Fmoc Cleavage on:
[Wikipedia]
[Google]
[Amazon]
 The fluorenylmethoxycarbonyl protecting group (Fmoc) is a base-labile
The fluorenylmethoxycarbonyl protecting group (Fmoc) is a base-labile 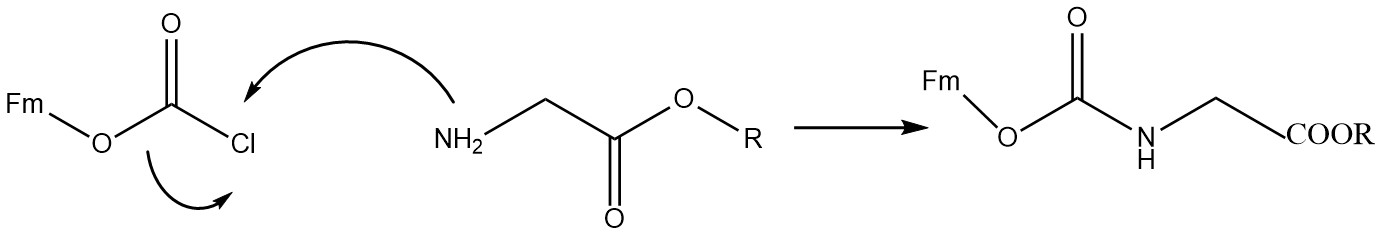
 The other common method for introducing the Fmoc group is through 9-fluorenylmethylsuccinimidyl carbonate (Fmoc-OSu), which may itself be obtained by the reaction of Fmoc-Cl with the dicyclohexylammonium salt of ''N''-hydroxysuccinimide.
Reacting with 9-fluorenylmethyloxycarbonyl azide (itself made by reacting Fmoc-Cl with
The other common method for introducing the Fmoc group is through 9-fluorenylmethylsuccinimidyl carbonate (Fmoc-OSu), which may itself be obtained by the reaction of Fmoc-Cl with the dicyclohexylammonium salt of ''N''-hydroxysuccinimide.
Reacting with 9-fluorenylmethyloxycarbonyl azide (itself made by reacting Fmoc-Cl with
 The fluorenylmethoxycarbonyl protecting group (Fmoc) is a base-labile
The fluorenylmethoxycarbonyl protecting group (Fmoc) is a base-labile amine
In chemistry, amines (, ) are organic compounds that contain carbon-nitrogen bonds. Amines are formed when one or more hydrogen atoms in ammonia are replaced by alkyl or aryl groups. The nitrogen atom in an amine possesses a lone pair of elec ...
protecting group
A protecting group or protective group is introduced into a molecule by chemical modification of a functional group to obtain chemoselectivity in a subsequent chemical reaction. It plays an important role in multistep organic synthesis.
In man ...
used in organic synthesis
Organic synthesis is a branch of chemical synthesis concerned with the construction of organic compounds. Organic compounds are molecules consisting of combinations of covalently-linked hydrogen, carbon, oxygen, and nitrogen atoms. Within the gen ...
, particularly in peptide synthesis
In organic chemistry, peptide synthesis is the production of peptides, compounds where multiple amino acids are linked via amide bonds, also known as peptide bonds. Peptides are chemically synthesized by the condensation reaction of the carboxyl ...
. It is popular for its stability toward acids
An acid is a molecule or ion capable of either donating a proton (i.e. hydrogen cation, H+), known as a Brønsted–Lowry acid, or forming a covalent bond with an electron pair, known as a Lewis acid.
The first category of acids are the ...
and hydrolysis
Hydrolysis (; ) is any chemical reaction in which a molecule of water breaks one or more chemical bonds. The term is used broadly for substitution reaction, substitution, elimination reaction, elimination, and solvation reactions in which water ...
and its selective removal by weak bases, such as piperidine
Piperidine is an organic compound with the molecular formula (CH2)5NH. This heterocyclic amine consists of a six-membered ring containing five methylene bridges (–CH2–) and one amine bridge (–NH–). It is a colorless liquid with an odor de ...
, without affecting most other protecting groups or sensitive functional group
In organic chemistry, a functional group is any substituent or moiety (chemistry), moiety in a molecule that causes the molecule's characteristic chemical reactions. The same functional group will undergo the same or similar chemical reactions r ...
s. Fmoc protection is especially advantageous in solid-phase peptide synthesis
In organic chemistry, peptide synthesis is the production of peptides, compounds where multiple amino acids are linked via amide bonds, also known as peptide bonds. Peptides are chemically synthesized by the condensation reaction of the carboxyl ...
(SPPS), where its compatibility with other reagents and ease of removal streamline synthesis workflows. Upon deprotection, Fmoc yields a byproduct
A by-product or byproduct is a secondary product derived from a production process, manufacturing process or chemical reaction; it is not the primary product or service being produced.
A by-product can be useful and marketable or it can be cons ...
(Dibenzofulvene
9-Methylene-fluorene or dibenzofulvene (DBF) is a polycyclic aromatic hydrocarbon with chemical formula . It is best known as one product from deprotection of the Fmoc group. It can be prepared by treatment of 9-hydroxymethylfluorene with strong ...
) that can be monitored by UV spectroscopy, allowing for efficient reaction tracking.

