|
Intermolecular Force
An intermolecular force (IMF; also secondary force) is the force that mediates interaction between molecules, including the electromagnetic forces of attraction or repulsion which act between atoms and other types of neighbouring particles (e.g. atoms or ions). Intermolecular forces are weak relative to intramolecular forces – the forces which hold a molecule together. For example, the covalent bond, involving sharing electron pairs between atoms, is much stronger than the forces present between neighboring molecules. Both sets of forces are essential parts of force fields frequently used in molecular mechanics. The first reference to the nature of microscopic forces is found in Alexis Clairaut's work ''Théorie de la figure de la Terre,'' published in Paris in 1743. Other scientists who have contributed to the investigation of microscopic forces include: Laplace, Gauss, Maxwell, Boltzmann and Pauling. Attractive intermolecular forces are categorized into the following ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
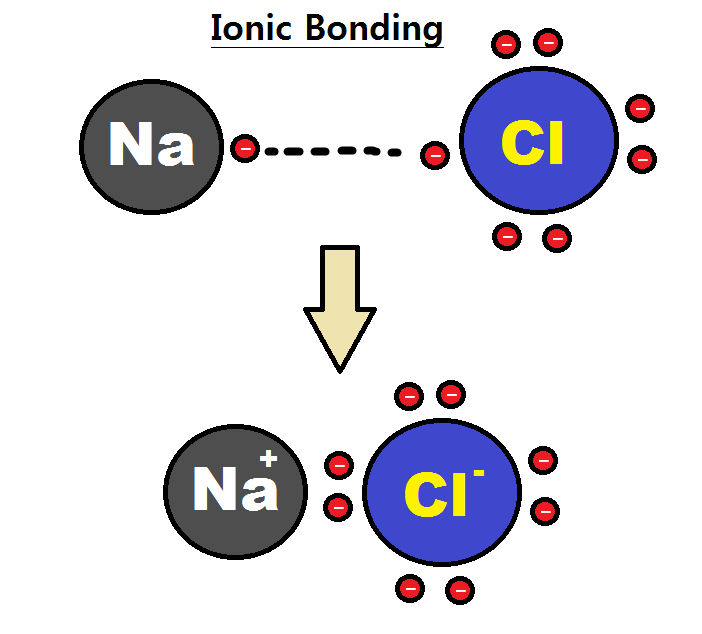
Intramolecular Force
An intramolecular force (from Latin ''intra-'' 'within') is any force that binds together the atoms making up a molecule. Intramolecular forces are stronger than the Intermolecular force, intermolecular forces that govern the interactions between molecules. Types The classical model identifies three main types of chemical bonds — ionic, covalent, and metallic — distinguished by the degree of charge separation between participating atoms. The characteristics of the bond formed can be predicted by the properties of constituent atoms, namely electronegativity. They differ in the magnitude of their Bond-dissociation energy, bond enthalpies, a measure of bond strength, and thus affect the physical and chemical properties of compounds in different ways. % of ionic character is directly proportional difference in electronegativity of bonded atom. Ionic bond An ionic bond can be approximated as complete transfer of one or more valence electrons of atoms participating in bond formati ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electromagnetism
In physics, electromagnetism is an interaction that occurs between particles with electric charge via electromagnetic fields. The electromagnetic force is one of the four fundamental forces of nature. It is the dominant force in the interactions of atoms and molecules. Electromagnetism can be thought of as a combination of electrostatics and magnetism, which are distinct but closely intertwined phenomena. Electromagnetic forces occur between any two charged particles. Electric forces cause an attraction between particles with opposite charges and repulsion between particles with the same charge, while magnetism is an interaction that occurs between charged particles in relative motion. These two forces are described in terms of electromagnetic fields. Macroscopic charged objects are described in terms of Coulomb's law for electricity and Ampère's force law for magnetism; the Lorentz force describes microscopic charged particles. The electromagnetic force is responsible for ma ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
London Dispersion Force
