being indigenous to and originating from the continent of Asia. However, the number of species is disputed, with as many as three species be ...
, and later diethyl ether, starting in the 1840s.Grattan, N. "Treatment of Uterine Haemorrhage". ''Provincial Medicine and Surgical Journal''. Vol. 1, No. 6 (Nov. 7, 1840), p. 107.
Ethanol is used as an antiseptic
An antiseptic ( and ) is an antimicrobial substance or compound that is applied to living tissue to reduce the possibility of sepsis, infection, or putrefaction. Antiseptics are generally distinguished from ''antibiotics'' by the latter's abil ...
in medical wipes and hand sanitizer
Hand sanitizer (also known as hand antiseptic, hand disinfectant, hand rub, or handrub) is a liquid, gel, or foam used to kill viruses, bacteria, and other microorganisms on the hands. It can also come in the form of a cream, spray, or wipe. W ...
gels for its bactericidal and anti-fungal effects. Ethanol kills microorganism
A microorganism, or microbe, is an organism of microscopic scale, microscopic size, which may exist in its unicellular organism, single-celled form or as a Colony (biology)#Microbial colonies, colony of cells. The possible existence of unseen ...
s by dissolving their membrane lipid bilayer
The lipid bilayer (or phospholipid bilayer) is a thin polar membrane made of two layers of lipid molecules. These membranes form a continuous barrier around all cell (biology), cells. The cell membranes of almost all organisms and many viruses a ...
and denaturing their protein
Proteins are large biomolecules and macromolecules that comprise one or more long chains of amino acid residue (biochemistry), residues. Proteins perform a vast array of functions within organisms, including Enzyme catalysis, catalysing metab ...
s, and is effective against most bacteria
Bacteria (; : bacterium) are ubiquitous, mostly free-living organisms often consisting of one Cell (biology), biological cell. They constitute a large domain (biology), domain of Prokaryote, prokaryotic microorganisms. Typically a few micr ...
, fungi
A fungus (: fungi , , , or ; or funguses) is any member of the group of eukaryotic organisms that includes microorganisms such as yeasts and mold (fungus), molds, as well as the more familiar mushrooms. These organisms are classified as one ...
and virus
A virus is a submicroscopic infectious agent that replicates only inside the living Cell (biology), cells of an organism. Viruses infect all life forms, from animals and plants to microorganisms, including bacteria and archaea. Viruses are ...
es. It is ineffective against bacterial spores
In biology, a spore is a unit of sexual (in fungi) or asexual reproduction that may be adapted for dispersal and for survival, often for extended periods of time, in unfavourable conditions. Spores form part of the life cycles of many plant ...
, which can be treated with hydrogen peroxide
Hydrogen peroxide is a chemical compound with the formula . In its pure form, it is a very pale blue liquid that is slightly more viscosity, viscous than Properties of water, water. It is used as an oxidizer, bleaching agent, and antiseptic, usua ...
.
A solution of 70% ethanol is more effective than pure ethanol because ethanol relies on water molecules for optimal antimicrobial activity. Absolute ethanol may inactivate microbes without destroying them because the alcohol is unable to fully permeate the microbe's membrane. Ethanol can also be used as a disinfectant and antiseptic by inducing cell dehydration through disruption of the osmotic balance across the cell membrane, causing water to leave the cell, leading to cell death.
Ethanol may be administered as an antidote
An antidote is a substance that can counteract a form of poisoning. The term ultimately derives from the Greek term φάρμακον ἀντίδοτον ''(pharmakon antidoton)'', "(medicine) given as a remedy". An older term in English which is ...
to ethylene glycol poisoning
Ethylene glycol poisoning is poisoning caused by drinking ethylene glycol. Early symptoms include intoxication, vomiting and abdominal pain. Later symptoms may include a decreased level of consciousness, headache, and seizures. Long term outco ...
and methanol poisoning
Methanol toxicity (also methanol poisoning) is poisoning from methanol, characteristically via ingestion. Symptoms may include an altered/decreased level of consciousness, poor or no coordination, vomiting, abdominal pain, and a specific smell o ...
. It does so by acting as a competitive inhibitor
Competitive inhibition is interruption of a chemical pathway owing to one chemical substance inhibiting the effect of another by competing with it for binding or bonding. Any metabolic or chemical messenger system can potentially be affected b ...
against methanol
Methanol (also called methyl alcohol and wood spirit, amongst other names) is an organic chemical compound and the simplest aliphatic Alcohol (chemistry), alcohol, with the chemical formula (a methyl group linked to a hydroxyl group, often ab ...
and ethylene glycol
Ethylene glycol ( IUPAC name: ethane-1,2-diol) is an organic compound (a vicinal diol) with the formula . It is mainly used for two purposes: as a raw material in the manufacture of polyester fibers and for antifreeze formulations. It is an odo ...
for alcohol dehydrogenase
Alcohol dehydrogenases (ADH) () are a group of dehydrogenase enzymes that occur in many organisms and facilitate the interconversion between alcohols and aldehydes or ketones with the reduction of nicotinamide adenine dinucleotide (NAD+) to N ...
(ADH). Though it has more side effects, ethanol is less expensive and more readily available than fomepizole
Fomepizole, also known as 4-methylpyrazole, is a medication used to treat methanol and ethylene glycol poisoning. It may be used alone or together with hemodialysis. It is given by injection into a vein.
Common side effects include headache ...
in the role.
Ethanol is used to dissolve many water-insoluble medications and related compounds. Liquid preparations of pain medications, cough and cold medicines, and mouth washes, for example, may contain up to 25% ethanol and may need to be avoided in individuals with adverse reactions to ethanol such as alcohol-induced respiratory reactions. Ethanol is present mainly as an antimicrobial preservative in over 700 liquid preparations of medicine including acetaminophen
Paracetamol, or acetaminophen, is a non-opioid analgesic and antipyretic agent used to treat fever and mild to moderate pain. It is a widely available over-the-counter drug sold under various brand names, including Tylenol and Panadol.
Parac ...