Protection & Formation
Fmoc-carbamate
In organic chemistry, a carbamate is a category of organic compounds with the general Chemical formula, formula and Chemical structure, structure , which are formally Derivative (chemistry), derived from carbamic acid (). The term includes orga ...
is frequently used as a protecting group
A protecting group or protective group is introduced into a molecule by chemical modification of a functional group to obtain chemoselectivity in a subsequent chemical reaction. It plays an important role in multistep organic synthesis.
In man ...
for primary and secondary amine
In chemistry, amines (, ) are organic compounds that contain carbon-nitrogen bonds. Amines are formed when one or more hydrogen atoms in ammonia are replaced by alkyl or aryl groups. The nitrogen atom in an amine possesses a lone pair of elec ...
s, where the Fmoc group can be introduced by reacting the amine with fluorenylmethyloxycarbonyl chloride
Fluorenylmethyloxycarbonyl chloride (Fmoc-Cl) is a chloroformate ester. It is used to introduce the fluorenylmethyloxycarbonyl protecting group as the Fmoc carbamate
In organic chemistry, a carbamate is a category of organic compounds with th ...
(Fmoc-Cl), e.g.:
: The other common method for introducing the Fmoc group is through 9-fluorenylmethylsuccinimidyl carbonate (Fmoc-OSu), which may itself be obtained by the reaction of Fmoc-Cl with the dicyclohexylammonium salt of ''N''-hydroxysuccinimide.
Reacting with 9-fluorenylmethyloxycarbonyl azide (itself made by reacting Fmoc-Cl with
The other common method for introducing the Fmoc group is through 9-fluorenylmethylsuccinimidyl carbonate (Fmoc-OSu), which may itself be obtained by the reaction of Fmoc-Cl with the dicyclohexylammonium salt of ''N''-hydroxysuccinimide.
Reacting with 9-fluorenylmethyloxycarbonyl azide (itself made by reacting Fmoc-Cl with sodium azide
Sodium azide is an inorganic compound with the formula . This colorless salt is the gas-forming component in some car airbag systems. It is used for the preparation of other azide compounds. It is highly soluble in water and is acutely poisonou ...
) in sodium bicarbonate
Sodium bicarbonate ( IUPAC name: sodium hydrogencarbonate), commonly known as baking soda or bicarbonate of soda (or simply “bicarb” especially in the UK) is a chemical compound with the formula NaHCO3. It is a salt composed of a sodium cat ...
and aqueous dioxane
Dioxane may refer to the following chemical compounds:
* 1,2-dioxane
* 1,3-dioxane
* 1,4-dioxane
{{Authority control ...
is also a method to install Fmoc group.
Because the fluorenyl group is highly fluorescent
Fluorescence is one of two kinds of photoluminescence, the emission of light by a substance that has absorbed light or other electromagnetic radiation. When exposed to ultraviolet radiation, many substances will glow (fluoresce) with color ...
, certain UV-inactive compounds may be reacted to give the Fmoc derivatives, suitable for analysis by reversed phase HPLC. Analytical uses of Fmoc-Cl that do not use chromatography may be limited by the requirement that excess Fmoc-Cl be removed before an analysis of fluorescence.
Cleavage & Deprotection
The Fmoc group is rapidly removed by base. Piperidine is usually preferred for Fmoc group removal as it forms a stable adduct with the dibenzofulvene byproduct, preventing it from reacting with the substrate.Role in Peptide Synthesis
The use of Fmoc as a temporary protecting group for amine at theN-terminus
The N-terminus (also known as the amino-terminus, NH2-terminus, N-terminal end or amine-terminus) is the start of a protein or polypeptide, referring to the free amine group (-NH2) located at the end of a polypeptide. Within a peptide, the amin ...
in solid phase synthesis is very widespread for Fmoc/tBu approach, because its removal with piperidine does not disturb the acid-labile linker between the peptide and the resin. A typical SPPS Fmoc deprotection is performed with a solution of 20% piperidine
Piperidine is an organic compound with the molecular formula (CH2)5NH. This heterocyclic amine consists of a six-membered ring containing five methylene bridges (–CH2–) and one amine bridge (–NH–). It is a colorless liquid with an odor de ...
in N,N-dimethylformamide
Dimethylformamide, DMF is an organic compound with the chemical formula . Its structure is . Commonly abbreviated as DMF (although this initialism is sometimes used for dimethylfuran, or dimethyl fumarate), this colourless liquid is miscible with ...
(DMF).
:
:
:
:
:
Common deprotection cocktails for Fmoc during SPPS
* 20% piperidine in DMF (Fmoc group has an approximate half life of 6 seconds in this solution) * 5%piperazine
Piperazine () is an organic compound with the formula . In term of its structure, it can be described as cyclohexane with the 1- and 4-CH2 groups replaced by NH. Piperazine exists as deliquescent solid with a saline taste. Piperazine is freely sol ...
, 1% DBU DBU may refer to:
Universities
* Dallas Baptist University, Dallas, Texas, U.S.
* Desh Bhagat University, Mandi Gobindgarh, Punjab, India
* Duluth Business University, Duluth, Minnesota, U.S.
Other uses
* 1,8-Diazabicyclo .4.0ndec-7-ene, an org ...
and 1% formic acid
Formic acid (), systematically named methanoic acid, is the simplest carboxylic acid. It has the chemical formula HCOOH and structure . This acid is an important intermediate in chemical synthesis and occurs naturally, most notably in some an ...
in DMF. This method avoids the use of strictly controlled piperidine. No side product was observed for a peptide with 9 residues synthesized with this method.
References
External links
*{{Commons category-inline Biochemistry methods Protecting groups