charge-transfer complex on:
[Wikipedia]
[Google]
[Amazon]
 In
In

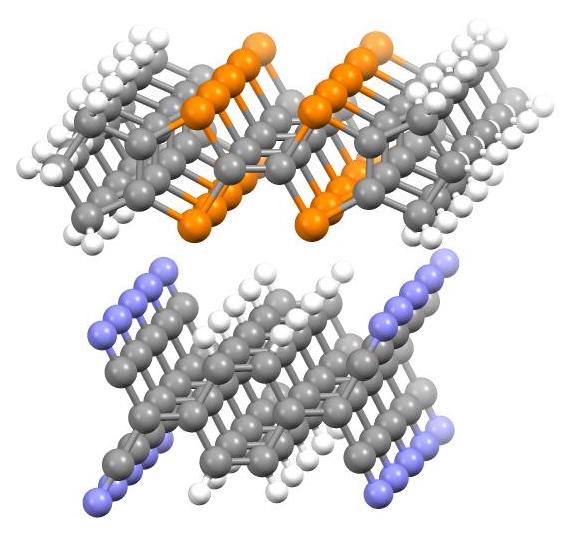 In 1954, charge-transfer salts derived from perylene with
In 1954, charge-transfer salts derived from perylene with
 In
In chemistry
Chemistry is the scientific study of the properties and behavior of matter. It is a physical science within the natural sciences that studies the chemical elements that make up matter and chemical compound, compounds made of atoms, molecules a ...
, charge-transfer (CT) complex, or electron donor-acceptor complex, describes a type of supramolecular assembly of two or more molecules or ions. The assembly consists of two molecule
A molecule is a group of two or more atoms that are held together by Force, attractive forces known as chemical bonds; depending on context, the term may or may not include ions that satisfy this criterion. In quantum physics, organic chemi ...
s that self-attract through electrostatic forces, i.e., one has at least partial negative charge and the partner has partial positive charge, referred to respectively as the electron acceptor
An electron acceptor is a chemical entity that accepts electrons transferred to it from another compound. Electron acceptors are oxidizing agents.
The electron accepting power of an electron acceptor is measured by its redox potential.
In the ...
and electron donor. In some cases, the degree of charge transfer is "complete", such that the CT complex can be classified as a salt. In other cases, the charge-transfer association is weak, and the interaction can be disrupted easily by polar solvents.
Examples
Electron donor-acceptor complexes
A number of organic compounds form charge-transfer complex, which are often described as electron-donor-acceptor complexes (EDA complexes). Typical acceptors are nitrobenzenes or tetracyanoethylene (TCNE). The strength of their interaction with electron donors correlates with the ionization potentials of the components. For TCNE, the stability constants (L/mol) for its complexes with benzene derivatives correlates with the number of methyl groups:benzene
Benzene is an Organic compound, organic chemical compound with the Chemical formula#Molecular formula, molecular formula C6H6. The benzene molecule is composed of six carbon atoms joined in a planar hexagonal Ring (chemistry), ring with one hyd ...
(0.128), 1,3,5-trimethylbenzene (1.11), 1,2,4,5-tetramethylbenzene (3.4), and hexamethylbenzene (16.8). A simple example for a prototypical electron-donor-acceptor complexes is nitroaniline.
1,3,5-Trinitrobenzene and related polynitrated aromatic compounds, being electron-deficient, form charge-transfer complexes with many arenes. Such complexes form upon crystallization, but often dissociate in solution to the components. Characteristically, these CT salts crystallize in stacks of alternating donor and acceptor (nitro aromatic) molecules, i.e. A-B-A-B.
Dihalogen/interhalogen CT complexes
Early studies on donor-acceptor complexes focused on the solvatochromism exhibited by iodine, which often results from I2 forming adducts with electron donors such as amines andether
In organic chemistry, ethers are a class of compounds that contain an ether group, a single oxygen atom bonded to two separate carbon atoms, each part of an organyl group (e.g., alkyl or aryl). They have the general formula , where R and R� ...
s. Dihalogens X2 (X = Cl, Br, I) and interhalogens XY(X = I; Y = Cl, Br) are Lewis acid species capable of forming a variety of products when reacted with donor species. Among these species (including oxidation or protonated products), CT adducts D·XY have been largely investigated. The CT interaction has been quantified and is the basis of many schemes for parameterizing donor and acceptor properties, such as those devised by Gutmann, Childs, Beckett, and the ECW model.
Many organic species featuring chalcogen or pnictogen donor atoms form CT salts. The nature of the resulting adducts can be investigated both in solution and in the solid state.
In solution, the intensity of charge-transfer bands in the UV-Vis absorbance spectrum is strongly dependent upon the degree (equilibrium constant) of this association reaction. Methods have been developed to determine the equilibrium constant for these complexes in solution by measuring the intensity of absorption bands as a function of the concentration of donor and acceptor components in solution. The Benesi-Hildebrand method, named for its developers, was first described for the association of iodine dissolved in aromatic hydrocarbons.