, iron supplements, ranitidine
Ranitidine, previously sold under the brand name Zantac among others, is a medication used to decrease stomach acid production. It was commonly used in treatment of peptic ulcer disease, gastroesophageal reflux disease, and Zollinger–Ellis ...
, furosemide
Furosemide, sold under the brand name Lasix among others, is a loop diuretic medication used to treat edema due to heart failure, liver scarring, or kidney disease. Furosemide may also be used for the treatment of high blood pressure. It can ...
, mannitol
Mannitol is a type of sugar alcohol used as a sweetener and medication. It is used as a low calorie sweetener as it is poorly absorbed by the intestines. As a medication, it is used to decrease pressure in the eyes, as in glaucoma, and to l ...
, phenobarbital
Phenobarbital, also known as phenobarbitone or phenobarb, sold under the brand name Luminal among others, is a medication of the barbiturate type. It is recommended by the World Health Organization (WHO) for the treatment of certain types of ...
, trimethoprim/sulfamethoxazole and over-the-counter cough medicine.
Some medicinal solutions of ethanol are also known as tinctures.
Energy source
 The largest single use of ethanol is as an engine fuel and fuel additive. Brazil in particular relies heavily upon the use of ethanol as an engine fuel, due in part to its role as one of the world's leading producers of ethanol. Gasoline sold in Brazil contains at least 25% anhydrous ethanol. Hydrous ethanol (about 95% ethanol and 5% water) can be used as fuel in more than 90% of new gasoline-fueled cars sold in the country.
The US and many other countries primarily use E10 (10% ethanol, sometimes known as gasohol) and E85 (85% ethanol) ethanol/gasoline mixtures. Over time, it is believed that a material portion of the ≈ per year market for gasoline will begin to be replaced with fuel ethanol.
The largest single use of ethanol is as an engine fuel and fuel additive. Brazil in particular relies heavily upon the use of ethanol as an engine fuel, due in part to its role as one of the world's leading producers of ethanol. Gasoline sold in Brazil contains at least 25% anhydrous ethanol. Hydrous ethanol (about 95% ethanol and 5% water) can be used as fuel in more than 90% of new gasoline-fueled cars sold in the country.
The US and many other countries primarily use E10 (10% ethanol, sometimes known as gasohol) and E85 (85% ethanol) ethanol/gasoline mixtures. Over time, it is believed that a material portion of the ≈ per year market for gasoline will begin to be replaced with fuel ethanol.
 Australian law limits the use of pure ethanol from sugarcane waste to 10% in automobiles. Older cars (and vintage cars designed to use a slower burning fuel) should have the engine valves upgraded or replaced.
According to an industry advocacy group, ethanol as a fuel reduces harmful tailpipe emissions of carbon monoxide, particulate matter, oxides of nitrogen, and other ozone-forming pollutants. Argonne National Laboratory analyzed greenhouse gas emissions of many different engine and fuel combinations, and found that biodiesel/petrodiesel blend (B20 (biodiesel), B20) showed a reduction of 8%, conventional E85 ethanol blend a reduction of 17% and cellulosic ethanol 64%, compared with pure gasoline. Ethanol has a much greater research octane number (RON) than gasoline, meaning it is less prone to pre-ignition, allowing for better ignition advance which means more torque, and efficiency in addition to the lower carbon emissions.
Ethanol combustion in an internal combustion engine yields many of the products of incomplete combustion produced by gasoline and significantly larger amounts of formaldehyde and related species such as acetaldehyde. This leads to a significantly larger photochemical reactivity and more ground level ozone. This data has been assembled into The Clean Fuels Report comparison of fuel emissions and show that ethanol exhaust generates 2.14 times as much ozone as gasoline exhaust. When this is added into the custom ''Localized Pollution Index'' of The Clean Fuels Report, the local pollution of ethanol (pollution that contributes to smog) is rated 1.7, where gasoline is 1.0 and higher numbers signify greater pollution. The California Air Resources Board formalized this issue in 2008 by recognizing control standards for formaldehydes as an emissions control group, much like the conventional NOx and reactive organic gases (ROGs).
More than 20% of Brazilian cars are able to use 100% ethanol as fuel, which includes ethanol-only engines and flex-fuel engines. Flex-fuel engines in Brazil are able to work with all ethanol, all gasoline or any mixture of both. In the United States, flex-fuel vehicles can run on 0% to 85% ethanol (15% gasoline) since higher ethanol blends are not yet allowed or efficient. Brazil supports this fleet of ethanol-burning automobiles with large national infrastructure that produces ethanol from domestically grown sugarcane.
Ethanol's high miscibility with water makes it unsuitable for shipping through modern Pipeline transport, pipelines like liquid hydrocarbons. Mechanics have seen increased cases of damage to small engines (in particular, the carburetor) and attribute the damage to the increased water retention by ethanol in fuel.
Ethanol was commonly used as fuel in early bipropellant rocket (liquid-propelled) vehicles, in conjunction with an oxidizer such as liquid oxygen. The German A-4 ballistic rocket of World War II (better known by its propaganda name ), which is credited as having begun the space age, used ethanol as the main constituent of . Under such nomenclature, the ethanol was mixed with 25% water to reduce the combustion chamber temperature. The design team helped develop U.S. rockets following World War II, including the ethanol-fueled Redstone (rocket family), Redstone rocket, which launched the first U.S. astronaut on suborbital spaceflight. Alcohols fell into general disuse as more energy-dense rocket fuels were developed, although ethanol was used in recent experimental Light aircraft, lightweight Mark-III X-racer, rocket-powered racing aircraft.
Commercial fuel cells operate on reformed natural gas, hydrogen or methanol. Ethanol is an attractive alternative due to its wide availability, low cost, high purity and low toxicity. There is a wide range of fuel cell concepts that have entered trials including direct-ethanol fuel cells, auto-thermal reforming systems and thermally integrated systems. The majority of work is being conducted at a research level although there are a number of organizations at the beginning of the commercialization of ethanol fuel cells.
