Polar Compound on:
[Wikipedia]
[Google]
[Amazon]
 In
In
 A polar molecule has a net dipole as a result of the opposing charges (i.e. having partial positive and partial negative charges) from polar bonds arranged asymmetrically. Water (H2O) is an example of a polar molecule since it has a slight positive charge on one side and a slight negative charge on the other. The dipoles do not cancel out, resulting in a net dipole. Due to the polar nature of the water molecule itself, other polar molecules are generally able to dissolve in water. The dipole moment of water depends on its state. In the gas phase the dipole moment is ≈ 1.86 debye (D), whereas liquid water (≈ 2.95 D) and ice (≈ 3.09 D) are higher due to differing hydrogen-bonded environments. Other examples include sugars (like
A polar molecule has a net dipole as a result of the opposing charges (i.e. having partial positive and partial negative charges) from polar bonds arranged asymmetrically. Water (H2O) is an example of a polar molecule since it has a slight positive charge on one side and a slight negative charge on the other. The dipoles do not cancel out, resulting in a net dipole. Due to the polar nature of the water molecule itself, other polar molecules are generally able to dissolve in water. The dipole moment of water depends on its state. In the gas phase the dipole moment is ≈ 1.86 debye (D), whereas liquid water (≈ 2.95 D) and ice (≈ 3.09 D) are higher due to differing hydrogen-bonded environments. Other examples include sugars (like  Ammonia, NH3, is a molecule whose three N−H bonds have only a slight polarity (toward the more electronegative nitrogen atom). The molecule has two lone electrons in an orbital that points towards the fourth apex of an approximately regular tetrahedron, as predicted by the VSEPR theory. This orbital is not participating in covalent bonding; it is electron-rich, which results in a powerful dipole across the whole ammonia molecule.
Ammonia, NH3, is a molecule whose three N−H bonds have only a slight polarity (toward the more electronegative nitrogen atom). The molecule has two lone electrons in an orbital that points towards the fourth apex of an approximately regular tetrahedron, as predicted by the VSEPR theory. This orbital is not participating in covalent bonding; it is electron-rich, which results in a powerful dipole across the whole ammonia molecule.
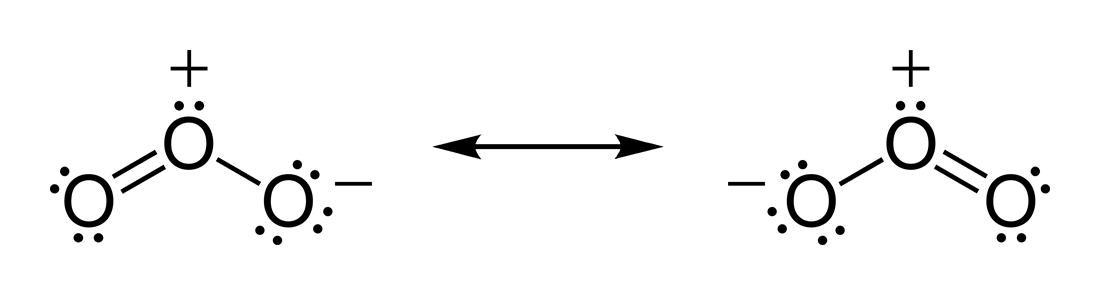 In ozone (O3) molecules, the two O−O bonds are nonpolar (there is no electronegativity difference between atoms of the same element). However, the distribution of other electrons is unevensince the central atom has to share electrons with two other atoms, but each of the outer atoms has to share electrons with only one other atom, the central atom is more deprived of electrons than the others (the central atom has a
In ozone (O3) molecules, the two O−O bonds are nonpolar (there is no electronegativity difference between atoms of the same element). However, the distribution of other electrons is unevensince the central atom has to share electrons with two other atoms, but each of the outer atoms has to share electrons with only one other atom, the central atom is more deprived of electrons than the others (the central atom has a

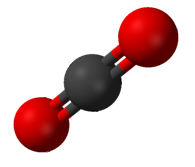 Carbon dioxide (CO2) has two polar C=O bonds, but the geometry of CO2 is linear so that the two bond dipole moments cancel and there is no net molecular dipole moment; the molecule is nonpolar.
Carbon dioxide (CO2) has two polar C=O bonds, but the geometry of CO2 is linear so that the two bond dipole moments cancel and there is no net molecular dipole moment; the molecule is nonpolar.