London dispersion forces (LDF, also known as dispersion forces, London forces, instantaneous dipole–induced dipole forces, fluctuating induced dipole bonds or loosely as van der Waals forces) are a type of intermolecular force acting between atoms and molecules that are normally electrically symmetric; that is, the electrons are symmetrically distributed with respect to the nucleus. They are part of the van der Waals forces. The LDF is named after the German physicist Fritz London. They are the weakest of the intermolecular forces. Introduction The electron distribution around an atom or molecule undergoes fluctuations in time. These fluctuations create instantaneous electric fields which are felt by other nearby atoms and molecules, which in turn adjust the spatial distribution of their own electrons. The net effect is that the fluctuations in electron positions in one atom induce a corresponding redistribution of electrons in other atoms, such that the electron motions be ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electronic Structure
Quantum chemistry, also called molecular quantum mechanics, is a branch of physical chemistry focused on the application of quantum mechanics to chemical systems, particularly towards the quantum-mechanical calculation of electronic contributions to physical and chemical properties of molecules, materials, and solutions at the atomic level. These calculations include systematically applied approximations intended to make calculations computationally feasible while still capturing as much information about important contributions to the computed wave functions as well as to observable properties such as structures, spectra, and thermodynamic properties. Quantum chemistry is also concerned with the computation of quantum effects on molecular dynamics and chemical kinetics. Chemists rely heavily on spectroscopy through which information regarding the quantization of energy on a molecular scale can be obtained. Common methods are infra-red (IR) spectroscopy, nuclear magnetic re ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Covalent Bond
A covalent bond is a chemical bond that involves the sharing of electrons to form electron pairs between atoms. These electron pairs are known as shared pairs or bonding pairs. The stable balance of attractive and repulsive forces between atoms, when they share electrons, is known as covalent bonding. For many molecules, the sharing of electrons allows each atom to attain the equivalent of a full valence shell, corresponding to a stable electronic configuration. In organic chemistry, covalent bonding is much more common than ionic bonding. Covalent bonding also includes many kinds of interactions, including σ-bonding, π-bonding, metal-to-metal bonding, agostic interactions, bent bonds, three-center two-electron bonds and three-center four-electron bonds. The term "covalence" was introduced by Irving Langmuir in 1919, with Nevil Sidgwick using "co-valent link" in the 1920s. Merriam-Webster dates the specific phrase ''covalent bond'' to 1939, recognizing its first known ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Molecular Ion
A molecule is a group of two or more atoms that are held together by attractive forces known as chemical bonds; depending on context, the term may or may not include ions that satisfy this criterion. In quantum physics, organic chemistry, and biochemistry, the distinction from ions is dropped and ''molecule'' is often used when referring to polyatomic ions. A molecule may be homonuclear, that is, it consists of atoms of one chemical element, e.g. two atoms in the oxygen molecule (O2); or it may be heteronuclear, a chemical compound composed of more than one element, e.g. water (two hydrogen atoms and one oxygen atom; H2O). In the kinetic theory of gases, the term ''molecule'' is often used for any gaseous particle regardless of its composition. This relaxes the requirement that a molecule contains two or more atoms, since the noble gases are individual atoms. Atoms and complexes connected by non-covalent interactions, such as hydrogen bonds or ionic bonds, are typicall ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