In the solid state a valuable parameter is the elongation of the X–X or X–Y bond length, resulting from the antibonding nature of the σ* LUMO. The elongation can be evaluated by means of structural determinations (XRD) and FT-Raman spectroscopy.
A well-known example is the complex formed by iodine
Iodine is a chemical element; it has symbol I and atomic number 53. The heaviest of the stable halogens, it exists at standard conditions as a semi-lustrous, non-metallic solid that melts to form a deep violet liquid at , and boils to a vi ...
when combined with starch
Starch or amylum is a polymeric carbohydrate consisting of numerous glucose units joined by glycosidic bonds. This polysaccharide is produced by most green plants for energy storage. Worldwide, it is the most common carbohydrate in human diet ...
, which exhibits an intense purple charge-transfer band. This has widespread use as a rough screen for counterfeit currency. Unlike most paper, the paper used in US currency is not sized with starch. Thus, formation of this purple color on application of an iodine solution indicates a counterfeit.
TTF-TCNQ: prototype for electrically conducting complexes

 In 1954, charge-transfer salts derived from perylene with
In 1954, charge-transfer salts derived from perylene with iodine
Iodine is a chemical element; it has symbol I and atomic number 53. The heaviest of the stable halogens, it exists at standard conditions as a semi-lustrous, non-metallic solid that melts to form a deep violet liquid at , and boils to a vi ...
or bromine
Bromine is a chemical element; it has chemical symbol, symbol Br and atomic number 35. It is a volatile red-brown liquid at room temperature that evaporates readily to form a similarly coloured vapour. Its properties are intermediate between th ...
were reported with resistivities as low as 8 ohm·cm. In 1973, it was discovered that a combination of tetracyanoquinodimethane (TCNQ) and tetrathiafulvalene (TTF) forms a strong charge-transfer complex referred to as ''TTF-TCNQ''. The solid shows almost metallic electrical conductance and was the first-discovered purely organic conductor. In a TTF-TCNQ crystal, TTF and TCNQ molecules are arranged independently in separate parallel-aligned stacks, and an electron transfer occurs from donor (TTF) to acceptor (TCNQ) stacks. Hence, electrons and electron hole
In physics, chemistry, and electronic engineering, an electron hole (often simply called a hole) is a quasiparticle denoting the lack of an electron at a position where one could exist in an atom or crystal structure, atomic lattice. Since in ...
s are separated and concentrated in the stacks and can traverse in a one-dimensional direction along the TCNQ and TTF columns, respectively, when an electric potential is applied to the ends of a crystal in the stack direction.
Superconductivity
Superconductivity is a set of physical properties observed in superconductors: materials where Electrical resistance and conductance, electrical resistance vanishes and Magnetic field, magnetic fields are expelled from the material. Unlike an ord ...
is exhibited by tetramethyl-tetraselenafulvalene-hexafluorophosphate (TMTSF2PF6), which is a semi-conductor at ambient conditions, shows superconductivity at low temperature
Temperature is a physical quantity that quantitatively expresses the attribute of hotness or coldness. Temperature is measurement, measured with a thermometer. It reflects the average kinetic energy of the vibrating and colliding atoms making ...
(critical temperature
Critical or Critically may refer to:
*Critical, or critical but stable, medical states
**Critical, or intensive care medicine
*Critical juncture, a discontinuous change studied in the social sciences.
*Critical Software, a company specializing in ...
) and high pressure
Pressure (symbol: ''p'' or ''P'') is the force applied perpendicular to the surface of an object per unit area over which that force is distributed. Gauge pressure (also spelled ''gage'' pressure)The preferred spelling varies by country and eve ...
: 0.9 K and 12 k bar. Critical current densities in these complexes are very small.
Mechanistic implications
Many reactions involving nucleophiles attacking electrophiles can be usefully assessed from the perspective of an incipient charge-transfer complex. Examples include electrophilic aromatic substitution, the addition of Grignard reagents to ketones, and brominolysis of metal-alkyl bonds.See also
*Exciplex
An excimer (originally short for excited dimer) is a short-lived polyatomic molecule formed from two species that do not form a stable molecule in the ground state. In this case, formation of molecules is possible only if such atom is in an elec ...
– a special case where one of the molecules is in an excited state
* Organic semiconductor
Organic semiconductors are solids whose building blocks are pi-bonded molecules or polymers made up by carbon and hydrogen atoms and – at times – heteroatoms such as nitrogen, sulfur and oxygen. They exist in the form of molecular crystals o ...
* Organic superconductor
References
Historical sources
* Y. Okamoto and W. Brenner ''Organic Semiconductors'', Rheinhold (1964) * {{DEFAULTSORT:Charge-Transfer Complex Physical organic chemistry Molecular electronics Organic semiconductors