 Examples of household nonpolar compounds include fats, oil, and petrol/gasoline. Most nonpolar molecules are water-insoluble ( hydrophobic) at room temperature. Many nonpolar organic solvents, such as turpentine, are able to dissolve non-polar substances.
In the methane molecule (CH4) the four C−H bonds are arranged tetrahedrally around the carbon atom. Each bond has polarity (though not very strong). The bonds are arranged symmetrically so there is no overall dipole in the molecule. The diatomic oxygen molecule (O2) does not have polarity in the covalent bond because of equal electronegativity, hence there is no polarity in the molecule.
Examples of household nonpolar compounds include fats, oil, and petrol/gasoline. Most nonpolar molecules are water-insoluble ( hydrophobic) at room temperature. Many nonpolar organic solvents, such as turpentine, are able to dissolve non-polar substances.
In the methane molecule (CH4) the four C−H bonds are arranged tetrahedrally around the carbon atom. Each bond has polarity (though not very strong). The bonds are arranged symmetrically so there is no overall dipole in the molecule. The diatomic oxygen molecule (O2) does not have polarity in the covalent bond because of equal electronegativity, hence there is no polarity in the molecule.
File:CHCA cleavable detergent.png, This amphiphilic molecule has several polar groups ( hydrophilic, ''water-loving'') on the right side and a long nonpolar chain ( lipophilic, ''fat-loving'') at the left side. This gives it
Chemical Bonding
Molecule Polarity
{{chemical bonds Physical chemistry Chemical properties Dimensionless numbers of chemistry
 In
In chemistry
Chemistry is the science, scientific study of the properties and behavior of matter. It is a natural science that covers the Chemical element, elements that make up matter to the chemical compound, compounds made of atoms, molecules and ions ...
, polarity is a separation of electric charge leading to a molecule or its chemical groups having an electric dipole moment
The electric dipole moment is a measure of the separation of positive and negative electrical charges within a system, that is, a measure of the system's overall polarity. The SI unit for electric dipole moment is the coulomb-meter (C⋅m). The ...
, with a negatively charged end and a positively charged end.
Polar molecules must contain one or more polar bonds due to a difference in electronegativity between the bonded atoms. Molecules containing polar bonds have no molecular polarity if the bond dipoles cancel each other out by symmetry.
Polar molecules interact through dipole–dipole intermolecular forces and hydrogen bonds
In chemistry, a hydrogen bond (or H-bond) is a primarily electrostatic force of attraction between a hydrogen (H) atom which is covalently bound to a more electronegative "donor" atom or group (Dn), and another electronegative atom bearing a ...
. Polarity underlies a number of physical properties including surface tension
Surface tension is the tendency of liquid surfaces at rest to shrink into the minimum surface area possible. Surface tension is what allows objects with a higher density than water such as razor blades and insects (e.g. water striders) to f ...
, solubility, and melting and boiling points.
Polarity of bonds
Not all atoms attract electrons with the same force. The amount of "pull" an atom exerts on its electrons is called its electronegativity. Atoms with high electronegativitiessuch asfluorine
Fluorine is a chemical element with the symbol F and atomic number 9. It is the lightest halogen and exists at standard conditions as a highly toxic, pale yellow diatomic gas. As the most electronegative reactive element, it is extremely reacti ...
, oxygen, and nitrogenexert a greater pull on electrons than atoms with lower electronegativities such as alkali metal
The alkali metals consist of the chemical elements lithium (Li), sodium (Na), potassium (K),The symbols Na and K for sodium and potassium are derived from their Latin names, ''natrium'' and ''kalium''; these are still the origins of the names ...
s and alkaline earth metals. In a bond, this leads to unequal sharing of electrons between the atoms, as electrons will be drawn closer to the atom with the higher electronegativity.
Because electrons have a negative charge, the unequal sharing of electrons within a bond leads to the formation of an electric dipole: a separation of positive and negative electric charge. Because the amount of charge separated in such dipoles is usually smaller than a fundamental charge, they are called partial charges, denoted as δ+ ( delta plus) and δ− (delta minus). These symbols were introduced by Sir Christopher Ingold and Dr. Edith Hilda (Usherwood) Ingold in 1926. The bond dipole moment is calculated by multiplying the amount of charge separated and the distance between the charges.