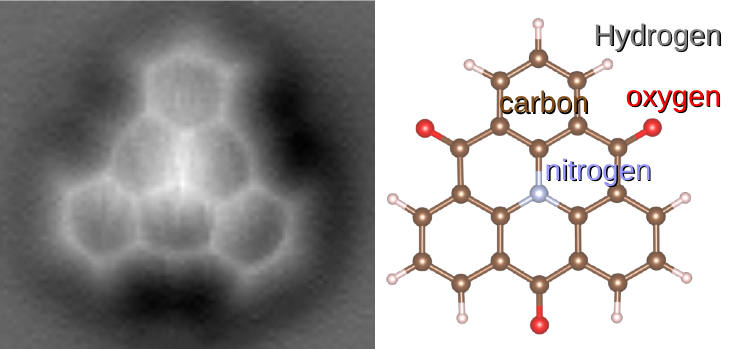
Molecule
A molecule is a group of two or more atoms that are held together by Force, attractive forces known as chemical bonds; depending on context, the term may or may not include ions that satisfy this criterion. In quantum physics, organic chemistry, and biochemistry, the distinction from ions is dropped and ''molecule'' is often used when referring to polyatomic ions. A molecule may be homonuclear, that is, it consists of atoms of one chemical element, e.g. two atoms in the oxygen molecule (O2); or it may be heteronuclear, a chemical compound composed of more than one element, e.g. water (molecule), water (two hydrogen atoms and one oxygen atom; H2O). In the kinetic theory of gases, the term ''molecule'' is often used for any gaseous particle regardless of its composition. This relaxes the requirement that a molecule contains two or more atoms, since the noble gases are individual atoms. Atoms and complexes connected by non-covalent interactions, such as hydrogen bonds or ionic ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
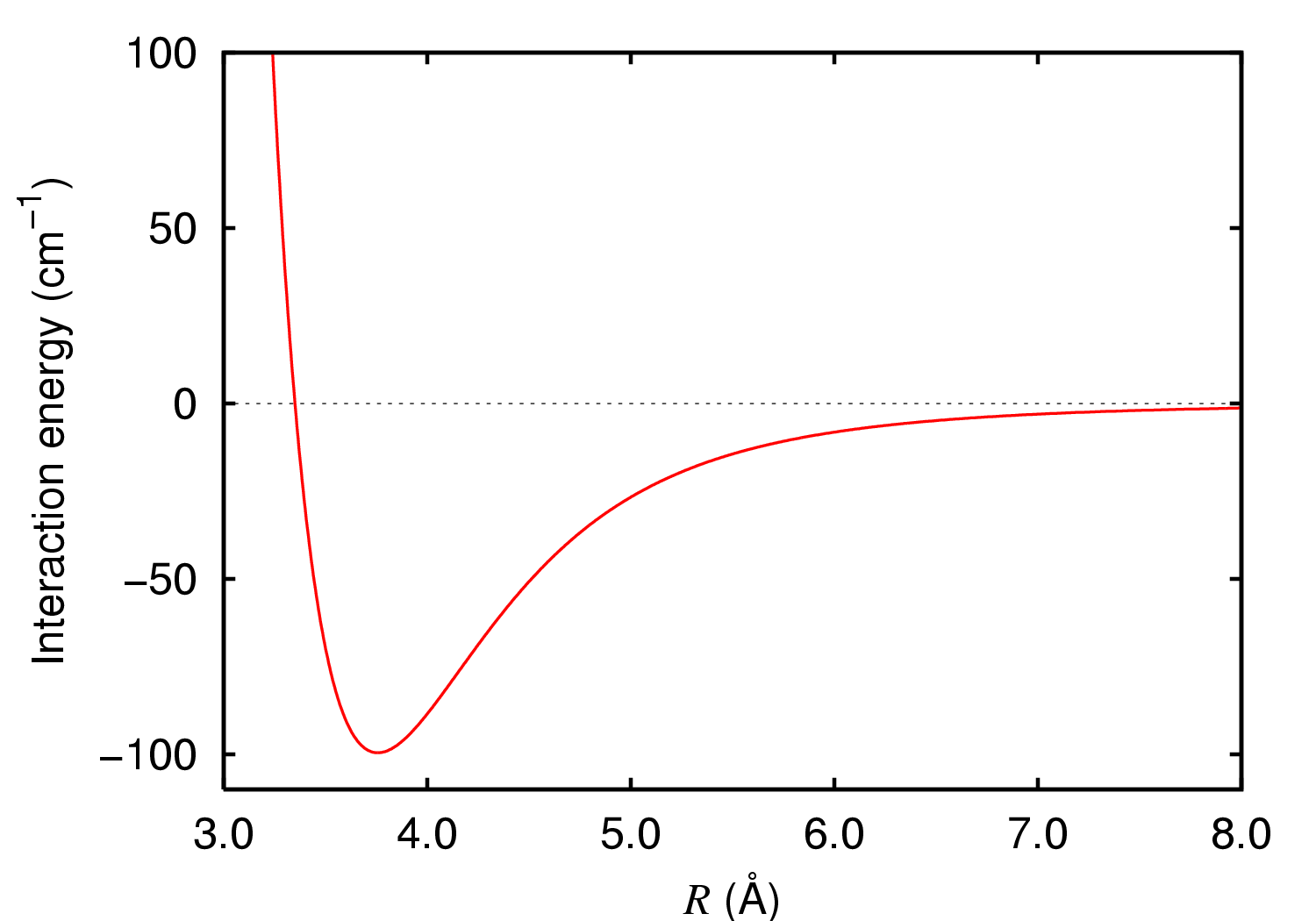
Lennard-Jones Potential
In computational chemistry, molecular physics, and physical chemistry, the Lennard-Jones potential (also termed the LJ potential or 12-6 potential; named for John Lennard-Jones) is an intermolecular pair potential. Out of all the intermolecular potentials, the Lennard-Jones potential is probably the one that has been the most extensively studied. It is considered an archetype model for simple yet realistic intermolecular interactions. The Lennard-Jones potential is often used as a building block in molecular models (a.k.a. force fields) for more complex substances. Many studies of the idealized "Lennard-Jones substance" use the potential to understand the physical nature of matter. Overview The Lennard-Jones potential is a simple model that still manages to describe the essential features of interactions between simple atoms and molecules: Two interacting particles repel each other at very close distance, attract each other at moderate distance, and eventually stop intera ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Buckingham Potential
In theoretical chemistry, the Buckingham potential is a formula proposed by Richard Buckingham which describes the Pauli exclusion principle and van der Waals energy \Phi_(r) for the interaction of two atoms that are not directly bonded as a function of the interatomic distance r. It is a variety of interatomic potentials. :\Phi_(r) = A \exp \left(-Br\right) - \frac Here, A, B and C are constants. The two terms on the right-hand side constitute a repulsion and an attraction, because their first derivatives with respect to r are negative and positive, respectively. Buckingham proposed this as a simplification of the Lennard-Jones potential, in a theoretical study of the equation of state for gaseous helium, neon and argon. As explained in Buckingham's original paper and, e.g., in section 2.2.5 of Jensen's text,F. Jensen, ''Introduction to Computational Chemistry'', 2nd ed., Wiley, 2007, the repulsion is due to the interpenetration of the closed electron shells. "There is there ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Mie Potential
