Nitrone on:
[Wikipedia]
[Google]
[Amazon]
 In
In
 However, a hydroxylamine with two α hydrogens may unsaturate on either side. Carbonyl condensation avoids this ambiguity...
However, a hydroxylamine with two α hydrogens may unsaturate on either side. Carbonyl condensation avoids this ambiguity... ...but is inhibited if both ketone substituents are bulky.
In principle, ''N''-
...but is inhibited if both ketone substituents are bulky.
In principle, ''N''-
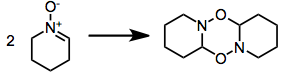 Syntheses with nitrone precursors obviate the issue with increased temperature, to exaggerate entropic factors; or with a nitrone excess.
Syntheses with nitrone precursors obviate the issue with increased temperature, to exaggerate entropic factors; or with a nitrone excess.
 Other ring-closing reactions are known, including formal +3and +2
Other ring-closing reactions are known, including formal +3and +2
 In
In organic chemistry
Organic chemistry is a subdiscipline within chemistry involving the science, scientific study of the structure, properties, and reactions of organic compounds and organic matter, organic materials, i.e., matter in its various forms that contain ...
, a nitrone is a functional group
In organic chemistry, a functional group is any substituent or moiety (chemistry), moiety in a molecule that causes the molecule's characteristic chemical reactions. The same functional group will undergo the same or similar chemical reactions r ...
consisting of an ''N''-oxide of an imine
In organic chemistry, an imine ( or ) is a functional group or organic compound containing a carbon–nitrogen double bond (). The nitrogen atom can be attached to a hydrogen or an organic group (R). The carbon atom has two additional single bon ...
. The general structure is , where R3 is not a hydrogen
Hydrogen is a chemical element; it has chemical symbol, symbol H and atomic number 1. It is the lightest and abundance of the chemical elements, most abundant chemical element in the universe, constituting about 75% of all baryon, normal matter ...
. Their primary application is intermediates in chemical synthesis
Chemical synthesis (chemical combination) is the artificial execution of chemical reactions to obtain one or several products. This occurs by physical and chemical manipulations usually involving one or more reactions. In modern laboratory uses ...
. A nitrone is a 1,3-dipole
In organic chemistry, a 1,3-dipolar compound or 1,3-dipole is a dipolar compound with delocalized electrons and a separation of charge over three atoms. They are reactants in 1,3-dipolar cycloadditions.
The dipole has at least one resonance st ...
used in cycloaddition
In organic chemistry, a cycloaddition is a chemical reaction in which "two or more Unsaturated hydrocarbon, unsaturated molecules (or parts of the same molecule) combine with the formation of a cyclic adduct in which there is a net reduction of th ...
s, and a carbonyl
In organic chemistry, a carbonyl group is a functional group with the formula , composed of a carbon atom double bond, double-bonded to an oxygen atom, and it is divalent at the C atom. It is common to several classes of organic compounds (such a ...
mimic.
Structure
Nitrones, as a tetrasubstituteddouble bond
In chemistry, a double bond is a covalent bond between two atoms involving four bonding electrons as opposed to two in a single bond. Double bonds occur most commonly between two carbon atoms, for example in alkenes. Many double bonds exist betw ...
, admit ''cis''–''trans'' isomerism.
Generation of nitrones
Typical nitrone sources arehydroxylamine
Hydroxylamine (also known as hydroxyammonia) is an inorganic compound with the chemical formula . The compound exists as hygroscopic colorless crystals.Greenwood and Earnshaw. ''Chemistry of the Elements.'' 2nd Edition. Reed Educational and Prof ...
oxidation or condensation
Condensation is the change of the state of matter from the gas phase into the liquid phase, and is the reverse of vaporization. The word most often refers to the water cycle. It can also be defined as the change in the state of water vapor ...
with carbonyl compounds
In organic chemistry, a carbonyl group is a functional group with the formula , composed of a carbon atom double-bonded to an oxygen atom, and it is divalent at the C atom. It is common to several classes of organic compounds (such as aldehydes ...
. Secondary hydroxylamines oxidize to nitrones in air over a timescale of several weeks, a process cupric salts accelerate. The most general reagent used for the oxidation of hydroxylamines is aqueous mercuric oxide
Mercury(II) oxide, also called mercuric oxide or simply mercury oxide, is the inorganic compound with the formula Hg O. It has a red or orange color. Mercury(II) oxide is a solid at room temperature and pressure. The mineral form montroydite is ...
:
 However, a hydroxylamine with two α hydrogens may unsaturate on either side. Carbonyl condensation avoids this ambiguity...
However, a hydroxylamine with two α hydrogens may unsaturate on either side. Carbonyl condensation avoids this ambiguity... ...but is inhibited if both ketone substituents are bulky.
In principle, ''N''-
...but is inhibited if both ketone substituents are bulky.