These dipoles within molecules can interact with dipoles in other molecules, creating dipole-dipole intermolecular forces.
Classification
Bonds can fall between one of two extremescompletely nonpolar or completely polar. A completely nonpolar bond occurs when the electronegativities are identical and therefore possess a difference of zero. A completely polar bond is more correctly called an ionic bond, and occurs when the difference between electronegativities is large enough that one atom actually takes an electron from the other. The terms "polar" and "nonpolar" are usually applied tocovalent bond
A covalent bond is a chemical bond that involves the sharing of electrons to form electron pairs between atoms. These electron pairs are known as shared pairs or bonding pairs. The stable balance of attractive and repulsive forces between atoms ...
s, that is, bonds where the polarity is not complete. To determine the polarity of a covalent bond using numerical means, the difference between the electronegativity of the atoms is used.
Bond polarity is typically divided into three groups that are loosely based on the difference in electronegativity between the two bonded atoms. According to the Pauling scale
Electronegativity, symbolized as , is the tendency for an atom of a given chemical element to attract shared electrons (or electron density) when forming a chemical bond. An atom's electronegativity is affected by both its atomic number and ...
:
* ''Nonpolar bonds'' generally occur when the difference in electronegativity between the two atoms is less than 0.5
* ''Polar bonds'' generally occur when the difference in electronegativity between the two atoms is roughly between 0.5 and 2.0
* '' Ionic bonds'' generally occur when the difference in electronegativity between the two atoms is greater than 2.0
Pauling Pauling is a surname. People, places, and organizations with it include:
*Linus Pauling
**Paulingite
**Pauling's rules
**4674 Pauling
**Linus Pauling Institute
**Linus Pauling Library
**Linus Pauling Award
** Pauling Field
*Ava Helen Pauling, wife ...
based this classification scheme on the ''partial ionic character'' of a bond, which is an approximate function of the difference in electronegativity between the two bonded atoms. He estimated that a difference of 1.7 corresponds to 50% ionic character, so that a greater difference corresponds to a bond which is predominantly ionic.
As a quantum-mechanical
Quantum mechanics is a fundamental theory in physics that provides a description of the physical properties of nature at the scale of atoms and subatomic particles. It is the foundation of all quantum physics including quantum chemistry, qua ...
description, Pauling proposed that the wave function for a polar molecule AB is a linear combination of wave functions for covalent and ionic molecules: ψ = aψ(A:B) + bψ(A+B−). The amount of covalent and ionic character depends on the values of the squared coefficients a2 and b2.
Bond dipole moments
The bond dipole moment uses the idea ofelectric dipole moment
The electric dipole moment is a measure of the separation of positive and negative electrical charges within a system, that is, a measure of the system's overall polarity. The SI unit for electric dipole moment is the coulomb-meter (C⋅m). The ...
to measure the polarity of a chemical bond within a molecule. It occurs whenever there is a separation of positive and negative charges.
The bond dipole μ is given by:
:.
The bond dipole is modeled as δ+ — δ– with a distance ''d'' between the partial charges δ+ and δ–. It is a vector, parallel to the bond axis, pointing from minus to plus, as is conventional for electric dipole moment
The electric dipole moment is a measure of the separation of positive and negative electrical charges within a system, that is, a measure of the system's overall polarity. The SI unit for electric dipole moment is the coulomb-meter (C⋅m). The ...
vectors.
Chemists often draw the vector pointing from plus to minus. This vector can be physically interpreted as the movement undergone by electrons when the two atoms are placed a distance ''d'' apart and allowed to interact, the electrons will move from their free state positions to be localised more around the more electronegative atom.
The SI unit for electric dipole moment is the coulomb–meter. This is too large to be practical on the molecular scale.
Bond dipole moments are commonly measured in debyes, represented by the symbol D, which is obtained by measuring the charge in units of 10−10 statcoulomb and the distance ''d'' in Angstroms. Based on the conversion factor
Conversion of units is the conversion between different units of measurement for the same quantity, typically through multiplicative conversion factors which change the measured quantity value without changing its effects.
Overview
The process ...
of
10−10 statcoulomb being 0.208 units of elementary charge, so 1.0 debye results from an electron and a proton separated by 0.208 Å. A useful conversion factor is 1 D = 3.335 64 C m.