Ethanol fireplaces can be used for home heating or for decoration. Ethanol can also be used as stove fuel for cooking.
Australian law limits the use of pure ethanol from sugarcane waste to 10% in automobiles. Older cars (and vintage cars designed to use a slower burning fuel) should have the engine valves upgraded or replaced.
According to an industry advocacy group, ethanol as a fuel reduces harmful tailpipe emissions of carbon monoxide, particulate matter, oxides of nitrogen, and other ozone-forming pollutants. Argonne National Laboratory analyzed greenhouse gas emissions of many different engine and fuel combinations, and found that biodiesel/petrodiesel blend (B20 (biodiesel), B20) showed a reduction of 8%, conventional E85 ethanol blend a reduction of 17% and cellulosic ethanol 64%, compared with pure gasoline. Ethanol has a much greater research octane number (RON) than gasoline, meaning it is less prone to pre-ignition, allowing for better ignition advance which means more torque, and efficiency in addition to the lower carbon emissions.
Ethanol combustion in an internal combustion engine yields many of the products of incomplete combustion produced by gasoline and significantly larger amounts of formaldehyde and related species such as acetaldehyde. This leads to a significantly larger photochemical reactivity and more ground level ozone. This data has been assembled into The Clean Fuels Report comparison of fuel emissions and show that ethanol exhaust generates 2.14 times as much ozone as gasoline exhaust. When this is added into the custom ''Localized Pollution Index'' of The Clean Fuels Report, the local pollution of ethanol (pollution that contributes to smog) is rated 1.7, where gasoline is 1.0 and higher numbers signify greater pollution. The California Air Resources Board formalized this issue in 2008 by recognizing control standards for formaldehydes as an emissions control group, much like the conventional NOx and reactive organic gases (ROGs).
More than 20% of Brazilian cars are able to use 100% ethanol as fuel, which includes ethanol-only engines and flex-fuel engines. Flex-fuel engines in Brazil are able to work with all ethanol, all gasoline or any mixture of both. In the United States, flex-fuel vehicles can run on 0% to 85% ethanol (15% gasoline) since higher ethanol blends are not yet allowed or efficient. Brazil supports this fleet of ethanol-burning automobiles with large national infrastructure that produces ethanol from domestically grown sugarcane.
Ethanol's high miscibility with water makes it unsuitable for shipping through modern Pipeline transport, pipelines like liquid hydrocarbons. Mechanics have seen increased cases of damage to small engines (in particular, the carburetor) and attribute the damage to the increased water retention by ethanol in fuel.
Ethanol was commonly used as fuel in early bipropellant rocket (liquid-propelled) vehicles, in conjunction with an oxidizer such as liquid oxygen. The German A-4 ballistic rocket of World War II (better known by its propaganda name ), which is credited as having begun the space age, used ethanol as the main constituent of . Under such nomenclature, the ethanol was mixed with 25% water to reduce the combustion chamber temperature. The design team helped develop U.S. rockets following World War II, including the ethanol-fueled Redstone (rocket family), Redstone rocket, which launched the first U.S. astronaut on suborbital spaceflight. Alcohols fell into general disuse as more energy-dense rocket fuels were developed, although ethanol was used in recent experimental Light aircraft, lightweight Mark-III X-racer, rocket-powered racing aircraft.
Commercial fuel cells operate on reformed natural gas, hydrogen or methanol. Ethanol is an attractive alternative due to its wide availability, low cost, high purity and low toxicity. There is a wide range of fuel cell concepts that have entered trials including direct-ethanol fuel cells, auto-thermal reforming systems and thermally integrated systems. The majority of work is being conducted at a research level although there are a number of organizations at the beginning of the commercialization of ethanol fuel cells.
Ethanol fireplaces can be used for home heating or for decoration. Ethanol can also be used as stove fuel for cooking.
Other uses
Ethanol is an important industrial ingredient. It has widespread use as a precursor for other organic compounds such as ethyl halides, ethyl esters, diethyl ether, acetic acid, and ethyl amines. It is considered a universalsolvent
A solvent (from the Latin language, Latin ''wikt:solvo#Latin, solvō'', "loosen, untie, solve") is a substance that dissolves a solute, resulting in a Solution (chemistry), solution. A solvent is usually a liquid but can also be a solid, a gas ...
, as its molecular structure allows for the dissolving of both Chemical polarity#Polar molecules, polar, hydrophilic and nonpolar, hydrophobic compounds. As ethanol also has a low boiling point, it is easy to remove from a solution that has been used to dissolve other compounds, making it a popular extracting agent for botanical oils. Cannabis oil extraction methods often use ethanol as an extraction solvent, and also as a post-processing solvent to remove oils, waxes, and chlorophyll from solution in a process known as Winterization of oil, winterization.
Ethanol is found in paints, tinctures, markers, personal care products such as mouthwashes, perfumes and deodorants, and Zoological specimens#Wet specimens, wet specimen preservatives. Polysaccharides Ethanol precipitation, precipitate from aqueous solution in the presence of alcohol, and ethanol precipitation is used for this reason in the purification of DNA and RNA. Because of its low freezing point of and low toxicity, ethanol is sometimes used in laboratories (with dry ice or other coolants) as a cooling bath to keep vessels at temperatures below the freezing point of water. For the same reason, it is also used as the active fluid in alcohol thermometers.
Chemistry
Ethanol is a 2-carbonalcohol
Alcohol may refer to:
Common uses
* Alcohol (chemistry), a class of compounds
* Ethanol, one of several alcohols, commonly known as alcohol in everyday life
** Alcohol (drug), intoxicant found in alcoholic beverages
** Alcoholic beverage, an alco ...
. Its molecular formula is . The structure of the molecule of ethanol is (an ethyl group
In organic chemistry, an ethyl group (abbr. Et) is an alkyl substituent with the formula , derived from ethane (). ''Ethyl'' is used in the International Union of Pure and Applied Chemistry
The International Union of Pure and Applied ...
linked to a hydroxyl group), which indicates that the carbon of a methyl group () is attached to the carbon of a methylene group (), which is attached to the oxygen of a hydroxyl group (). It is a constitutional isomer of dimethyl ether. Ethanol is sometimes abbreviated as EtOH, using the common organic chemistry notation of representing the ethyl group () with Et.
Physical properties
 Ethanol is a volatile, colorless liquid that has a slight odor. It burns with a smokeless blue flame that is not always visible in normal light. The physical properties of ethanol stem primarily from the presence of its hydroxyl group and the shortness of its carbon chain. Ethanol's hydroxyl group is able to participate in hydrogen bonding, rendering it more viscous and less volatile than less polar organic compounds of similar molecular weight, such as propane. Ethanol's adiabatic flame temperature for combustion in air is 2082 °C or 3779 °F.
Ethanol is slightly more refractive than water, having a refractive index of 1.36242 (at λ=589.3 nm and ). The triple point for ethanol is .
Ethanol is a volatile, colorless liquid that has a slight odor. It burns with a smokeless blue flame that is not always visible in normal light. The physical properties of ethanol stem primarily from the presence of its hydroxyl group and the shortness of its carbon chain. Ethanol's hydroxyl group is able to participate in hydrogen bonding, rendering it more viscous and less volatile than less polar organic compounds of similar molecular weight, such as propane. Ethanol's adiabatic flame temperature for combustion in air is 2082 °C or 3779 °F.
Ethanol is slightly more refractive than water, having a refractive index of 1.36242 (at λ=589.3 nm and ). The triple point for ethanol is .
Solvent properties
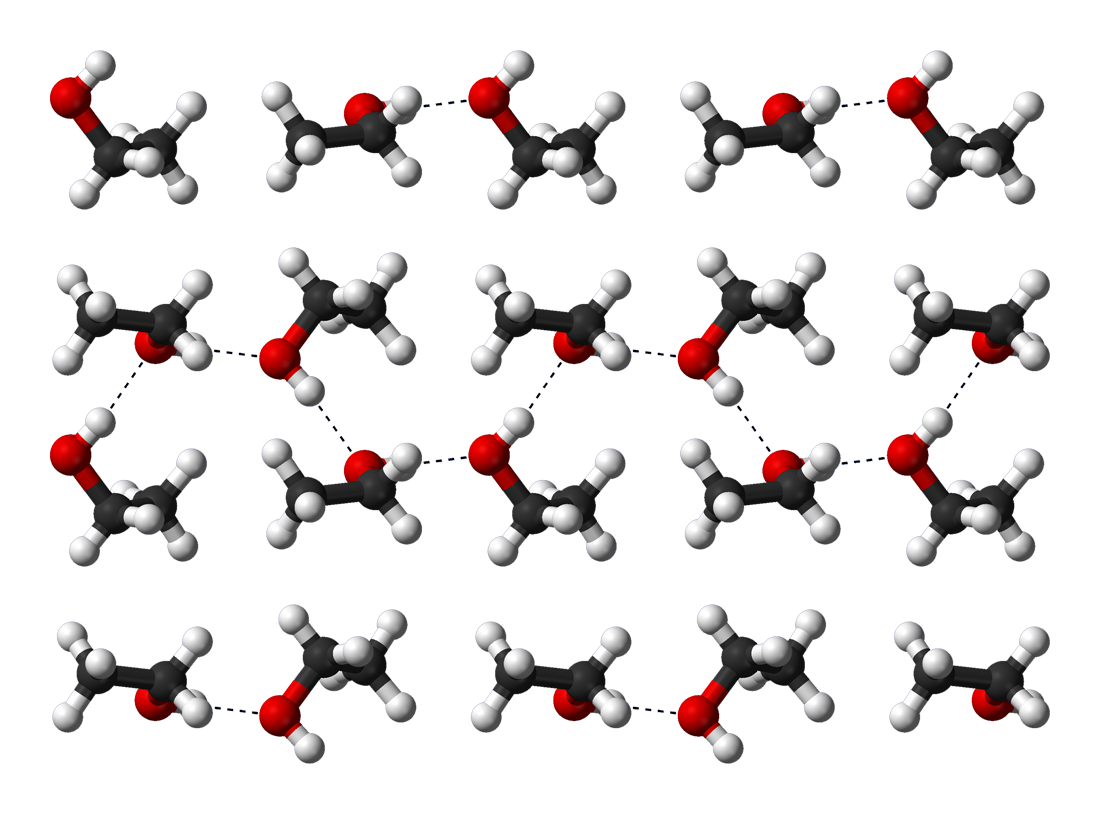
Ethanol is a versatile solvent, miscible with water and with many organic solvents, including acetic acid, acetone, benzene, carbon tetrachloride, chloroform, diethyl ether, ethylene glycol, glycerol, nitromethane, pyridine, and toluene. Its main use as a solvent is in making tincture of iodine, cough syrups, etc. It is also miscible with light aliphatic hydrocarbons, such as pentane and hexane, and with aliphatic chlorides such as 1,1,1-Trichloroethane, trichloroethane and tetrachloroethylene. Ethanol's miscibility with water contrasts with the immiscibility of longer-chain alcohols (five or more carbon atoms), whose water miscibility decreases sharply as the number of carbons increases. The miscibility of ethanol with alkanes is limited to alkanes up to undecane: mixtures with dodecane and higher alkanes show a miscibility gap below a certain temperature (about 13 °C for dodecane). The miscibility gap tends to get wider with higher alkanes, and the temperature for complete miscibility increases. Ethanol-water mixtures have less volume than the sum of their individual components at the given fractions. Mixing equal volumes of ethanol and water results in only 1.92 volumes of mixture. Mixing ethanol and water is exothermic, with up to 777 J/mol being released at 298 K. Hydrogen bonding causes pure ethanol to be hygroscopic to the extent that it readily absorbs water from the air. The polar nature of the hydroxyl group causes ethanol to dissolve many ionic compounds, notably sodium hydroxide, sodium and potassium hydroxides, magnesium chloride, calcium chloride, ammonium chloride, ammonium bromide, and sodium bromide. Sodium chloride, Sodium and potassium chlorides are slightly soluble in ethanol. Because the ethanol molecule also has a nonpolar end, it will also dissolve nonpolar substances, including most essential oils''Merck Index of Chemicals and Drugs'', 9th ed.; monographs 6575 through 6669 and numerous flavoring, coloring, and medicinal agents.
The addition of even a few percent of ethanol to water sharply reduces the surface tension of water. This property partially explains the "tears of wine" phenomenon. When wine is swirled in a glass, ethanol evaporates quickly from the thin film of wine on the wall of the glass. As the wine's ethanol content decreases, its surface tension increases and the thin film "beads up" and runs down the glass in channels rather than as a smooth sheet.
Hydrogen bonding causes pure ethanol to be hygroscopic to the extent that it readily absorbs water from the air. The polar nature of the hydroxyl group causes ethanol to dissolve many ionic compounds, notably sodium hydroxide, sodium and potassium hydroxides, magnesium chloride, calcium chloride, ammonium chloride, ammonium bromide, and sodium bromide. Sodium chloride, Sodium and potassium chlorides are slightly soluble in ethanol. Because the ethanol molecule also has a nonpolar end, it will also dissolve nonpolar substances, including most essential oils''Merck Index of Chemicals and Drugs'', 9th ed.; monographs 6575 through 6669 and numerous flavoring, coloring, and medicinal agents.
The addition of even a few percent of ethanol to water sharply reduces the surface tension of water. This property partially explains the "tears of wine" phenomenon. When wine is swirled in a glass, ethanol evaporates quickly from the thin film of wine on the wall of the glass. As the wine's ethanol content decreases, its surface tension increases and the thin film "beads up" and runs down the glass in channels rather than as a smooth sheet.
Azeotrope with water
At atmospheric pressure, mixtures of ethanol and water form an azeotrope at about 89.4 mol% ethanol (95.6% ethanol by mass, 97% alcohol by volume), with a boiling point of 351.3 K (78.1 °C). At lower pressure, the composition of the ethanol-water azeotrope shifts to more ethanol-rich mixtures. The minimum-pressure azeotrope has an ethanol fraction of 100% and a boiling point of 306 K (33 °C), corresponding to a pressure of roughly 70 torr (9.333 kPa). Below this pressure, there is no azeotrope, and it is possible to distill absolute ethanol from an ethanol-water mixture.Flammability
An ethanol–water solution will catch fire if heated above a temperature called its flash point and an ignition source is then applied to it. For 20% alcohol by mass (about 25% by volume), this will occur at about . The flash point of pure ethanol is , but may be influenced very slightly by atmospheric composition such as pressure and humidity. Ethanol mixtures can ignite below average room temperature. Ethanol is considered a flammable liquid (Class 3 Hazardous Material) in concentrations above 2.35% by mass (3.0% by volume; 6 Alcohol proof, proof). Dishes using burning alcohol for culinary effects are called flambé.Natural occurrence
Ethanol is a byproduct of the metabolic process of yeast. As such, ethanol will be present in any yeast habitat. Ethanol can commonly be found in overripe fruit. Ethanol produced by symbiotic yeast can be found in palm wine, bertam palm blossoms. Although some animal species, such as the pentailed treeshrew, exhibit ethanol-seeking behaviors, most show no interest or avoidance of food sources containing ethanol. Ethanol is also produced during the germination of many plants as a result of natural anaerobiosis. Ethanol has been detected in outer space, forming an icy coating around dust grains in interstellar clouds. Minute quantity amounts (average 196 parts per billion, ppb) of endogenous ethanol and acetaldehyde were found in the exhaled breath of healthy volunteers. Auto-brewery syndrome, also known as gut fermentation syndrome, is a rare medical condition in which intoxicating quantities of ethanol are produced through endogenousfermentation
Fermentation is a type of anaerobic metabolism which harnesses the redox potential of the reactants to make adenosine triphosphate (ATP) and organic end products. Organic molecules, such as glucose or other sugars, are catabolized and reduce ...
within the digestive system.
Production
 Ethanol is produced both as a
Ethanol is produced both as a petrochemical
Petrochemicals (sometimes abbreviated as petchems) are the chemical products obtained from petroleum by refining. Some chemical compounds made from petroleum are also obtained from other fossil fuels, such as coal or natural gas, or renewable s ...
, through the hydration of ethylene
Ethylene (IUPAC name: ethene) is a hydrocarbon which has the formula or . It is a colourless, flammable gas with a faint "sweet and musky" odour when pure. It is the simplest alkene (a hydrocarbon with carbon–carbon bond, carbon–carbon doub ...
and, via biological processes, by fermenting sugar
Sugar is the generic name for sweet-tasting, soluble carbohydrates, many of which are used in food. Simple sugars, also called monosaccharides, include glucose
Glucose is a sugar with the Chemical formula#Molecular formula, molecul ...
s with yeast
Yeasts are eukaryotic, single-celled microorganisms classified as members of the fungus kingdom (biology), kingdom. The first yeast originated hundreds of millions of years ago, and at least 1,500 species are currently recognized. They are est ...
. Which process is more economical depends on prevailing prices of petroleum and grain feed stocks.
Sources
World production of ethanol in 2006 was , with 69% of the world supply coming from Brazil and the U.S. Brazilian ethanol is produced from sugarcane, which has relatively high yields (830% more fuel than the fossil fuels used to produce it) compared to some other energy crops. Sugarcane not only has a greater concentration of sucrose than corn (by about 30%), but is also much easier to extract. The bagasse generated by the process is not discarded, but burned by power plants to produce electricity. Bagasse burning accounts for around 9% of the electricity produced in Brazil. In the 1970s most industrial ethanol in the U.S. was made as a petrochemical, but in the 1980s the U.S. introduced subsidies for corn-based ethanol. According to the Renewable Fuels Association, as of 30 October 2007, 131 grain ethanol bio-refineries in the U.S. have the capacity to produce of ethanol per year. An additional 72 construction projects underway (in the U.S.) can add of new capacity in the next 18 months. In India ethanol is made from sugarcane. Sweet sorghum is another potential source of ethanol, and is suitable for growing in dryland conditions. The International Crops Research Institute for the Semi-Arid Tropics is investigating the possibility of growing sorghum as a source of fuel, food, and animal feed in arid parts of Asia and Africa. Sweet sorghum has one-third the water requirement of sugarcane over the same time period. It also requires about 22% less water than corn. The world's first sweet sorghum ethanol distillery began commercial production in 2007 in Andhra Pradesh, India. Ethanol has been produced in the laboratory by converting carbon dioxide via biological and electrochemical reactions.Hydration
Ethanol can be produced from petrochemical feed stocks, primarily by the acid-catalysis, catalyzed Hydration reaction, hydration of ethylene. It is often referred to as synthetic ethanol. : The catalyst is most commonly phosphoric acid, adsorption, adsorbed onto a porous support such as silica gel or diatomaceous earth. This catalyst was first used for large-scale ethanol production by the Shell Oil Company in 1947. The reaction is carried out in the presence of high pressure steam at where a 5:3 ethylene to steam ratio is maintained. This process was used on an industrial scale by Union Carbide Corporation and others. It is no longer practiced in the US as fermentation ethanol produced from corn is more economical. In an older process, first practiced on the industrial scale in 1930 by Union Carbide Republished as but now almost entirely obsolete, ethylene was hydrated indirectly by reacting it with concentrated sulfuric acid to produce ethyl sulfate, which was hydrolyzed to yield ethanol and regenerate the sulfuric acid: : :Fermentation
Ethanol inalcoholic beverage
Drinks containing alcohol (drug), alcohol are typically divided into three classes—beers, wines, and Distilled beverage, spirits—with alcohol content typically between 3% and 50%. Drinks with less than 0.5% are sometimes considered Non-al ...
s and fuel is produced by fermentation. Certain species of yeast (e.g., ''Saccharomyces cerevisiae'') metabolize sugar (namely polysaccharides), producing ethanol and carbon dioxide. The chemical equations below summarize the conversion:
Fermentation is the process of culturing yeast under favorable thermal conditions to produce alcohol. This process is carried out at around . Toxicity of ethanol to yeast limits the ethanol concentration obtainable by brewing; higher concentrations, therefore, are obtained by Fortified wine, fortification or distillation
Distillation, also classical distillation, is the process of separating the component substances of a liquid mixture of two or more chemically discrete substances; the separation process is realized by way of the selective boiling of the mixt ...
. The most ethanol-tolerant yeast strains can survive up to approximately 18% ethanol by volume.
To produce ethanol from starchy materials such as cereals, the starch must first be converted into sugars. In brewing beer, this has traditionally been accomplished by allowing the grain to germinate, or malt, which produces the enzyme amylase. When the malted grain is mashing, mashed, the amylase converts the remaining starches into sugars.
Sugars for ethanol fermentation can be obtained from cellulose. Deployment of this technology could turn a number of cellulose-containing agricultural by-products, such as corncobs, straw, and sawdust, into renewable energy resources. Other agricultural residues such as sugarcane bagasse and energy crops such as switchgrass may also be fermentable sugar sources.
Testing
Purification
Ethylene hydration or brewing produces an ethanol–water mixture. For most industrial and fuel uses, the ethanol must be purified. Fractional distillation at atmospheric pressure can concentrate ethanol to 95.6% by weight (89.5 mole%). This mixture is an azeotrope with a boiling point of , and ''cannot'' be further purified by distillation. Addition of an entraining agent, such as benzene, cyclohexane, or heptane, allows a new ternary azeotrope comprising the ethanol, water, and the entraining agent to be formed. This lower-boiling ternary azeotrope is removed preferentially, leading to water-free ethanol. Apart from distillation, ethanol may be dried by addition of a desiccant, such as molecular sieves, cellulose, or cornmeal. The desiccants can be dried and reused. Molecular sieves can be used to selectively absorb the water from the 95.6% ethanol solution. Molecular sieves of pore-size 3 Angstrom, Å, a type of zeolite, effectively sequester water molecules while excluding ethanol molecules. Heating the wet sieves drives out the water, allowing regeneration of their desiccant capability. Membranes can also be used to separate ethanol and water. Membrane-based separations are not subject to the limitations of the water-ethanol azeotrope because the separations are not based on vapor-liquid equilibria. Membranes are often used in the so-called hybrid membrane distillation process. This process uses a pre-concentration distillation column as the first separating step. The further separation is then accomplished with a membrane operated either in vapor permeation or pervaporation mode. Vapor permeation uses a vapor membrane feed and pervaporation uses a liquid membrane feed. A variety of other techniques have been discussed, including the following: * Salting using potassium carbonate to exploit its insolubility will cause a phase separation with ethanol and water. This offers a very small potassium carbonate impurity to the alcohol that can be removed by distillation. This method is very useful in purification of ethanol by distillation, as ethanol forms an azeotrope with water. * Direct electrochemical reduction of carbon dioxide to ethanol under ambient conditions using copper nanoparticles on a carbon nanospike film as the catalyst; * Extraction of ethanol from grain mash by supercritical carbon dioxide; * Pervaporation; * Fractional freezing is also used to concentrate fermented alcoholic solutions, such as traditionally made Applejack (beverage); * Pressure swing adsorption.Grades of ethanol
Pure ethanol and alcoholic beverages are heavily Sin tax, taxed as psychoactive drugs, but ethanol has many uses that do not involve its consumption. To relieve the tax burden on these uses, most jurisdictions waive the tax when an agent has been added to the ethanol to render it unfit to drink. These include bitterant, bittering agents such as denatonium benzoate and toxins such as methanol, naphtha, and pyridine. Products of this kind are called ''denatured alcohol.'' Absolute or anhydrous alcohol refers to ethanol with a low water content. There are various grades with maximum water contents ranging from 1% to a few parts per million (ppm). If azeotropic distillation is used to remove water, it will contain trace amounts of the material separation agent (e.g. benzene). Absolute alcohol is not intended for human consumption. Absolute ethanol is used as a solvent for laboratory and industrial applications, where water will react with other chemicals, and as fuel alcohol. Spectroscopic ethanol is an absolute ethanol with a low absorbance in ultraviolet and visible light, fit for use as a solvent in ultraviolet-visible spectroscopy. Pure ethanol is classed as 200 proof (alcohol), proof in the US, equivalent to 175 degrees proof in the UK system. Rectified spirit, an azeotropic composition of 96% ethanol containing 4% water, is used instead of anhydrous ethanol for various purposes. Spirits of wine are about 94% ethanol (188 proof). The impurities are different from those in 95% (190 proof) laboratory ethanol.Reactions
Ethanol is classified as a primary alcohol, meaning that the carbon that its hydroxyl group attaches to has at least two hydrogen atoms attached to it as well. Many ethanol reactions occur at its hydroxyl group.Ester formation
In the presence of acid catalysts, ethanol reacts with carboxylic acids to produce ethyl esters and water: :carboxylic acid, RCOOH + HOCH2CH3 → ester, RCOOCH2CH3 + H2O This reaction, which is conducted on large scale industrially, requires the removal of the water from the reaction mixture as it is formed. Esters react in the presence of an acid or base to give back the alcohol and a salt. This reaction is known as saponification because it is used in the preparation of soap. Ethanol can also form esters with inorganic acids. Diethyl sulfate and triethyl phosphate are prepared by treating ethanol with sulfur trioxide and phosphorus pentoxide respectively. Diethyl sulfate is a useful ethylating agent in organic synthesis. Ethyl nitrite, prepared from the reaction of ethanol with sodium nitrite and sulfuric acid, was formerly used as a diuretic.Dehydration
In the presence of acid catalysts, alcohols can be converted to alkenes such as ethanol to ethylene. Typically solid acids such as alumina are used. :CH3CH2OH → H2C=CH2 + H2O Since water is removed from the same molecule, the reaction is known as Dehydration of alcohols to alkenes, intramolecular dehydration. Intramolecular dehydration of an alcohol requires a high temperature and the presence of an acid catalyst such as sulfuric acid. Ethylene produced from sugar-derived ethanol (primarily in Brazil) competes with ethylene produced from petrochemical feedstocks such as naphtha and ethane. At a lower temperature than that of intramolecular dehydration, Dehydration reaction, intermolecular alcohol dehydration may occur producing a symmetrical ether. This is a condensation reaction. In the following example, diethyl ether is produced from ethanol: :2 CH3CH2OH → CH3CH2OCH2CH3 + H2OCombustion
Complete combustion of ethanol forms carbon dioxide and water: :C2H5OH (l) + 3 O2 (g) → 2 CO2 (g) + 3 H2O (l); −Δc''H'' = 1371 kJ/mol = 29.8 kJ/g = 327 kcal/mol = 7.1 kcal/g :C2H5OH (l) + 3 O2 (g) → 2 CO2 (g) + 3 H2O (g); −Δc''H'' = 1236 kJ/mol = 26.8 kJ/g = 295.4 kcal/mol = 6.41 kcal/g Specific heat = 2.44 kJ/(kg·K)Acid-base chemistry
Ethanol is a neutral molecule and the pH of a solution of ethanol in water is nearly 7.00. Ethanol can be quantitatively converted to its conjugate base, the Alkoxide, ethoxide ion (CH3CH2O−), by reaction with an alkali metal such as sodium: :2 CH3CH2OH + 2 Na → 2 CH3CH2ONa + H2 or a very strong base such as sodium hydride: :CH3CH2OH + NaH → CH3CH2ONa + H2 The acidities of water and ethanol are nearly the same, as indicated by their Acid dissociation constant, pKa of 15.7 and 16 respectively. Thus, sodium ethoxide and sodium hydroxide exist in an equilibrium that is closely balanced: :CH3CH2OH + NaOH CH3CH2ONa + H2OHalogenation
Ethanol is not used industrially as a precursor to ethyl halides, but the reactions are illustrative. Ethanol reacts with hydrogen halides to produce haloalkane, ethyl halides such as ethyl chloride and ethyl bromide via an SN2 reaction, SN2 reaction: :CH3CH2OH + hydrogen chloride, HCl → CH3CH2Cl + H2O HCl requires a catalyst such as zinc chloride. HBr requires refluxing with a sulfuric acid catalyst. Ethyl halides can, in principle, also be produced by treating ethanol with more specialized Halogenation, halogenating agents, such as thionyl chloride or phosphorus tribromide. :CH3CH2OH + SOCl2 → CH3CH2Cl + SO2 + HCl Upon treatment with halogens in the presence of base, ethanol gives the corresponding haloform (CHX3, where X = Cl, Br, I). This conversion is called the haloform reaction. An intermediate in the reaction with chlorine is the aldehyde called chloral, which forms chloral hydrate upon reaction with water: :4 Cl2 + CH3CH2OH → CCl3CHO + 5 HCl :CCl3CHO + H2O → CCl3C(OH)2HOxidation
Ethanol can be oxidized to acetaldehyde and further oxidized to acetic acid, depending on the reagents and conditions. This oxidation is of no importance industrially, but in the human body, these oxidation reactions are catalyzed by the enzyme liver alcohol dehydrogenase, liver ADH. The oxidation product of ethanol, acetic acid, is a nutrient for humans, being a precursor to acetyl CoA, where the acetyl group can be spent as energy or used for biosynthesis.Metabolism
Ethanol is similar to macronutrients such as proteins, fats, and carbohydrates in that it provides calories. When consumed and metabolized, it contributes 7 kilocalories per gram via ethanol metabolism.Safety
Ethanol is very flammable and should not be used around an open flame. Pure ethanol will irritate the skin and eyes. Nausea, vomiting, and intoxication are symptoms of ingestion. Long-term use by ingestion can result in serious liver damage. Atmospheric concentrations above one part per thousand are above the European Union occupational exposure limits.History
The fermentation of sugar into ethanol is one of the earliest biotechnology, biotechnologies employed by humans. Ethanol has historically been identified variously as spirit of wine or ardent spirits, and as aqua vitae (Latin for "water of life") or aqua vita. The intoxicating effects of its consumption have been known since ancient times. Ethanol has been used by humans since prehistory as the intoxicating ingredient of alcoholic beverages. Dried residue on 9,000-year-old pottery found in China suggests that Neolithic people consumed alcoholic beverages. The inflammable nature of the exhalations of wine was already known to ancient natural philosophers such as Aristotle (384–322 BCE), Theophrastus (–287 BCE), and Pliny the Elder (23/24–79 CE). However, this did not immediately lead to the isolation of ethanol, despite the development of more advanced distillation techniques in second- and third-century Roman Egypt. An important recognition, first found in one of the writings attributed to Jabir ibn Hayyan, Jābir ibn Ḥayyān (ninth century CE), was that by Salt-effect distillation, adding salt to boiling wine, which increases the wine's relative volatility, the flammability of the resulting vapors may be enhanced. The distillation of wine is attested in Arabic works attributed to Al-Kindi, al-Kindī (–873 CE) and to Al-Farabi, al-Fārābī (–950), and in the 28th book of Al-Zahrawi, al-Zahrāwī's (Latin: Abulcasis, 936–1013) ''Kitāb al-Taṣrīf'' (later translated into Latin as ''Liber servatoris''). In the twelfth century, recipes for the production of ''aqua ardens'' ("burning water", i.e., ethanol) by distilling wine with salt started to appear in a number of Latin works, and by the end of the thirteenth century it had become a widely known substance among Western European chemists. The works of Taddeo Alderotti (1223–1296) describe a method for concentrating ethanol involving repeated fractional distillation through a water-cooled still, by which an ethanol purity of 90% could be obtained. The medicinal properties of ethanol were studied by Arnald of Villanova (1240–1311 CE) and John of Rupescissa (–1366), the latter of whom regarded it as a life-preserving substance able to prevent all diseases (the ''aqua vitae'' or "water of life", also called by John the ''Aether (classical element), quintessence'' of wine). In China, archaeological evidence indicates that the true distillation of alcohol began during the Jin dynasty (1115–1234), Jin (1115–1234) or Southern Song dynasty, Southern Song (1127–1279) dynasties. A still has been found at an archaeological site in Qinglong, Hebei, dating to the 12th century. In India, the true distillation of alcohol was introduced from the Middle East, and was in wide use in the Delhi Sultanate by the 14th century. In 1796, German-Russian chemist Johann Tobias Lowitz obtained pure ethanol by mixing partially purified ethanol (the alcohol-water azeotrope) with an excess of anhydrous alkali and then distilling the mixture over low heat. French chemist Antoine Lavoisier described ethanol as a compound of carbon, hydrogen, and oxygen, and in 1807 Nicolas-Théodore de Saussure determined ethanol's chemical formula. Fifty years later, Archibald Scott Couper published the structural formula of ethanol, one of the first structural formulas determined. Ethanol was first prepared synthetically in 1825 by Michael Faraday. He found that sulfuric acid could absorb large volumes of coal gas. He gave the resulting solution to Henry Hennell, a British chemist, who found in 1826 that it contained "sulphovinic acid" (ethyl hydrogen sulfate). In 1828, Hennell and the French chemist Georges-Simon Serullas independently discovered that sulphovinic acid could be decomposed into ethanol. On page 368, Hennell produces ethanol from "sulfovinic acid" (Ethyl sulfate, ethyl hydrogen sulfate). Thus, in 1825 Faraday had unwittingly discovered that ethanol could be produced from ethylene (a component of coal gas) by Acid catalysis, acid-catalyzed hydration, a process similar to current industrial ethanol synthesis.In 1855, the French chemist Marcellin Berthelot confirmed Faraday's discovery by preparing ethanol from pure ethylene. (Note: The chemical formulas in Berthelot's paper are wrong because chemists at that time used the wrong atomic masses for the elements; e.g., carbon (6 instead of 12), oxygen (8 instead of 16), etc.) Ethanol was used as lamp fuel in the U.S. as early as 1840, but a tax levied on industrial alcohol during the American Civil War, Civil War made this use uneconomical. The tax was repealed in 1906. Use as an automotive fuel dates back to 1908, with the Ford Model T able to run on petrol (gasoline) or ethanol. It fuels some spirit lamps. Ethanol intended for industrial use is often produced from ethylene. Ethanol has widespread use as a solvent of substances intended for human contact or consumption, including scents, flavorings, colorings, and medicines. In chemistry, it is both a solvent and a feedstock for the synthesis of other products. It has a long history as a fuel for heat and light, and more recently as a fuel for internal combustion engines.See also
* Ethanol-induced non-lamellar phases in phospholipids * Methanol * 1-Propanol * 2-Propanol * Rubbing alcohol * tert-Butyl alcohol * Butanol fuel * Timeline of alcohol fuelReferences
Further reading
* * * * *External links
Alcohol (Ethanol)
at ''The Periodic Table of Videos'' (University of Nottingham)
International Labour Organization
ethanol safety information
* [http://webbook.nist.gov/cgi/cbook.cgi?Name=ethanol&Units=SI National Institute of Standards and Technology] chemical data on ethanol
Chicago Board of Trade
news and market data on ethanol futures * Calculation o
vapor pressure
of ethanol
Ethanol History
A look into the history of ethanol
Industrial ethanol production process flow diagram using ethylene and sulphuric acid
{{Authority control Ethanol, Alcohol solvents Alkanols Anatomical preservation Chemical hazards Commodity chemicals Disinfectants Hepatotoxins Household chemicals Human metabolites IARC Group 1 carcinogens Oxygenates Primary alcohols Rocket fuels Teratogens Alcohol chemistry