In principle, ''N''-alkylation Alkylation is a chemical reaction that entails transfer of an alkyl group. The alkyl group may be transferred as an alkyl carbocation, a free radical, a carbanion, or a carbene (or their equivalents). Alkylating agents are reagents for effecting al ...
could produce nitrones from oximes
In organic chemistry, an oxime is an organic compound belonging to the imines, with the general formula , where R is an organic side-chain and R' may be hydrogen, forming an aldoxime, or another organic group, forming a ketoxime. O-substituted ...
, but in practice electrophiles
In chemistry, an electrophile is a chemical species that forms bonds with nucleophiles by accepting an electron pair. Because electrophiles accept electrons, they are Lewis acids. Most electrophiles are positively charged, have an atom that carries ...
typically perform a mixture of ''N''- and ''O''-attack.
Reactions
Some nitrones oligomerize: Syntheses with nitrone precursors obviate the issue with increased temperature, to exaggerate entropic factors; or with a nitrone excess.
Syntheses with nitrone precursors obviate the issue with increased temperature, to exaggerate entropic factors; or with a nitrone excess.
Carbonyl mimic
Like many other unsaturated functional groups, nitrones activate the α and β carbons towards reaction. The α carbon is an electrophile and the β carbon a nucleophile; that is, nitrones polarize like carbonyls and nitriles but unlikenitro compounds
In organic chemistry, nitro compounds are organic compounds that contain one or more nitro functional groups (). The nitro group is one of the most common explosophores (functional group that makes a compound explosive) used globally. The nitr ...
and vinyl sulfur derivatives.
Nitrones hydrolyze extremely easily to the corresponding carbonyl and N-hydroxylamine.
1,3-dipolar cycloadditions
As 1,3dipoles, nitrones perform +2cycloadditions. For example, a dipolarophilicalkene
In organic chemistry, an alkene, or olefin, is a hydrocarbon containing a carbon–carbon double bond. The double bond may be internal or at the terminal position. Terminal alkenes are also known as Alpha-olefin, α-olefins.
The Internationa ...
combines to form isoxazolidine
Isoxazolidine is the organic compound with the formula (CH2)3(NH)O. It is the parent of a family of compounds called Isoxazolidines, which are saturated C3NO heterocyclic rings where the nitrogen and oxygen occupy adjacent positions (1 and 2). T ...
:
 Other ring-closing reactions are known, including formal +3and +2
Other ring-closing reactions are known, including formal +3and +2cycloaddition
In organic chemistry, a cycloaddition is a chemical reaction in which "two or more Unsaturated hydrocarbon, unsaturated molecules (or parts of the same molecule) combine with the formation of a cyclic adduct in which there is a net reduction of th ...
s.
Isomerization
Deoxygenating reagents,light
Light, visible light, or visible radiation is electromagnetic radiation that can be visual perception, perceived by the human eye. Visible light spans the visible spectrum and is usually defined as having wavelengths in the range of 400– ...
, or heat all catalyze rearrangement to the amide
In organic chemistry, an amide, also known as an organic amide or a carboxamide, is a chemical compound, compound with the general formula , where R, R', and R″ represent any group, typically organyl functional group, groups or hydrogen at ...
. Acids catalyze rearrangement to the oxime ether.
Reduction
Hydrides
In chemistry, a hydride is formally the anion of hydrogen (H−), a hydrogen ion with two electrons. In modern usage, this is typically only used for ionic bonds, but it is sometimes (and has been more frequently in the past) applied to all co ...
add to give hydroxylamines
Hydroxylamine (also known as hydroxyammonia) is an inorganic compound with the chemical formula . The compound exists as hygroscopic colorless crystals.Greenwood and Earnshaw. ''Chemistry of the Elements.'' 2nd Edition. Reed Educational and Prof ...
. Reducing Lewis acids
A Lewis acid (named for the American physical chemist Gilbert N. Lewis) is a chemical species that contains an empty orbital which is capable of accepting an electron pair from a Lewis base to form a Lewis adduct. A Lewis base, then, is any sp ...
(e.g. metals
A metal () is a material that, when polished or fractured, shows a lustrous appearance, and conducts electricity and heat relatively well. These properties are all associated with having electrons available at the Fermi level, as against no ...
, ) deoxygenate to the imine
In organic chemistry, an imine ( or ) is a functional group or organic compound containing a carbon–nitrogen double bond (). The nitrogen atom can be attached to a hydrogen or an organic group (R). The carbon atom has two additional single bon ...
instead.
See also
* ''N''-Oxoammonium salt *Nitronate
A nitronate (IUPAC: azinate) in organic chemistry is an anion with the general structure , containing the functional group, where R can be hydrogen, halogen, organyl group or other groups. It is the anion of nitronic acid (sometimes also called ...
References
{{reflist Functional groups