For diatomic molecules there is only one (single or multiple) bond so the bond dipole moment is the molecular dipole moment, with typical values in the range of 0 to 11 D. At one extreme, a symmetrical molecule such as chlorine, , has zero dipole moment, while near the other extreme, gas phase potassium bromide
Potassium bromide ( K Br) is a salt, widely used as an anticonvulsant and a sedative in the late 19th and early 20th centuries, with over-the-counter use extending to 1975 in the US. Its action is due to the bromide ion (sodium bromide is equall ...
, KBr, which is highly ionic, has a dipole moment of 10.41 D.
For polyatomic molecules, there is more than one bond. The total molecular dipole moment may be approximated as the vector sum of the individual bond dipole moments. Often bond dipoles are obtained by the reverse process: a known total dipole of a molecule can be decomposed into bond dipoles. This is done to transfer bond dipole moments to molecules that have the same bonds, but for which the total dipole moment is not yet known. The vector sum of the transferred bond dipoles gives an estimate for the total (unknown) dipole of the molecule.
Polarity of molecules
While the molecules can be described as "polar covalent", "nonpolar covalent", or "ionic", this is often a relative term, with one molecule simply being ''more polar'' or ''more nonpolar'' than another. However, the following properties are typical of such molecules. A molecule is composed of one or more chemical bonds between molecular orbitals of different atoms. A molecule may be polar either as a result of polar bonds due to differences in electronegativity as described above, or as a result of an asymmetric arrangement of nonpolar covalent bonds and non-bonding pairs of electrons known as a full molecular orbital.Polar molecules
 A polar molecule has a net dipole as a result of the opposing charges (i.e. having partial positive and partial negative charges) from polar bonds arranged asymmetrically. Water (H2O) is an example of a polar molecule since it has a slight positive charge on one side and a slight negative charge on the other. The dipoles do not cancel out, resulting in a net dipole. Due to the polar nature of the water molecule itself, other polar molecules are generally able to dissolve in water. The dipole moment of water depends on its state. In the gas phase the dipole moment is ≈ 1.86 debye (D), whereas liquid water (≈ 2.95 D) and ice (≈ 3.09 D) are higher due to differing hydrogen-bonded environments. Other examples include sugars (like
A polar molecule has a net dipole as a result of the opposing charges (i.e. having partial positive and partial negative charges) from polar bonds arranged asymmetrically. Water (H2O) is an example of a polar molecule since it has a slight positive charge on one side and a slight negative charge on the other. The dipoles do not cancel out, resulting in a net dipole. Due to the polar nature of the water molecule itself, other polar molecules are generally able to dissolve in water. The dipole moment of water depends on its state. In the gas phase the dipole moment is ≈ 1.86 debye (D), whereas liquid water (≈ 2.95 D) and ice (≈ 3.09 D) are higher due to differing hydrogen-bonded environments. Other examples include sugars (like sucrose
Sucrose, a disaccharide, is a sugar composed of glucose and fructose subunits. It is produced naturally in plants and is the main constituent of white sugar. It has the molecular formula .
For human consumption, sucrose is extracted and refined ...
), which have many polar oxygen–hydrogen (−OH) groups and are overall highly polar.
If the bond dipole moments of the molecule do not cancel, the molecule is polar. For example, the water molecule (H2O) contains two polar O−H bonds in a bent
Bent may refer to:
Places
* Bent, Iran, a city in Sistan and Baluchestan Province, Iran
* Bent District, an administrative subdivision of Iran
* Bent, Netherlands, a village in the municipality of Rijnwoude, the Netherlands
* Bent County, Colo ...
(nonlinear) geometry. The bond dipole moments do not cancel, so that the molecule forms a molecular dipole
In physics, a dipole () is an electromagnetic phenomenon which occurs in two ways:
*An electric dipole deals with the separation of the positive and negative electric charges found in any electromagnetic system. A simple example of this system i ...
with its negative pole at the oxygen and its positive pole midway between the two hydrogen atoms. In the figure each bond joins the central O atom with a negative charge (red) to an H atom with a positive charge (blue).
The hydrogen fluoride
Hydrogen fluoride (fluorane) is an inorganic compound with the chemical formula . This colorless gas or liquid is the principal industrial source of fluorine, often as an aqueous solution called hydrofluoric acid. It is an important feedstock i ...
, HF, molecule is polar by virtue of polar covalent bondsin the covalent bond electrons are displaced toward the more electronegative fluorine atom.
 Ammonia, NH3, is a molecule whose three N−H bonds have only a slight polarity (toward the more electronegative nitrogen atom). The molecule has two lone electrons in an orbital that points towards the fourth apex of an approximately regular tetrahedron, as predicted by the VSEPR theory. This orbital is not participating in covalent bonding; it is electron-rich, which results in a powerful dipole across the whole ammonia molecule.
Ammonia, NH3, is a molecule whose three N−H bonds have only a slight polarity (toward the more electronegative nitrogen atom). The molecule has two lone electrons in an orbital that points towards the fourth apex of an approximately regular tetrahedron, as predicted by the VSEPR theory. This orbital is not participating in covalent bonding; it is electron-rich, which results in a powerful dipole across the whole ammonia molecule.
 In ozone (O3) molecules, the two O−O bonds are nonpolar (there is no electronegativity difference between atoms of the same element). However, the distribution of other electrons is unevensince the central atom has to share electrons with two other atoms, but each of the outer atoms has to share electrons with only one other atom, the central atom is more deprived of electrons than the others (the central atom has a
In ozone (O3) molecules, the two O−O bonds are nonpolar (there is no electronegativity difference between atoms of the same element). However, the distribution of other electrons is unevensince the central atom has to share electrons with two other atoms, but each of the outer atoms has to share electrons with only one other atom, the central atom is more deprived of electrons than the others (the central atom has a formal charge
In chemistry, a formal charge (F.C. or q), in the covalent view of chemical bonding, is the charge assigned to an atom in a molecule, assuming that electrons in all chemical bonds are shared equally between atoms, regardless of relative electroneg ...
of +1, while the outer atoms each have a formal charge of −). Since the molecule has a bent geometry, the result is a dipole across the whole ozone molecule.
When comparing a polar and nonpolar molecule with similar molar masses, the polar molecule in general has a higher boiling point, because the dipole–dipole interaction between polar molecules results in stronger intermolecular attractions. One common form of polar interaction is the hydrogen bond
In chemistry, a hydrogen bond (or H-bond) is a primarily electrostatic force of attraction between a hydrogen (H) atom which is covalently bound to a more electronegative "donor" atom or group (Dn), and another electronegative atom bearing a ...
, which is also known as the H-bond. For example, water forms H-bonds and has a molar mass M = 18 and a boiling point of +100 °C, compared to nonpolar methane with M = 16 and a boiling point of –161 °C.
Nonpolar molecules
A molecule may be nonpolar either when there is an equal sharing of electrons between the two atoms of a diatomic molecule or because of the symmetrical arrangement of polar bonds in a more complex molecule. For example,boron trifluoride
Boron trifluoride is the inorganic compound with the formula BF3. This pungent, colourless, and toxic gas forms white fumes in moist air. It is a useful Lewis acid and a versatile building block for other boron compounds.
Structure and bondin ...
(BF3) has a trigonal planar arrangement of three polar bonds at 120°. This results in no overall dipole in the molecule.

 Carbon dioxide (CO2) has two polar C=O bonds, but the geometry of CO2 is linear so that the two bond dipole moments cancel and there is no net molecular dipole moment; the molecule is nonpolar.
Carbon dioxide (CO2) has two polar C=O bonds, but the geometry of CO2 is linear so that the two bond dipole moments cancel and there is no net molecular dipole moment; the molecule is nonpolar.
 Examples of household nonpolar compounds include fats, oil, and petrol/gasoline. Most nonpolar molecules are water-insoluble ( hydrophobic) at room temperature. Many nonpolar organic solvents, such as turpentine, are able to dissolve non-polar substances.
In the methane molecule (CH4) the four C−H bonds are arranged tetrahedrally around the carbon atom. Each bond has polarity (though not very strong). The bonds are arranged symmetrically so there is no overall dipole in the molecule. The diatomic oxygen molecule (O2) does not have polarity in the covalent bond because of equal electronegativity, hence there is no polarity in the molecule.
Examples of household nonpolar compounds include fats, oil, and petrol/gasoline. Most nonpolar molecules are water-insoluble ( hydrophobic) at room temperature. Many nonpolar organic solvents, such as turpentine, are able to dissolve non-polar substances.
In the methane molecule (CH4) the four C−H bonds are arranged tetrahedrally around the carbon atom. Each bond has polarity (though not very strong). The bonds are arranged symmetrically so there is no overall dipole in the molecule. The diatomic oxygen molecule (O2) does not have polarity in the covalent bond because of equal electronegativity, hence there is no polarity in the molecule.
Amphiphilic molecules
Large molecules that have one end with polar groups attached and another end with nonpolar groups are described asamphiphile
An amphiphile (from the Greek αμφις amphis, both, and φιλíα philia, love, friendship), or amphipath, is a chemical compound possessing both hydrophilic (''water-loving'', polar) and lipophilic (''fat-loving'') properties. Such a compoun ...
s or ''amphiphilic'' molecules. They are good surfactant
Surfactants are chemical compounds that decrease the surface tension between two liquids, between a gas and a liquid, or interfacial tension between a liquid and a solid. Surfactants may act as detergents, wetting agents, emulsifiers, foaming ...
s and can aid in the formation of stable emulsions, or blends, of water and fats. Surfactants reduce the interfacial tension between oil and water by adsorbing at the liquid–liquid interface.
surfactant
Surfactants are chemical compounds that decrease the surface tension between two liquids, between a gas and a liquid, or interfacial tension between a liquid and a solid. Surfactants may act as detergents, wetting agents, emulsifiers, foaming ...
properties
File:Micelle scheme-en.svg, A micellethe lipophilic ends of the surfactant molecules dissolve in the oil, while the hydrophilic charged ends remain outside in the water phase, shielding the rest of the hydrophobic micelle. In this way, the small oil droplet becomes water-soluble.
File:Phospholipid schematic representation.png, Phospholipid
Phospholipids, are a class of lipids whose molecule has a hydrophilic "head" containing a phosphate group and two hydrophobic "tails" derived from fatty acids, joined by an alcohol residue (usually a glycerol molecule). Marine phospholipids typ ...
s are effective natural surfactants that have important biological functions
File:Phospholipids aqueous solution structures.svg, Cross section view of the structures that can be formed by phospholipid
Phospholipids, are a class of lipids whose molecule has a hydrophilic "head" containing a phosphate group and two hydrophobic "tails" derived from fatty acids, joined by an alcohol residue (usually a glycerol molecule). Marine phospholipids typ ...
s. They can form a micelle and are vital in forming cell membranes
Predicting molecule polarity
Determining thepoint group
In geometry, a point group is a mathematical group of symmetry operations (isometries in a Euclidean space) that have a fixed point in common. The coordinate origin of the Euclidean space is conventionally taken to be a fixed point, and every p ...
is a useful way to predict polarity of a molecule. In general, a molecule will not possess dipole moment if the individual bond dipole moments of the molecule cancel each other out. This is because dipole moments are euclidean vector quantities with magnitude and direction, and a two equal vectors who oppose each other will cancel out.
Any molecule with a centre of inversion ("i") or a horizontal mirror plane ("σh") will not possess dipole moments.
Likewise, a molecule with more than one C''n'' axis of rotation will not possess a dipole moment because dipole moments cannot lie in more than one dimension. As a consequence of that constraint, all molecules with dihedral symmetry (D''n'') will not have a dipole moment because, by definition, D point groups have two or multiple C''n'' axes.
Since C1, Cs,C∞h C''n'' and C''n''v point groups do not have a centre of inversion, horizontal mirror planes or multiple C''n'' axis, molecules in one of those point groups will have dipole moment.
Electrical deflection of water
Contrary to popular misconception, the electrical deflection of a stream of water from a charged object is not based on polarity. The deflection occurs because of electrically charged droplets in the stream, which the charged object induces. A stream of water can also be deflected in a uniform electrical field, which cannot exert force on polar molecules. Additionally, after a stream of water is grounded, it can no longer be deflected. Weak deflection is even possible for nonpolar liquids.See also
* Chemical properties *Colloid
A colloid is a mixture in which one substance consisting of microscopically dispersed insoluble particles is suspended throughout another substance. Some definitions specify that the particles must be dispersed in a liquid, while others extend ...
* Detergent
A detergent is a surfactant or a mixture of surfactants with cleansing properties when in dilute solutions. There are a large variety of detergents, a common family being the alkylbenzene sulfonates, which are soap-like compounds that are more ...
* Electronegativities of the elements (data page)
* Polar point group
References
External links
Chemical Bonding
Molecule Polarity
{{chemical bonds Physical chemistry Chemical properties Dimensionless numbers of chemistry