The Mie potential is an interaction potential describing the interactions between particles on the atomic level. It is mostly used for describing intermolecular interactions, but at times also for modeling intramolecular interaction, i.e. bonds. The Mie potential is named after the German physicist Gustav Mie; yet the history of intermolecular potentials is more complicated. The Mie potential is the generalized case of the Lennard-Jones (LJ) potential, which is perhaps the most widely used pair potential. The Mie potential V(r) is a function of r, the distance between two particles, and is written as V(r) = C \, \varepsilon \left \left(\frac \right)^- \left( \frac\right)^m \right,~~~~~~ (1) with C = \frac \left( \frac\right)^ . The Lennard-Jones potential corresponds to the special case where n=12 and m=6 in Eq. (1). In Eq. (1), \varepsilon is the dispersion energy, and \sigma indicates the distance at which V = 0 , which is sometimes called the "collision radius." The p ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pair Potential
In physics, a pair potential is a function that describes the potential energy of two interacting objects solely as a function of the distance between them. Some interactions, like Coulomb's law in electrodynamics or Newton's law of universal gravitation in mechanics naturally have this form for simple spherical objects. For other types of more complex interactions or objects it is useful and common to approximate the interaction by a pair potential, for example interatomic potentials in physics and computational chemistry that use approximations like the Lennard-Jones and Morse potentials. Functional form The total energy of a system of N objects at positions \vec_i, that interact through pair potential v is given by E=\frac12\sum_^N\sum_^Nv\left(\left, \vec_i - \vec_j\\right)\ . Equivalently, this can be expressed as E=\sum_^N\sum_^Nv\left(\left, \vec_i - \vec_j\\right)\ . This expression uses the fact that interaction is symmetric between particles i and j. It also ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Virial Coefficient
Virial coefficients B_i appear as coefficients in the virial expansion of the pressure of a many-particle system in powers of the density, providing systematic corrections to the ideal gas law. They are characteristic of the interaction potential between the particles and in general depend on the temperature. The second virial coefficient B_2 depends only on the pair interaction between the particles, the third (B_3) depends on 2- and non-additive 3-body interactions, and so on. Derivation The first step in obtaining a closed expression for virial coefficients is a cluster expansion of the grand canonical partition function : \Xi = \sum_ = e^ Here p is the pressure, V is the volume of the vessel containing the particles, k_\text is the Boltzmann constant, T is the absolute temperature, \lambda =\exp mu/(k_\textT) is the fugacity, with \mu the chemical potential. The quantity Q_n is the canonical partition function of a subsystem of n particles: : Q_n = \operatorname e ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |